Collateral pathways from the ventromedial hypothalamus mediate defensive behaviors - PubMed (original) (raw)
Collateral pathways from the ventromedial hypothalamus mediate defensive behaviors
Li Wang et al. Neuron. 2015.
Abstract
The ventromedial hypothalamus (VMH) was thought to be essential for coping with threat, although its circuit mechanism remains unclear. To investigate this, we optogenetically activated steroidogenic factor 1 (SF1)-expressing neurons in the dorsomedial and central parts of the VMH (VMHdm/c), and observed a range of context-dependent somatomotor and autonomic responses resembling animals' natural defensive behaviors. By activating independent pathways emanating from the VMHdm/c, we demonstrated that VMHdm/c projection to the dorsolateral periaqueductal gray (dlPAG) induces inflexible immobility, while the VMHdm/c to anterior hypothalamic nucleus (AHN) pathway promotes avoidance. Consistent with the behavior changes induced by VMH to AHN pathway activation, direct activation of the AHN elicited avoidance and escape jumping, but not immobility. Retrograde tracing studies revealed that nearly 50% of PAG-projecting VMHdm/c neurons send collateral projection to the AHN and vice versa. Thus, VMHdm/c neurons employ a one-to-many wiring configuration to orchestrate multiple aspects of defensive behaviors.
Copyright © 2015 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no conflict-of-interest.
Figures
Figure 1. Optogenetic stimulation of SF1 neurons in the VMHdm/c induced spiking activity
(A) GFP (green) staining reveals SF1 positive cells in the VMH of an SF1:CRE × RCE:loxP mouse. Blue: NeuN. Scale bar: 500 μm. (B) Virally expressed ChR2-EYFP (green) is concentrated in the VMHdm/c. Blue: DAPI. Scale bar: 200 μm. (C) The ratio of accumulated pixel values of EYFP expression (green channel) in the VMHdm/c relative to in the VMHvl (N=8) is significantly more than 1 (Student's t test). Error bar: standard error. Each dot represents data from one animal. (D) Optrode (yellow arrow) track in the VMHdm/c. Green: ChR2-EYFP; blue: DAPI; red: DiI painted on the optrode. Scale bar: 200 μm. (E) 50 overlaid electrophysiological traces aligned to the 1-ms light onset. Red arrow: spontaneous spike; black arrow: evoked spikes; blue arrow: stimulation artifacts. (F) Spiking probability (number of spikes/number of trials) aligned to the light onset. Bin size = 0.5 ms. Blue shading: light on.
Figure 2. Optogenetic stimulation of SF1 neurons in VMHdm/c-induced defensive behaviors
(A and B) The average percentage of time that the animal spent immobile (A) and the average percentage of trials with running and jumping events (B) were significantly higher during light delivery (N=31). (C) Increased light intensity increased the percentage of trials with running and jumping events but did not significantly change the time of immobility (N=8). (D) Examples of tracking traces with 10-Hz (5 trials) and 20-Hz (6 trials) light pulses. Blue and red traces: light on and off periods, respectively. (E) PSTHs of locomotion velocity aligned to the 10-Hz (top) and 20-Hz (bottom) stimulation onset (left blue shading) and offset (right blue shading). (F–H) Increases in light-pulse frequency decreased the time spent immobile (F), increased the percentage of trials with running and jumping events (G), and shortened latency to running and jumping but did not change latency to immobility (H). N=12. (I) Representative tracking traces during the hiding box test. Different colors represent different stimulation trials. The white dot indicates the animal's position at light onset. (J–K) Light stimulation shortened latency to return to the box (J) and increased total percentage of time in the box (K). N=6. Gray and red lines indicate data of individuals and population average, respectively. Error bars: standard errors. Paired t-test: *p<0.05, **p<0.01, ***p<0.001. See also Figure S1-2 and Movie S1-2.
Figure 3. VMH SF1 neuron stimulation induced autonomic responses
(A) PSTHs of the pupil diameter aligned to light onset. Gray lines indicate individual trials. Blue and red lines indicate the average response curve under low- (blue) and high- (red) intensity light. Upper right corner shows a representative image with computer detected pupil (yellow circle). (B) Pupil diameter aligned to spontaneous running (N=102 trials; 5 animals). (C–D) Significantly increased pupil diameter (C) and breathing rate (D) during low (N=4) or high (N=6) intensity light stimulation. Colored squares show individual animals. (E) Raw ECG trace with computer detected heart beats (red dots). (F) PSTHs of heart rate aligned to light onset. Note opposite HR response during low- (left) and high- (right) intensity light. (G, H) HR (top) and HRV (bottom) changes during low- and high-intensity light stimulation of an example animal. Gray: individual trials. Red: average. (I, J) HR (top) and HRV (bottom) changes of all 6 animals during low- (left) and high- (right) intensity light stimulation. Red line indicates response during light is significantly different from before or after period (FDR adjusted p< 0.05, paired t-test across trials for each animal). (K) PSTHs of HR aligned to the onset of running events during either low (top, N=11) or high (bottom, N=22) intensity light stimulation. See also Movie S3.
Figure 4. VMH SF1 neuron activation promoted avoidance
(A) Experimental schematic of real time place preference test. (B) Tracking traces from one test session. (C) Time spent in safe and stimulation chamber during each entry of the session shown in (B). Red arrow indicates the first entry into the stimulation chamber during test. (D–F) Stimulation of the VMHdm/c decreased the duration in the stimulation chamber per entry (D), increased the duration in the safe chamber per entry (E), and decreased the percentage of time in the stimulation chamber (F). (G) The animal spent a smaller percentage of time immobile during and between stimulation compared with the pretest. (G) Percentage of time immobile during the last two entries to the stimulation chamber was much less than that during the first two entries. D–H: N=18. (I) Schematics of punishment operant conditioning. (J) Tracking traces during sham (left) and VMH (right) stimulation. Yellow arrows indicate nose ports. Note the limited movement during light stimulation (blue) and increased movement along the perimeter of the cage (green) after stimulation. (K) Locomotion velocity (mean ± SEM) aligned to the 2-s light stimulation (light blue) onset in the home cage (N=66 trials from one animal). (L) Increased immobility during VMH stimulation in comparison to sham stimulation. (M) The poking number significantly decreased with VMH stimulation but returned to baseline one day after stimulation. (N) Animals moved away from the poker after VMH stimulation. N=12 for K–N. Gray and red lines indicate results from individual animals and population average, respectively. Error bars: standard errors. Paired _t-_test. *p<0.05, ***p<0.001. See also Movie S4.
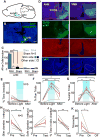
Figure 5. VMH→AHN and VMH→PAG terminal stimulation induced defense-like motor responses and avoidance
(A) Schematic representation of VMH→AHN and VMH→PAG terminal stimulation. Image adopted from
http://connectivity.brain-map.org/
experiment ID: 182337561. (B) Native expression of EYFP in an SF1:CRE mouse bilaterally injected with AAV2 expressing CRE-dependent ChR2-EFYP. (C–F) EYFP staining reveals the terminal fields of VMHdm/c SF1 neurons (green). Scale bars in B–F: 1 mm. (G–H) An example of fiber terminals (green) and cannula/optic fiber tracks (yellow arrows) in the AHN (G) and the PAG (H). Blue: DAPI. Scale bar: 200 μm. (I–K) Stimulating the VMH→PAG pathway significantly increased immobility (I, left) but not running (J, left) or jumping (K, left) whereas VMH→AHN axon terminal stimulation increased immobility (I, right), running (J, right) and jumping (K, right). (L) VMH→AHN terminal stimulation (right) decreased the percentage of time spent in stimulation chamber in all animals, whereas the VMH→PAG terminal stimulation (left) induced avoidance only in 5/9 animals (light blue lines). Dashed line indicates 40% time in stimulation chamber. (M)VMH→AHN stimulation (right) decreased immobility while VMH→PAG stimulation (left) induced heterogeneous changes in immobility. Gray and red lines indicate individual result and population average. (PAG: N=9; AHN: N=7). Error bars: standard errors. Paired t test *p<0.05, **p<0.01, ***p<0.001. See also Figure S3 and 4 and Movie S5.
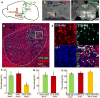
Figure 6. VMHdm/c neurons sent collateral projections to the AHN and PAG
(A) Retrograde tracing experiment. (B–C) Injection sites in the AHN (B) and the dlPAG (C) of one animal. Scale bars: 0.5 mm (D) Retrogradely labeled neurons in the VMH from the PAG (green) and the AHN (red), co-stained with SF1 antibody (blue). Scale bar: 100 μm. (E) Individual channels and merged view of the boxed area in (D). White arrows indicate triply labeled neurons. Scale bar: 20 μm. (F) Percentages of PAG (green), AHN (red), and dual projecting neurons (yellow) among all the VMHdm/c neurons. (G) Percentages of dual projecting neurons in PAG-projecting (green) or AHN-projecting (red) populations. (H) Percentages of SF1-positive neurons in the PAG-, AHN-, or dual-projecting populations. N=5 for F and G. N=3 for H. Error bars: standard errors. See also Figure S5 and Table S1.
Figure 7. VMH→PAG and VMH→AHN activation with VMH blocking induced distinctive aspects of defensive behaviors
(A) Schematic representation of the experiment. (B) Before blocking, VMH stimulation induced avoidance to the stimulation chamber in all animals (N = 11). (C) Bupivacaine injection blocked VMH stimulation induced avoidance in 6 of 11 animals (red dots) but had no effect on the remaining 5 animals (blue dots). (D) VMH→AHN stimulation (right) but not VMH→PAG stimulation (left) induced avoidance to the stimulation chamber in the RTPP test. (E) VMH→PAG (left) but not VMH→AHN (right) stimulation increased immobility. (F) The immobility induced by VMH→ PAG stimulation (left) is similar and high in the first two and last two entries to the stimulation chamber, whereas 5/6 animals showed low immobility during later VMH→AHN stimulation trials. (G) In the operant punishment test, VMH→PAG stimulation induced higher level of immobility than VMH→AHN or sham stimulation. (H) The animal showed a trend (p = 0.06) to move away from the poker after VMH→AHN stimulation but not after VMH→PAG stimulation. (I) VMH→AHN activation reduced poke number, while VMH→PAG activation did not. Gray and red lines indicate data of individual animals and population average, respectively. Error bars: standard errors. PAG: N = 5; AHN: N = 6; Sham: N = 6. Paired t test in D–F; Unpaired t test in G–I. *p<0.05, ***p<0.001 See also Figure S6-7.
Figure 8. AHN stimulation induced jump and avoidance but no immobility
(A) Schematic showing injection and stimulation sites. (B) A coronal section with cannula track and ChR2-EYFP expression (green). Scale bar: 1 mm. (C) Average number of Fos-expressing cells per section at the AHN and VMH after AHN stimulation with light. Two-way ANOVA showed significant difference between stimulation side, stimulation condition and their interaction term for AHN (p<0.01 for all terms) but not VMH (p>0.1 for all terms). Unpaired t-test**:** *p<0.05, ***p<0.001. (D) Representative images showing the ChR2-EYFP (green) and Fos expression (red) at AHN (left) and VMH (right) after AHN light stimulation. Dashed lines mark the areas for Fos counting. Scale bar: 500 μm. (E–G) AHN light stimulation did not change immobility level (E) but increased the percentage of trials with running (F) and jumping events (G). (H–J) In the RTPP test, AHN stimulation decreased the time in the stimulation chamber per entry (H), did not change the percentage of time in the safe chamber per entry (I), and decreased the total percentage of time in the stimulation chamber (J). (K) AHN stimulation decreased immobility in the RTPP test. N = 5. Paired t test for E–K. *p<0.05, **p<0.01, ***p<0.001.
Comment in
- Behavioural neuroscience: Going on the defensive.
Yates D. Yates D. Nat Rev Neurosci. 2015 May;16(5):246-7. doi: 10.1038/nrn3952. Epub 2015 Apr 10. Nat Rev Neurosci. 2015. PMID: 25858005 No abstract available.
Similar articles
- Fos-like immunoreactive neurons following electrical stimulation of the dorsal periaqueductal gray at freezing and escape thresholds.
Vianna DM, Borelli KG, Ferreira-Netto C, Macedo CE, Brandão ML. Vianna DM, et al. Brain Res Bull. 2003 Dec 30;62(3):179-89. doi: 10.1016/j.brainresbull.2003.09.010. Brain Res Bull. 2003. PMID: 14698351 - High-frequency stimulation of the dorsolateral periaqueductal gray and ventromedial hypothalamus fails to inhibit panic-like behaviour.
Lim LW, Blokland A, Visser-Vandewalle V, Vlamings R, Sesia T, Steinbusch H, Schruers K, Griez E, Temel Y. Lim LW, et al. Behav Brain Res. 2008 Nov 21;193(2):197-203. doi: 10.1016/j.bbr.2008.05.020. Epub 2008 Jun 4. Behav Brain Res. 2008. PMID: 18582503 - Neuroanatomical and neurochemical mechanisms underlying amygdaloid control of defensive rage behavior in the cat.
Shaikh MB, Siegel A. Shaikh MB, et al. Braz J Med Biol Res. 1994 Dec;27(12):2759-79. Braz J Med Biol Res. 1994. PMID: 7549999 Review. - The medial hypothalamic defensive system: hodological organization and functional implications.
Canteras NS. Canteras NS. Pharmacol Biochem Behav. 2002 Mar;71(3):481-91. doi: 10.1016/s0091-3057(01)00685-2. Pharmacol Biochem Behav. 2002. PMID: 11830182 Review.
Cited by
- Encoding innately recognized odors via a generalized population code.
Qiu Q, Wu Y, Ma L, Yu CR. Qiu Q, et al. Curr Biol. 2021 May 10;31(9):1813-1825.e4. doi: 10.1016/j.cub.2021.01.094. Epub 2021 Mar 1. Curr Biol. 2021. PMID: 33651991 Free PMC article. - Posterior amygdala regulates sexual and aggressive behaviors in male mice.
Yamaguchi T, Wei D, Song SC, Lim B, Tritsch NX, Lin D. Yamaguchi T, et al. Nat Neurosci. 2020 Sep;23(9):1111-1124. doi: 10.1038/s41593-020-0675-x. Epub 2020 Jul 27. Nat Neurosci. 2020. PMID: 32719562 Free PMC article. - Chemo- and optogenetic activation of hypothalamic _Foxb1_-expressing neurons and their terminal endings in the rostral-dorsolateral PAG leads to tachypnea, bradycardia, and immobility.
Cola RB, Roccaro-Waldmeyer DM, Naim S, Babalian A, Seebeck P, Alvarez-Bolado G, Celio MR. Cola RB, et al. Elife. 2024 Feb 1;12:RP86737. doi: 10.7554/eLife.86737. Elife. 2024. PMID: 38300670 Free PMC article. - Neural and Genetic Basis of Evasion, Approach and Predation.
Park S, Ryoo J, Kim D. Park S, et al. Mol Cells. 2022 Feb 28;45(2):93-97. doi: 10.14348/molcells.2022.2032. Mol Cells. 2022. PMID: 35236784 Free PMC article. Review. - Editorial: Current Views of Hypothalamic Contributions to the Control of Motivated Behaviors.
Hahn JD, Fink G, Kruk MR, Stanley BG. Hahn JD, et al. Front Syst Neurosci. 2019 Aug 13;13:32. doi: 10.3389/fnsys.2019.00032. eCollection 2019. Front Syst Neurosci. 2019. PMID: 31456668 Free PMC article. No abstract available.
References
- Akesson TR, Ulibarri C, Truitt S. Divergent axon collaterals originate in the estrogen receptive ventromedial nucleus of hypothalamus in the rat. J Neurobiol. 1994;25:406–414. - PubMed
- Bandler R, Keay KA. Columnar organization in the midbrain periaqueductal gray and the integration of emotional expression. Prog Brain Res. 1996;107:285–300. - PubMed
- Bard P. A diencephalic mechanism for the expression of rage with special reference to the sympathetic nervous system. Am J Physiol. 1928;84:490–515.
- Blanchard RJ, Hebert MA, Ferrari PF, Palanza P, Figueira R, Blanchard DC, Parmigiani S. Defensive behaviors in wild and laboratory (Swiss) mice: the mouse defense test battery. Physiol Behav. 1998;65:201–209. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases