PDIA1/P4HB is required for efficient proinsulin maturation and ß cell health in response to diet induced obesity - PubMed (original) (raw)
doi: 10.7554/eLife.44528.
Anita Pottekat 1, Juthakorn Poothong 1, Jing Yong 1, Jacqueline Lagunas-Acosta 1, Adriana Charbono 1, Zhouji Chen 1, Donalyn L Scheuner 2, Ming Liu 3, Pamela Itkin-Ansari 4, Peter Arvan 3, Randal J Kaufman 1
Affiliations
- PMID: 31184304
- PMCID: PMC6559792
- DOI: 10.7554/eLife.44528
PDIA1/P4HB is required for efficient proinsulin maturation and ß cell health in response to diet induced obesity
Insook Jang et al. Elife. 2019.
Abstract
Regulated proinsulin biosynthesis, disulfide bond formation and ER redox homeostasis are essential to prevent Type two diabetes. In ß cells, protein disulfide isomerase A1 (PDIA1/P4HB), the most abundant ER oxidoreductase of over 17 members, can interact with proinsulin to influence disulfide maturation. Here we find Pdia1 is required for optimal insulin production under metabolic stress in vivo. ß cell-specific Pdia1 deletion in young high-fat diet fed mice or aged mice exacerbated glucose intolerance with inadequate insulinemia and increased the proinsulin/insulin ratio in both serum and islets compared to wildtype mice. Ultrastructural abnormalities in _Pdia1_-null ß cells include diminished insulin granule content, ER vesiculation and distention, mitochondrial swelling and nuclear condensation. Furthermore, Pdia1 deletion increased accumulation of disulfide-linked high molecular weight proinsulin complexes and islet vulnerability to oxidative stress. These findings demonstrate that PDIA1 contributes to oxidative maturation of proinsulin in the ER to support insulin production and ß cell health.
Keywords: PDIA1; beta cell function; cell biology; disulfide bond formation; glucose homeostasis; mouse; proinsulin maturation; type 2 diabetes.
© 2019, Jang et al.
Conflict of interest statement
IJ, AP, JP, JY, JL, AC, ZC, DS, ML, PI, PA, RK No competing interests declared
Figures
Figure 1.. Conditional ß cell-specific Pdia1 deleted mice were generated with Tamoxifen (Tam) induction.
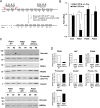
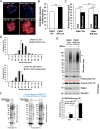
(A) Diagram depicts the generation of Pdia1:RIP-CreERT mice. Mice with floxed Pdia1 alleles (Hahm et al., 2013) were crossed with RIP-CreERT transgenic mice (Dor et al., 2004) and progeny were injected IP with Tam to induce CreERT function and Pdia1 deletion. Control littermate mice with one or two floxed Pdia1 alleles, but not harboring the RIP-CreERT transgene, were injected in parallel with Tam. (B–D) Pdia1 deletion is specific. (B) Total RNA was extracted from islets isolated from female mice at eight wks after Tam injection. mRNA levels were measured by qRT-PCR. Mean ± SEM, n = 3 for each group (p<0.01**). (C) Western blot illustrates expression of Vinculin, PDIA1, BiP, PDIA6, GRP94, Proinsulin, and Insulin in islets isolated from female mice at 14 wks after Tam injection. (D) Quantification of indicated proteins by Western blotting (from C) is shown. Each value was normalized to vinculin except for the proinsulin to insulin ratio. Pdia1 fl/fl (n = 2), Pdia1 fl/+ (n = 2), Pdia1 fl/fl;Cre (n = 2).
Figure 1—figure supplement 1.. The RIP-CreERT allele does not impact the ß cell-specific Pdia1 deletion phenotype.
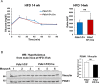
(A) Pdia1 fl/+ mice harboring the RIP-CreERT allele (n = 3) exhibit similar glucose tolerance tests as Pdia1 fl/+ mice without the RIP-CreERT allele (n = 4) at 14 wks after HFD. All mice were injected with Tam after 3 wks of HFD. (B) Δ-AUC of glucose tolerance test in (A). (C) PDIA1 protein levels did not change in hypothalamic brain tissue after 37 wks of HFD. Pdia1 fl/fl (n = 3), Pdia1 fl/fl;RIP-CreERT (n = 3). All data are shown as Mean ± SEM.
Figure 2.. Pdia1 is specifically and persistently deleted in murine ß cells.
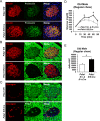
(A–C) Pancreas tissue sections were prepared from female mice at 49 wks after Tam injection and immuno-stained with anti- proinsulin, insulin, PDIA1, and glucagon antibodies. Images were merged with DAPI stain. Scale bar, 20 µm. (D) Old KO mice developed glucose intolerance compared to control genotypes measured by glucose tolerance testing (GTT) at nine wks after Tam injection. Male mice at 9 mon of age were injected with Tam and fed a regular chow. Mice were fasted (4 hr) prior to IP glucose injection (2 g/Kg body weight) and glucose levels were measured by tail bleeding at each time point (0; non-injected, 15, 30, 60, 90 min). control genotypes: Pdia1 fl/+ (n = 2), Pdia1 fl/+;Cre (n = 5), KO; n = 5. (E) Area under the GTT curve (Δ-AUC) of (D) is indicated in graph.
Figure 3.. ß cell-specific Pdia1 deleted male mice are glucose intolerant with defective insulin secretion when fed a 45% High Fat Diet (HFD).
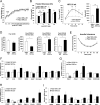
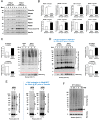
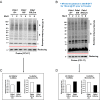
All mice were Tam injected at three wks after HFD was started. (A) No difference was observed in body weight (g) between control genotypes and KO mice. Mean ± SEM, controls; n = 17, KO; n = 12. (B) Fasting (4 hr) blood glucose levels were elevated in KO mice at 11, 16 and 20 wks after HFD. Glucose levels were measured by tail bleeding. Mean ± SEM, control genotypes; n = 17, KO; n = 12. (C) KO mice displayed higher blood glucose levels and area under the GTT curve (Δ-AUC) compared to control genotypes during glucose tolerance testing (GTT) after HFD for 25 wks. GTT were performed at multiple time points after HFD in two independent cohorts and representative results are shown. Mice were fasted (4 hr) prior to IP glucose injection (1 g/Kg body weight) and glucose levels were measured by tail bleeding at each time point (0; non-injected, 15, 30, 60, 90 min). control genotypes; n = 17, KO; n = 6. (D) KO mice exhibited decreased serum insulin levels and an increased serum proinsulin/insulin ratio compared to control genotypes. Insulin and proinsulin ELISAs were performed with the serum obtained from mice after fasting (overnight) and re-feeding (4 r) after HFD for 17 wks. control genotypes; n = 8, KO; n = 12. (E) No difference was observed in insulin tolerance tests performed after HFD for 20 wks. Mice were fasted for 4 hr before IP injection of insulin (1.5units/Kg). Glucose levels were measured by tail bleeding at each time point (0; non-injection, 15, 30, 60, 90, 120 min). control genotypes; n = 17, KO; n = 12. (F–I) Total RNA was extracted from islets isolated from mice after HFD for 30 wks. mRNA expression was measured by qRT-PCR. control genotypes; n = 3, KO; n = 3 mice. (F). ß cell-, α cell- and insulin processing genes. (G) PDI family and SERCA genes. (H) UPR genes. (I) Antioxidant response- and cell death- related genes. All data are shown as Mean ± SEM. p<0.05*, p<0.01**, p<0.001***.
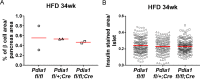
Figure 3—figure supplement 1.. ß cell area relative to pancreas area and β cell number relative to islet area were not changed in ß cell-specific Pdia1 deleted male mice after 34 wks of HFD.
Pancreata were harvested, fixed in 4% PFA, and embedded in paraffin. For each pancreas three sections were prepared at 200 µm intervals and stained with guinea pig α-insulin antibody and DAPI. Images were taken by Aperio Imaging system. Insulin stained ß cell area, islet area and pancreas area were measured by Aperio Imagescope software. A. The percent of ß cell area relative to total pancreas area is shown. B. The number of insulin positive cells per islet is shown. Two mice for each genotype were combined and the mean value is indicated.
Figure 4.. Pdia1 deletion induces morphological abnormalities including decreased insulin granule numbers, ER vesiculation and distention, mitochondrial swelling and nuclear condensation in ß cells.
(A–H). Transmission electron microscopy was performed on pancreata obtained from genetic controls (A, B, E, F) and KO (C, D, G, H) male mice after 40 wks of HFD. Images were obtained at 1900X (A, C, E, G) or 4800X (B, D, F, H) magnification. Scale bar represents 5 µm or 1 µm as indicated. Marked area is two times enlarged on the right side: N, nucleus; M, mitochondria; *, distended endoplasmic reticulum; GA, Golgi apparatus. Yellow arrows, mature granules. Orange filled open arrowheads, immature granules. (I) Pdia1 KO mice had reduced numbers of mature and immature granules. For each genotype 80–110 images were quantified. genetic controls; n = 3, KO; n = 2. Mean value is indicated. (J) Mature granule sizes were larger in Pdia1 KO mice compared to genetic controls without differences in mature granule dense core size. For each genotype, 40 images were quantified. genetic controls; n = 3, KO; n = 2.
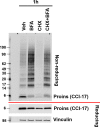
Figure 5.. Pdia1 deletion in HFD fed mice increases islet steady state proinsulin to insulin ratio with accumulation of high molecular weight (HMW) proinsulin complexes.
(A, C) Western blotting was performed for murine islets isolated after HFD for 30 wks. After overnight recovery, islets were lysed and analyzed under reducing (A) or non-reducing (C, D) conditions by SDS-PAGE and Western blotting. Image exposed for a different time for reduced proinsulin (CCI-17) in (A) was used to represent the total proinsulin levels in (D). Six independent mice were analyzed and technical duplicates are indicated as’ on the top of gel. (B). Quantification of indicated proteins was performed under reducing conditions (A). Each value was normalized to vinculin. Proinsulin/insulin ratios were calculated based on the quantification of proinsulin and insulin species under reducing conditions (A). (C) Five samples from A were analyzed under non-reducing conditions on a 4–12% Bis-Tris SDS gel. The proinsulin blot under reducing conditions is under the red line. Left side. Quantification of HMW proinsulin complexes under non-reducing conditions is indicated. (C), left side, upper) The ratio of 49–198 kDa (a) to 14–49 kDa (b) proinsulin complexes is shown. (C), left side, lower) The ratio of 14–49 kDa (b) proinsulin complexes to proinsulin monomer under reducing conditions is shown. Mean value is indicated in graph. (D) The ten samples in A were analyzed under non-reducing conditions after the gel was incubated in 25 mM DTT for 10 min at RT prior to transfer. To control for variable transfer from a gradient gel, we used a fixed percentage gel (12% Bis-Tris SDS). Right side. Quantification is shown for HMW proinsulin complexes under non-reducing conditions. (D), right side, upper) The ratio of 49–198 kDa (a) to 14–49 kDa (b) proinsulin complexes is shown. (D), right side, lower) The ratio of 14–49 kDa (b) proinsulin complexes to proinsulin monomer under reducing conditions is shown. (A–D) genetic controls; n = 3, KO; n = 3 mice. Mean ± SEM, p<0.05*, p<0.01**, p<0.001***. (E) WT murine islets were treated with or without NEM and lysates were analyzed under non-reducing conditions. Equal numbers of islets were divided into two tubes. Left side islets were rinsed with cold-PBS and lysed on ice. Right side islets were rinsed with cold-PBS containing 20 mM NEM and lysed in lysis buffer containing 2 mM NEM. Samples were prepared alongside and lysates were boiled for 5 min. Equal amounts of lysates were loaded and analyzed with or without gel incubation in 25 mM DTT for 10 min at RT prior to transfer. Two different exposure time images (short, long) are shown after DTT incubation. (F) WT murine islets were treated with increasing concentrations of DTT for 20 min in culture at room temperature and then analyzed by non-reducing and reducing SDS-PAGE and Western blotting with proinsulin antibody (CCI-17). The range of oligomeric proinsulin species are identified by an open arrowhead and monomeric proinsulin is indicated by black arrowhead.
Figure 5—figure supplement 1.. Pdia1 deletion increases accumulation of HMW proinsulin complexes under regular diet.
(A) Islets were isolated from female mice at 14 wks after Tam injection, as described in Figure 1C, and analyzed by Western blotting under non-reducing conditions. The longer exposed image of reduced proinsulin (CCI-17) in Figure 1C is shown to represent the total proinsulin levels. (B) The same amounts of lysates from A were analyzed under non-reducing conditions after the gel was incubated in 25 mM DTT for 10 min at RT prior to transfer. To avoid unequal transfer from a gradient gel, we used a fixed percentage gel (12% Bis-Tris SDS). The proinsulin blot under reducing conditions is located under the red line. (D) The quantification of disulfide-linked proinsulin complexes (B) is shown. The left side of (C–D) shows the ratio of 49–198 kDa (a) to 14–49 kDa (b) proinsulin complexes. The right side of (C–D) shows the ratio of 14–49 kDa (b) proinsulin complexes to proinsulin monomer under reducing conditions.
Figure 5—figure supplement 2.. Inhibition of ER to Golgi trafficking increases proinsulin disulfide linked HMW complex formation.
After overnight recovery, WT islets were incubated in media containing brefeldin A (BFA, 5 µg/ml) and/or cycloheximide (CHX, 100 μg/ml) for 1 hr at 37°C and then analyzed by non-reducing and reducing SDS-PAGE and Western blotting with proinsulin antibody (CCI-17).
Figure 5—figure supplement 3.. PDIA1 overexpression reduces proinsulin.
Adenoviruses that express human proinsulin (Ad-hProins) or human PDIA1 (Ad-hPDI) or catalytically inactive PDIA1 mutant (Ad-hPDImut) were infected into WT MEFs. After 48 hr, cells were treated with DTT (2.5, 5 mM) for 20 min or Menadione (50, 100 μM) for 1 hr. Lysates were analyzed by non-reducing and reducing SDS-PAGE and Western blotting with anti-human proinsulin antibody (1B24). Unfortunately, this antibody for human proinsulin does not recognize HMW proinsulin aggregates.
Figure 6.. Pdia1 deletion increases sensitivity to menadione oxidant: Increased ROS, nuclear condensation, and HMW proinsulin complexes were observed in menadione-treated KO islets.
A. Islets isolated from mice after 30 wks HFD were treated with or without Menadione (10 µM, 3 hr) and co-stained with CellROX Deep Red (red) and Hoechst 33342 (blue). Live islet images were obtained by an Opera Phenix high content screening system (63X objective lens) and seven z-stack images (1 µm interval) were combined. Scale bar, 20 μm. genetic controls; n = 3, KO; n = 3. (B) Quantification of ROS mean intensity is shown. CellROX Deep Red mean intensity (divided by area) was measured by image J software. Mean ± SEM, p<0.001***. (C). Quantification of nuclear mean area (µm2) measured in Hoechst 33342 stained images by ImageJ software is shown. Mean ± SEM, p<0.001***. (D). Histogram analysis of nuclear sizes is shown. Percent frequencies are indicated in the graph. (E) Western blot of islets isolated from mice after 37 wks of HFD by SDS-PAGE under reducing and non-reducing conditions is shown. After overnight recovery, islets were treated with menadione (100 µM) for 1 hr. Islet preparations from five independent control and KO mice were performed and representative images are shown. Quantification of the ratio of HMW proinsulin complexes (a) to monomeric proinsulin under reducing conditions is shown in graph (lower). Mean ± SEM, p<0.05*, p<0.01**, p<0.001***. Controls; n = 5, KO; n = 5 mice. (F) WT murine islets were treated with Menadione (100 µM) for 1 hr, treated with or without NEM as in Figure 5E, and lysates were prepared and analyzed under non-reducing conditions.
Figure 6—figure supplement 1.. PDIA1 inhibitor KSC-34 recapitulates effects of Pdia1 deletion.
WT murine islets were treated with menadione in the presence or absence of 30 μM KSC-34 for 3 hr and then treated with menadione (100μM) for 1 hr at 37°C. Five independent experiments were performed and representative results are shown. (A) Islet lysates were prepared and analyzed by non-reducing and reducing SDS-PAGE for Western blotting with CCI-17 antibody. (B) Quantification shows the proportion of HMW complexes (49–198 kDa) relative to the amount of reduced proinsulin monomer. Mean ± SEM, p<0.05*, p<0.01**, p<0.001***. Controls; n = 5, KO; n = 5 mice.
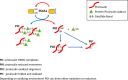
Figure 7.. The role of PDIA1 in proinsulin disulfide bond formation.
Here, we show that PDIA1 is not required but increases the efficiency of proinsulin maturation, possibly by reducing HMW proinsulin complexes. In the absence of PDIA1, disulfide bond formation, reduction and/or isomerization in proinsulin are inadequate during increased demand so HMW proinsulin complexes accumulate in the ER upon metabolic pressure, such as a HFD. The figure depicts the formation of proinsulin HMW complexes with cellular proteins and the role of PDIA1 in interconversions to reduced proinsulin with subsequent oxidation to dimer/oligomeric proinsulin and oxidized folded proinsulin.
Similar articles
- Protein disulfide isomerase A1 regulates breast cancer cell immunorecognition in a manner dependent on redox state.
Alhammad R, Khunchai S, Tongmuang N, Limjindaporn T, Yenchitsomanus PT, Mutti L, Krstic-Demonacos M, Demonacos C. Alhammad R, et al. Oncol Rep. 2020 Dec;44(6):2406-2418. doi: 10.3892/or.2020.7816. Epub 2020 Oct 20. Oncol Rep. 2020. PMID: 33125139 Free PMC article. - OSGEP regulates islet β-cell function by modulating proinsulin translation and maintaining ER stress homeostasis in mice.
Liu Y, Yang X, Zhou J, Yang H, Yang R, Zhu P, Zhou R, Wu T, Gao Y, Ye Z, Li X, Liu R, Zhang W, Zhou H, Li Q. Liu Y, et al. Nat Commun. 2024 Dec 2;15(1):10479. doi: 10.1038/s41467-024-54905-8. Nat Commun. 2024. PMID: 39622811 Free PMC article. - Proinsulin misfolding is an early event in the progression to type 2 diabetes.
Arunagiri A, Haataja L, Pottekat A, Pamenan F, Kim S, Zeltser LM, Paton AW, Paton JC, Tsai B, Itkin-Ansari P, Kaufman RJ, Liu M, Arvan P. Arunagiri A, et al. Elife. 2019 Jun 11;8:e44532. doi: 10.7554/eLife.44532. Elife. 2019. PMID: 31184302 Free PMC article. - Protein disulfide isomerases: Redox connections in and out of the endoplasmic reticulum.
Soares Moretti AI, Martins Laurindo FR. Soares Moretti AI, et al. Arch Biochem Biophys. 2017 Mar 1;617:106-119. doi: 10.1016/j.abb.2016.11.007. Epub 2016 Nov 24. Arch Biochem Biophys. 2017. PMID: 27889386 Review. - Proinsulin misfolding and endoplasmic reticulum stress during the development and progression of diabetes.
Sun J, Cui J, He Q, Chen Z, Arvan P, Liu M. Sun J, et al. Mol Aspects Med. 2015 Apr;42:105-18. doi: 10.1016/j.mam.2015.01.001. Epub 2015 Jan 8. Mol Aspects Med. 2015. PMID: 25579745 Free PMC article. Review.
Cited by
- The Post-Translational Role of UFMylation in Physiology and Disease.
Wang X, Xu X, Wang Z. Wang X, et al. Cells. 2023 Oct 29;12(21):2543. doi: 10.3390/cells12212543. Cells. 2023. PMID: 37947621 Free PMC article. Review. - Lipid Modulation in the Formation of β-Sheet Structures. Implications for De Novo Design of Human Islet Amyloid Polypeptide and the Impact on β-Cell Homeostasis.
Martínez-Navarro I, Díaz-Molina R, Pulido-Capiz A, Mas-Oliva J, Luna-Reyes I, Rodríguez-Velázquez E, Rivero IA, Ramos-Ibarra MA, Alatorre-Meda M, García-González V. Martínez-Navarro I, et al. Biomolecules. 2020 Aug 19;10(9):1201. doi: 10.3390/biom10091201. Biomolecules. 2020. PMID: 32824918 Free PMC article. - Abnormal ECA-Binding Membrane Glycans and Galactosylated CAT and P4HB in Lesion Tissues as Potential Biomarkers for Hepatocellular Carcinoma Diagnosis.
Kong Y, Chen H, Chen M, Li Y, Li J, Liu Q, Xiong H, Guo T, Xie Y, Yuan Y, Zhang XL. Kong Y, et al. Front Oncol. 2022 Mar 22;12:855952. doi: 10.3389/fonc.2022.855952. eCollection 2022. Front Oncol. 2022. PMID: 35392238 Free PMC article. - Investigating the Role of 17-Beta Estradiol in the Regulation of the Unfolded Protein Response (UPR) in Pancreatic Beta Cells.
De Paoli M, Shah D, Zakharia A, Patel Z, Patel Z, Pakhi P, Werstuck GH. De Paoli M, et al. Int J Mol Sci. 2024 Feb 2;25(3):1816. doi: 10.3390/ijms25031816. Int J Mol Sci. 2024. PMID: 38339098 Free PMC article. - Distinct states of proinsulin misfolding in MIDY.
Haataja L, Arunagiri A, Hassan A, Regan K, Tsai B, Dhayalan B, Weiss MA, Liu M, Arvan P. Haataja L, et al. Cell Mol Life Sci. 2021 Aug;78(16):6017-6031. doi: 10.1007/s00018-021-03871-1. Epub 2021 Jul 10. Cell Mol Life Sci. 2021. PMID: 34245311 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- R01DK103185/NH/NIH HHS/United States
- P30 CA030199/NH/NIH HHS/United States
- R24 DK110973/DK/NIDDK NIH HHS/United States
- U24 DK098085/DK/NIDDK NIH HHS/United States
- R01 DK048280/DK/NIDDK NIH HHS/United States
- P30 DK063491/DK/NIDDK NIH HHS/United States
- R01 DK113171/DK/NIDDK NIH HHS/United States
- P30 DK020572/DK/NIDDK NIH HHS/United States
- R01DK113171/NH/NIH HHS/United States
- P30 CA030199/CA/NCI NIH HHS/United States
- P30 DK063491/NH/NIH HHS/United States
- R01 DK103185/DK/NIDDK NIH HHS/United States
- R24DK110973/NH/NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous