Repeated nebulisation of non-viral CFTR gene therapy in patients with cystic fibrosis: a randomised, double-blind, placebo-controlled, phase 2b trial - PubMed (original) (raw)
Clinical Trial
doi: 10.1016/S2213-2600(15)00245-3. Epub 2015 Jul 3.
David K Armstrong 2, Deborah Ashby 3, Katie J Bayfield 3, Diana Bilton 4, Emily V Bloomfield 3, A Christopher Boyd 5, June Brand 5, Ruaridh Buchan 6, Roberto Calcedo 7, Paula Carvelli 3, Mario Chan 3, Seng H Cheng 8, D David S Collie 9, Steve Cunningham 2, Heather E Davidson 5, Gwyneth Davies 3, Jane C Davies 3, Lee A Davies 10, Maria H Dewar 6, Ann Doherty 5, Jackie Donovan 4, Natalie S Dwyer 3, Hala I Elgmati 6, Rosanna F Featherstone 3, Jemyr Gavino 3, Sabrina Gea-Sorli 3, Duncan M Geddes 4, James S R Gibson 5, Deborah R Gill 10, Andrew P Greening 6, Uta Griesenbach 3, David M Hansell 4, Katharine Harman 3, Tracy E Higgins 3, Samantha L Hodges 3, Stephen C Hyde 10, Laura Hyndman 5, J Alastair Innes 6, Joseph Jacob 4, Nancy Jones 4, Brian F Keogh 4, Maria P Limberis 7, Paul Lloyd-Evans 11, Alan W Maclean 5, Michelle C Manvell 3, Dominique McCormick 10, Michael McGovern 6, Gerry McLachlan 9, Cuixiang Meng 3, M Angeles Montero 4, Hazel Milligan 6, Laura J Moyce 10, Gordon D Murray 12, Andrew G Nicholson 4, Tina Osadolor 4, Javier Parra-Leiton 5, David J Porteous 5, Ian A Pringle 10, Emma K Punch 3, Kamila M Pytel 3, Alexandra L Quittner 13, Gina Rivellini 3, Clare J Saunders 3, Ronald K Scheule 8, Sarah Sheard 4, Nicholas J Simmonds 4, Keith Smith 11, Stephen N Smith 3, Najwa Soussi 3, Samia Soussi 3, Emma J Spearing 3, Barbara J Stevenson 5, Stephanie G Sumner-Jones 10, Minna Turkkila 3, Rosa P Ureta 3, Michael D Waller 3, Marguerite Y Wasowicz 3, James M Wilson 7, Paul Wolstenholme-Hogg 8; UK Cystic Fibrosis Gene Therapy Consortium
Affiliations
- PMID: 26149841
- PMCID: PMC4673100
- DOI: 10.1016/S2213-2600(15)00245-3
Clinical Trial
Repeated nebulisation of non-viral CFTR gene therapy in patients with cystic fibrosis: a randomised, double-blind, placebo-controlled, phase 2b trial
Eric W F W Alton et al. Lancet Respir Med. 2015 Sep.
Erratum in
- Corrections.
[No authors listed] [No authors listed] Lancet Respir Med. 2015 Sep;3(9):e33. doi: 10.1016/S2213-2600(15)00314-8. Epub 2015 Sep 6. Lancet Respir Med. 2015. PMID: 26380890 Free PMC article. No abstract available.
Abstract
Background: Lung delivery of plasmid DNA encoding the CFTR gene complexed with a cationic liposome is a potential treatment option for patients with cystic fibrosis. We aimed to assess the efficacy of non-viral CFTR gene therapy in patients with cystic fibrosis.
Methods: We did this randomised, double-blind, placebo-controlled, phase 2b trial in two cystic fibrosis centres with patients recruited from 18 sites in the UK. Patients (aged ≥12 years) with a forced expiratory volume in 1 s (FEV1) of 50-90% predicted and any combination of CFTR mutations, were randomly assigned, via a computer-based randomisation system, to receive 5 mL of either nebulised pGM169/GL67A gene-liposome complex or 0.9% saline (placebo) every 28 days (plus or minus 5 days) for 1 year. Randomisation was stratified by % predicted FEV1 (<70 vs ≥70%), age (<18 vs ≥18 years), inclusion in the mechanistic substudy, and dosing site (London or Edinburgh). Participants and investigators were masked to treatment allocation. The primary endpoint was the relative change in % predicted FEV1. The primary analysis was per protocol. This trial is registered with ClinicalTrials.gov, number NCT01621867.
Findings: Between June 12, 2012, and June 24, 2013, we randomly assigned 140 patients to receive placebo (n=62) or pGM169/GL67A (n=78), of whom 116 (83%) patients comprised the per-protocol population. We noted a significant, albeit modest, treatment effect in the pGM169/GL67A group versus placebo at 12 months' follow-up (3.7%, 95% CI 0.1-7.3; p=0.046). This outcome was associated with a stabilisation of lung function in the pGM169/GL67A group compared with a decline in the placebo group. We recorded no significant difference in treatment-attributable adverse events between groups.
Interpretation: Monthly application of the pGM169/GL67A gene therapy formulation was associated with a significant, albeit modest, benefit in FEV1 compared with placebo at 1 year, indicating a stabilisation of lung function in the treatment group. Further improvements in efficacy and consistency of response to the current formulation are needed before gene therapy is suitable for clinical care; however, our findings should also encourage the rapid introduction of more potent gene transfer vectors into early phase trials.
Funding: Medical Research Council/National Institute for Health Research Efficacy and Mechanism Evaluation Programme.
Copyright © 2015 Alton et al. Open Access article distributed under the terms of CC BY. Published by Elsevier Ltd.. All rights reserved.
Figures
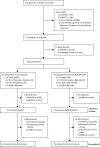
Figure 1
Trial profile Numbers of patients in the intention-to-treat population are unequal because of the 2:1 allocation in the mechanistic substudy. FEV1=forced expiratory volume in 1 s.
Figure 2
Timecourse of the primary outcome response to either placebo or pGM169/GL67A (A) and the individual patient responses in the pGM169/GL67A (B) and placebo (C) groups Error bars in panel A show the standard error of the mean. Primary outcome measurements were taken at each treatment visit before administration of study drugs. Pre and post values indicate the mean of two measurements at the respective timepoints. Positive values in panels B and C show an improvement. FEV1=forced expiratory volume in 1 s.
Figure 3
Forest plot showing secondary outcome responses to placebo or pGM169/GL67A Data are mean (SD) or mean (95% CI), unless otherwise indicated. The size of the circles is proportional to the number of patients represented and the error bars show 95% CIs. Values shown for FEV1 are the relative change in the % predicted FEV1. To allow results from different endpoints to be plotted on a common scale, the estimated treatment effects were standardised to be presented as multiples of the underlying SD (standardised treatment effect). FEV1=forced expiratory volume in 1 s. MEF25–75=mid-expiratory flow between 25% and 75% of FVC. KCOc=diffusion capacity of the alveolar capillary membrane, corrected for haemoglobin concentrations. TLCOc=transfer factor of the lung for carbon monoxide, corrected for haemoglobin concentrations. *Refers to scores from the Cystic Fibrosis Questionnaire-Revised.
Figure 4
Assessment of DNA from bronchial brushings in the placebo (n=7) and pGM169/GL67A (n=14) subgroups (A) and the response of the bronchial epithelium to perfusion with a zero chloride solution containing isoprenaline 10 μM (B, C) Horizontal bars show median values. Each circle in panel A represents an individual patient. Each symbol in panels B and C shows the change in response from trial start to finish for the relevant treatment in an individual patient. Of the 16 participants in the bronchoscopy subgroup, 15 individuals had post-dose bronchoscopies, of whom 14 individuals generated samples for DNA and mRNA molecular analysis. The plotted value in panel B is the mean of all interpretable recordings (range 1–3), and in panel C is the most negative value obtained from all interpretable recordings, at each timepoint for that patient. A more negative value is in the non-cystic fibrosis direction. LOQ=limit of quantification, PBNQ=positive but not quantifiable.
Comment in
- A new phase of CFTR treatment for cystic fibrosis?
Bell SC. Bell SC. Lancet Respir Med. 2015 Sep;3(9):662-663. doi: 10.1016/S2213-2600(15)00282-9. Epub 2015 Jul 22. Lancet Respir Med. 2015. PMID: 26208999 No abstract available.
References
- Griesenbach U, Alton EW. Moving forward: cystic fibrosis gene therapy. Hum Mol Genet. 2013;22:R52–R58. -PubMed
- Cystic Fibrosis Trust UK Cystic Fibrosis Registry annual data report 2013. 2014. http://www.cysticfibrosis.org.uk/media/598466/annual-data-report-2013-ju... (accessed April 26, 2015).
- Gill DR, Hyde SC. Delivery of genes into the CF airway. Thorax. 2014;69:962–964. -PubMed
- Davies LA, Nunez-Alonso GA, McLachlan G, Hyde SC, Gill DR. Aerosol delivery of DNA/liposomes to the lung for cystic fibrosis gene therapy. Hum Gene Ther Clin Dev. 2014;25:97–107. -PubMed
- McLachlan G, Davidson H, Holder E. Pre-clinical evaluation of three non-viral gene transfer agents for cystic fibrosis after aerosol delivery to the ovine lung. Gene Ther. 2011;18:996–1005. -PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical