White matter hyperintensities in vascular contributions to cognitive impairment and dementia (VCID): Knowledge gaps and opportunities - PubMed (original) (raw)
Review
doi: 10.1016/j.trci.2019.02.001. eCollection 2019.
Suvarna Alladi 2, Hee-Joon Bae 3, David A Barton 4, Laurel A Beckett 5, Joanne M Bell 6, Sara E Berman 7, Geert Jan Biessels 8, Sandra E Black 9, Isabelle Bos 10, Gene L Bowman 11 12 13, Emanuele Brai 14, Adam M Brickman 15, Brandy L Callahan 16, Roderick A Corriveau 16, Silvia Fossati 17, Rebecca F Gottesman 18, Deborah R Gustafson 19, Vladimir Hachinski 20, Kathleen M Hayden 21, Alex M Helman 22, Timothy M Hughes 23, Jeremy D Isaacs 24, Angela L Jefferson 25, Sterling C Johnson 26, Alifiya Kapasi 27, Silke Kern 28, Jay C Kwon 29, Juraj Kukolja 30, Athene Lee 31, Samuel N Lockhart 32, Anne Murray 33, Katie E Osborn 25, Melinda C Power 34, Brittani R Price 35, Hanneke F M Rhodius-Meester 36, Jacqueline A Rondeau 37, Allyson C Rosen 38, Douglas L Rosene 39, Julie A Schneider 40, Henrieta Scholtzova 41, C Elizabeth Shaaban 42, Narlon C B S Silva 43, Heather M Snyder 44, Walter Swardfager 45, Aron M Troen 46, Susanne J van Veluw 47, Prashanthi Vemuri 48, Anders Wallin 49, Cheryl Wellington 50, Donna M Wilcock 51, Sharon Xiangwen Xie 52, Atticus H Hainsworth 53
Affiliations
- PMID: 31011621
- PMCID: PMC6461571
- DOI: 10.1016/j.trci.2019.02.001
Review
White matter hyperintensities in vascular contributions to cognitive impairment and dementia (VCID): Knowledge gaps and opportunities
Jessica Alber et al. Alzheimers Dement (N Y). 2019.
Abstract
White matter hyperintensities (WMHs) are frequently seen on brain magnetic resonance imaging scans of older people. Usually interpreted clinically as a surrogate for cerebral small vessel disease, WMHs are associated with increased likelihood of cognitive impairment and dementia (including Alzheimer's disease [AD]). WMHs are also seen in cognitively healthy people. In this collaboration of academic, clinical, and pharmaceutical industry perspectives, we identify outstanding questions about WMHs and their relation to cognition, dementia, and AD. What molecular and cellular changes underlie WMHs? What are the neuropathological correlates of WMHs? To what extent are demyelination and inflammation present? Is it helpful to subdivide into periventricular and subcortical WMHs? What do WMHs signify in people diagnosed with AD? What are the risk factors for developing WMHs? What preventive and therapeutic strategies target WMHs? Answering these questions will improve prevention and treatment of WMHs and dementia.
Keywords: Leukoaraiosis; Small vessel disease; Vascular cognitive impairment; Vascular dementia; White matter lesions.
Figures
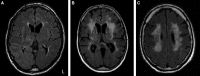
Fig. 1
MRI scans showing typical examples of WMHs of presumed vascular origin. (A) Punctate deep subcortical WMH in the left hemisphere and periventricular caps. This scan is Fazekas grade 1, on the Fazekas scale of WMH severity (range: 0-3). In the right thalamus, a lacune can be seen. (B, C) Two examples of severe confluent WMH. Note that borders between periventricular and deep subcortical WMHs become difficult to define. Scans B and C are Fazekas grade 3. Scans A-C are FLAIR sequences. Figure provided by GJ Biessels. Abbreviations: MRI, magnetic resonance imaging; WMHs, white matter hyperintensities; FLAIR, fluid-attenuated inversion recovery.
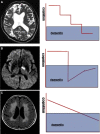
Fig. 2
Conceptual clinical courses leading to vascular dementia. (A) Multi-infarct dementia, stepwise pattern of cognitive decline. (B) Strategic vascular dementia due to a focal lesion in a clinically eloquent site. One-step pattern, with some recovery. (C) WMH-associated subcortical vascular dementia. Slow progression without stepwise pattern. Figure provided by J Kwon. Abbreviation: WMH, white matter hyperintensity.
References
- Prins N.D., Scheltens P. White matter hyperintensities, cognitive impairment and dementia: an update. Nat Rev Neurol. 2015;11:157–165. -PubMed
- Kloppenborg R.P., Nederkoorn P.J., Geerlings M.I., van den Berg E. Presence and progression of white matter hyperintensities and cognition: a meta-analysis. Neurology. 2014;82:2127–2138. -PubMed
Publication types
Grants and funding
- P30 AG010124/AG/NIA NIH HHS/United States
- RF1 AG059421/AG/NIA NIH HHS/United States
- G0800701/1/NC3RS_/National Centre for the Replacement, Refinement and Reduction of Animals in Research/United Kingdom
- U54 HD083211/HD/NICHD NIH HHS/United States
- F31 AG054084/AG/NIA NIH HHS/United States
- R01 NS104127/NS/NINDS NIH HHS/United States
- MR/L023784/2/MRC_/Medical Research Council/United Kingdom
- P30 AG028383/AG/NIA NIH HHS/United States
- P50 AG033514/AG/NIA NIH HHS/United States
- F30 AG054115/AG/NIA NIH HHS/United States
- R01 NS017950/NS/NINDS NIH HHS/United States
- R01 AG054076/AG/NIA NIH HHS/United States
- MR/R005567/1/MRC_/Medical Research Council/United Kingdom
- F32 AG058395/AG/NIA NIH HHS/United States
- T32 GM008692/GM/NIGMS NIH HHS/United States
- R01 AG062572/AG/NIA NIH HHS/United States
- P30 AG049638/AG/NIA NIH HHS/United States
- P30 AG062715/AG/NIA NIH HHS/United States
- P30 AG010129/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical