Characteristics of funding of clinical trials: cross-sectional survey and proposed guidance - PubMed (original) (raw)
. 2017 Oct 5;7(10):e015997.
doi: 10.1136/bmjopen-2017-015997.
Nahla Jouni 2, Eliane A Abou-Jaoude 3, Divina Justina Hasbani 4, Elias A Abou-Jaoude 5, Luciane Cruz Lopes 6, Mariam Khaldieh 7, Mira Zein Hammoud 8, Mounir Al-Gibbawi 4, Sirine Anouti 9, Gordon Guyatt 10, Elie A Akl 1 10 11
Affiliations
- PMID: 28982811
- PMCID: PMC5639984
- DOI: 10.1136/bmjopen-2017-015997
Characteristics of funding of clinical trials: cross-sectional survey and proposed guidance
Maram B Hakoum et al. BMJ Open. 2017.
Abstract
Objectives: To provide a detailed and current characterisation of funding of a representative sample clinical trials. We also aimed to develop guidance for standardised reporting of funding information.
Methods: We addressed the extent to which clinical trials published in 2015 in any of the 119 Core Clinical Journals included a statement on the funding source (eg, whether a not-for-profit organisation was supported by a private-for-profit organisation), type of funding, amount and role of funder. We used a stepwise approach to develop a guidance and an instrument for standardised reporting of funding information.
Results: Of 200 trials, 178 (89%) included a funding statement, of which 171 (96%) reported being funded. Funding statements in the 171 funded trials indicated the source in 100%, amount in 1% and roles of funders in 50%. The most frequent sources were governmental (58%) and private-for-profit (40%). Of 54 funding statements in which the source was a not-for-profit organisation, we found evidence of undisclosed support of those from private-for-profit organisation(s) in 26 (48%). The most frequently reported roles of funders in the 171 funded trials related to study design (42%) and data analysis, interpretation or management (41%). Of 139 randomised controlled trials (RCTs) addressing pharmacological or surgical interventions, 29 (21%) reported information on the supplier of the medication or device. The proposed guidance addresses both the funding information that RCTs should report and the reporting process. Attached to the guidance is a fillable PDF document for use as an instrument for standardised reporting of funding information.
Conclusion: Although the majority of RCTs report funding, there is considerable variability in the reporting of funding source, amount and roles of funders. A standardised approach to reporting of funding information would address these limitations. Future research should explore the implications of funding by not-for-profit organisations that are supported by for-profit organisations.
Keywords: funding; randomised controlled trial; role of funder.
© Article author(s) (or their employer(s) unless otherwise stated in the text of the article) 2017. All rights reserved. No commercial use is permitted unless otherwise expressly granted.
Conflict of interest statement
Competing interests: None declared.
Figures
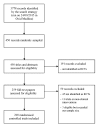
Figure 1
Study flow diagram. RCTs, randomised controlled trials.
Similar articles
- The association of funding source on effect size in randomized controlled trials: 2013-2015 - a cross-sectional survey and meta-analysis.
Falk Delgado A, Falk Delgado A. Falk Delgado A, et al. Trials. 2017 Mar 14;18(1):125. doi: 10.1186/s13063-017-1872-0. Trials. 2017. PMID: 28292317 Free PMC article. - Clinical trial design and dissemination: comprehensive analysis of clinicaltrials.gov and PubMed data since 2005.
Zwierzyna M, Davies M, Hingorani AD, Hunter J. Zwierzyna M, et al. BMJ. 2018 Jun 6;361:k2130. doi: 10.1136/bmj.k2130. BMJ. 2018. PMID: 29875212 Free PMC article. - Impact of industry collaboration on randomised controlled trials in oncology.
Linker A, Yang A, Roper N, Whitaker E, Korenstein D. Linker A, et al. Eur J Cancer. 2017 Feb;72:71-77. doi: 10.1016/j.ejca.2016.11.005. Epub 2016 Dec 24. Eur J Cancer. 2017. PMID: 28027518 Free PMC article. Review. - Reporting of conflicts of interest in meta-analyses of trials of pharmacological treatments.
Roseman M, Milette K, Bero LA, Coyne JC, Lexchin J, Turner EH, Thombs BD. Roseman M, et al. JAMA. 2011 Mar 9;305(10):1008-17. doi: 10.1001/jama.2011.257. JAMA. 2011. PMID: 21386079 Review.
Cited by
- Identifying competing interest disclosures in systematic reviews of surgical interventions and devices: a cross-sectional survey.
Yu J, Su G, Hirst A, Yang Z, Zhang Y, Li Y. Yu J, et al. BMC Med Res Methodol. 2020 Oct 19;20(1):260. doi: 10.1186/s12874-020-01144-2. BMC Med Res Methodol. 2020. PMID: 33076823 Free PMC article. - Challenging traditional research: A synopsis of the National Research Collaborative Meeting (NRCM) in 2017.
National Research Collaborative Meeting 2017 Organising Committee; West Midlands Research Collaborative. National Research Collaborative Meeting 2017 Organising Committee, et al. Int J Surg Protoc. 2019 May 13;15:8-11. doi: 10.1016/j.isjp.2019.03.001. eCollection 2019. Int J Surg Protoc. 2019. PMID: 31851748 Free PMC article. - Transparency of COVID-19-Related Research in Dental Journals.
Sofi-Mahmudi A, Raittio E. Sofi-Mahmudi A, et al. Front Oral Health. 2022 Apr 6;3:871033. doi: 10.3389/froh.2022.871033. eCollection 2022. Front Oral Health. 2022. PMID: 35464778 Free PMC article. - Promoting equity in clinical research: The role of social determinants of health.
Idnay B, Fang Y, Stanley E, Ruotolo B, Chung WK, Marder K, Weng C. Idnay B, et al. J Biomed Inform. 2024 Aug;156:104663. doi: 10.1016/j.jbi.2024.104663. Epub 2024 Jun 4. J Biomed Inform. 2024. PMID: 38838949 - Discontinuation and nonpublication of clinical trials in orthopaedic oncology.
Singh G, Wague A, Arora A, Rao V, Ward D, Barry J. Singh G, et al. J Orthop Surg Res. 2024 Feb 5;19(1):121. doi: 10.1186/s13018-024-04601-6. J Orthop Surg Res. 2024. PMID: 38317223 Free PMC article.
References
- Mugambi MN, Musekiwa A, Lombard M, et al. Association between funding source, methodological quality and research outcomes in randomized controlled trials of synbiotics, probiotics and prebiotics added to infant formula: a systematic review. BMC Med Res Methodol 2013;13:137 10.1186/1471-2288-13-137 - DOI - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources