Acute responses to opioidergic blockade as a biomarker of hedonic eating among obese women enrolled in a mindfulness-based weight loss intervention trial - PubMed (original) (raw)
Randomized Controlled Trial
doi: 10.1016/j.appet.2015.04.062. Epub 2015 Apr 27.
Robert H Lustig 2, Rashida R Brown 3 4, Michael Acree 1, Peter Bacchetti 5, Patricia J Moran 1, Mary Dallman 3, Barbara Laraia 5, Nancy Adler 3, Frederick M Hecht # 1, Jennifer Daubenmier # 1, Elissa S Epel # 1 3
Affiliations
- PMID: 25931433
- PMCID: PMC4485926
- DOI: 10.1016/j.appet.2015.04.062
Randomized Controlled Trial
Acute responses to opioidergic blockade as a biomarker of hedonic eating among obese women enrolled in a mindfulness-based weight loss intervention trial
Ashley E Mason et al. Appetite. 2015 Aug.
Abstract
There are currently no commonly used or easily accessible 'biomarkers' of hedonic eating. Physiologic responses to acute opioidergic blockade, indexed by cortisol changes and nausea, may represent indirect functional measures of opioid-mediated hedonic eating drive and predict weight loss following a mindfulness-based intervention for stress eating. In the current study, we tested whether cortisol and nausea responses induced by oral ingestion of an opioidergic antagonist (naltrexone) correlated with weight and self-report measures of hedonic eating and predicted changes in these measures following a mindfulness-based weight loss intervention. Obese women (N = 88; age = 46.7 ± 13.2 years; BMI = 35.8 ± 3.8) elected to complete an optional sub-study prior to a 5.5-month weight loss intervention with or without mindfulness training. On two separate days, participants ingested naltrexone and placebo pills, collected saliva samples, and reported nausea levels. Supporting previous findings, naltrexone-induced cortisol increases were associated with greater hedonic eating (greater food addiction symptoms and reward-driven eating) and less mindful eating. Among participants with larger cortisol increases (+1 SD above mean), mindfulness participants (relative to control participants) reported greater reductions in food addiction symptoms, b = -0.95, SE(b) = 0.40, 95% CI [-1.74, -0.15], p = .021. Naltrexone-induced nausea was marginally associated with reward-based eating. Among participants who endorsed naltrexone-induced nausea (n = 38), mindfulness participants (relative to control participants) reported greater reductions in food addiction symptoms, b = -1.00, 95% CI [-1.85, -0.77], p = .024, and trended toward reduced reward-based eating, binge eating, and weight, post-intervention. Single assessments of naltrexone-induced cortisol increases and nausea responses may be useful time- and cost-effective biological markers to identify obese individuals with greater opioid-mediated hedonic eating drive who may benefit from weight loss interventions with adjuvant mindfulness training that targets hedonic eating.
Keywords: Cortisol; Hedonic eating; Mindfulness intervention; Naltrexone; Nausea.
Published by Elsevier Ltd.
Figures
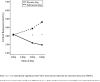
Figure 1
Naltrexone-induced cortisol increases from 1 PM to 4 PM on placebo and naltrexone days at baseline.
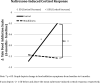
Figure 2
Interaction of naltrexone-induced cortisol response and treatment arm (active control vs. mindfulness) as a predictor of changes in self-reported food addiction symptoms.
Figure 3
Interaction of naltrexone-induced nausea (present vs. absent) and treatment arm (active control vs. mindfulness) as a predictor of changes in self-report measures of food addiction and binge eating symptoms.
References
- Adam TC, Epel ES. Stress, eating and the reward system. Physiology & Behavior. 2007;91(4):449–458. - PubMed
- American Psychological Association . Diagnostic and statistical manual of mental disorders: DSM-IV-TR®. American Psychiatric Pub.; 2000.
Publication types
MeSH terms
Substances
Grants and funding
- K24AT007827/AT/NCCIH NIH HHS/United States
- 1U01HL097973/HL/NHLBI NIH HHS/United States
- P01 AT005013/AT/NCCIH NIH HHS/United States
- K24 AT007827/AT/NCCIH NIH HHS/United States
- P30 DK098722/DK/NIDDK NIH HHS/United States
- 1P01AT005013/AT/NCCIH NIH HHS/United States
- K01 AT004199/AT/NCCIH NIH HHS/United States
- K01AT004199/AT/NCCIH NIH HHS/United States
- U01 HL097973/HL/NHLBI NIH HHS/United States
- T32 AT003997/AT/NCCIH NIH HHS/United States
- UL1 TR000004/TR/NCATS NIH HHS/United States
- T32AT003997/AT/NCCIH NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical