Acetyl-CoA and the regulation of metabolism: mechanisms and consequences - PubMed (original) (raw)
Review
Acetyl-CoA and the regulation of metabolism: mechanisms and consequences
Lei Shi et al. Curr Opin Cell Biol. 2015 Apr.
Abstract
Acetyl-CoA represents a key node in metabolism due to its intersection with many metabolic pathways and transformations. Emerging evidence reveals that cells monitor the levels of acetyl-CoA as a key indicator of their metabolic state, through distinctive protein acetylation modifications dependent on this metabolite. We offer the following conceptual model for understanding the role of this sentinel metabolite in metabolic regulation. High nucleocytosolic acetyl-CoA amounts are a signature of a 'growth' or 'fed' state and promote its utilization for lipid synthesis and histone acetylation. In contrast, under 'survival' or 'fasted' states, acetyl-CoA is preferentially directed into the mitochondria to promote mitochondrial-dependent activities such as the synthesis of ATP and ketone bodies. Fluctuations in acetyl-CoA within these subcellular compartments enable the substrate-level regulation of acetylation modifications, but also necessitate the function of sirtuin deacetylases to catalyze removal of spontaneous modifications that might be unintended. Thus, understanding the sources, fates, and consequences of acetyl-CoA as a carrier of two-carbon units has started to reveal its underappreciated but profound influence on the regulation of numerous life processes.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures
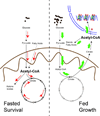
Figure 1. Schematic model proposing a general logic of acetyl-CoA utilization under fed versus fasted or growth versus survival states
Under fed or growth states, acetyl-CoA is directed out of the mitochondria and to the cytosol and nucleus for use in lipid synthesis or histone acetylation. Nucleocytosolic amounts of acetyl-CoA increase relative to mitochondrial amounts. Under fasted or survival states, acetyl-CoA is channeled into the mitochondria for synthesis of ATP and ketone bodies. Mitochondrial amounts of acetyl-CoA increase relative to nucleocytosolic amounts. Fatty acid oxidation significantly increases mitochondrial acetyl-CoA.
Figure 2. Dynamic acetylation and deacetylation of proteins
(A) The acetylation of proteins may be catalyzed by acetyltransferase enzymes or can occur spontaneously through reaction with acetyl-CoA directly. Deacetylase enzymes catalyze the removal of acetylation modifications. Liberated acetate can be converted back to acetyl-CoA. (B) Sirtuins utilize NAD+ to catalyze protein deacetylation, yielding nicotinamide and O-acetyl-ADP-ribose (OAADPr). The removal of aberrant acetylation or acylation modifications may restore protein function. Abbreviations: HAT (histone acetyltransferase), KAT (lysine acetyltransferase), HDAC (histone deacetylase), KDAC (lysine deacetylase), Ac (acetate), CoA (Coenzyme A).
References
- Wellen KE, Thompson CB. A two-way street: reciprocal regulation of metabolism and signalling. Nat Rev Mol Cell Biol. 2012;13:270–276. -PubMed
- Hardie DG. AMPK: positive and negative regulation, and its role in whole-body energy homeostasis. Curr Opin Cell Biol. 2014;33C:1–7. -PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources