Dysregulation of neurotrophin signaling in the pathogenesis of Alzheimer disease and of Alzheimer disease in Down syndrome - PubMed (original) (raw)
Review
Dysregulation of neurotrophin signaling in the pathogenesis of Alzheimer disease and of Alzheimer disease in Down syndrome
Xu-Qiao Chen et al. Free Radic Biol Med. 2018 Jan.
Abstract
Neurotrophic factors, including the members of the neurotrophin family, play important roles in the development and maintenance of the nervous system. Trophic factor signals must be transmitted over long distances from axons and dendrites to the cell bodies of neurons. A mode of signaling well suited to the challenge of robust long distance signaling is the signaling endosome. We review the biology of signaling endosomes and the "signaling endosome hypothesis". Evidence for disruption of signaling endosome function in disorders of the nervous system is also reviewed. Changes in endosome structure in Alzheimer disease (AD) and Down syndrome (DS) are present early in these disorders. Data for the APP products responsible are reviewed and the consequent changes in signaling from endosomes discussed. We conclude by pointing to the need for additional studies to explore the biology of signaling endosomes in normal neurons and to elucidate their role in the pathogenesis of neurodegeneration.
Keywords: Alzheimer's disease; Axonal transport deficit; Degeneration; Down syndrome; Endosome enlargement; Neurotrophin; Signaling endosome.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures
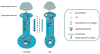
Fig. 1. Endosomal dysfunction in DS compromises signaling of neurotrophic factors
Neurons derive an important source of neurotrophic support from postsynaptic neurons. The latter synthesize and release neurotrophin molecules which then diffuse to presynaptic neurons where they bind to specific cognate receptors on surface membranes. Signaling endosomes are formed when the neurotrophin/receptor complex undergoes endocytosis. This remarkable organelle carries on its cytosolic surface activated isoforms of the downstream signaling pathways activated by the neurotrophin/receptor complex. Once created, signaling endosomes convey signaling to all parts of the presynaptic neuron, including the synapse, the axon, the cell body, and in some cases also dendrites. In normal, healthy neurons, there is a steady flow of signaling endosomes from synapses to cell bodies via dynein-mediated retrograde transport in axons. Long-distance axonal transport allows for the postsynaptic neuron to influence both somal and nuclear events in the presynaptic neuron that serve to ensure the structure and function of the presynaptic neuron, including its ability to synaptically partner with the postsynaptic neuron. Due to increased activation of Rab5, in AD and AD-DS there is enlargement of early endosomes, a change that is correlated with changes in trafficking of signaling endosomes. Though the mechanisms by which APP and its products act are yet to be defined, existing data point to two consequences of disrupted trafficking of signaling endosomes: increased neurotrophin signaling from endosomes in distal axons and deficient transport of signaling endosomes to cell bodies. Studies to explore these changes will be important for understanding their biological significance. Existing data support the view that decreased transport of signaling endosomes to cell bodies results in decreased trophic signaling with resulting atrophy, a hallmark of neurodegeneration in AD and AD-DS. As yet undefined are which signaling pathways are impacted by reduced delivery of signaling endosomes to cell bodies. Nevertheless, that such deficits are present is data showing that p-CREB levels were reduced in neurons overexpressing C99 (62).
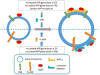
Fig. 2. Schematic illustrating proposed effects of C99 and Fl-APP on regulation of Rab5 activity and early endosome size
The left panel portrays an early endosome from cells expressing normal levels of C99 (above the dotted line), or Fl-APP (below the dotted line). Increased levels of C99 and Fl-APP result from increases in APP gene dose in DS, as well as in FAD due to APP duplication. Increased C99 is also seen in certain other FADs. Increases in C99 and Fl-APP induce activation of Rab5. The right panel portrays the result of increased levels of C99 (above the dotted line) or Fl-APP (below the dotted line). C99 recruits APPL1 to early endosome to stabilize activated Rab5 and induce enlargement of early endosomes. Independent of C99, Fl-APP also induces activation of Rab5 and increases the size of early endosomes. The proteins that link Fl-APP to Rab5 are less well defined. Evidence supports involvement of APP-BP1, but other adaptors may play a role, including APPL1. It is unclear to what extent Rab5 activity is regulated by C99 and Fl-APP in cells that do not overexpress APP, but a gene dose effect of APP has been shown (62).
References
- Ballard C, Mobley W, Hardy J, Williams G, Corbett A. Dementia in Down’s syndrome. The Lancet Neurology. 2016;15(6):622–36. -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- PN1 EY016525/EY/NEI NIH HHS/United States
- R01 AG061151/AG/NIA NIH HHS/United States
- PN2 EY016525/EY/NEI NIH HHS/United States
- R01 NS066072/NS/NINDS NIH HHS/United States
- P01 NS092525/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical