Omadacycline: a novel aminomethylcycline - PubMed (original) (raw)
Review
. 2019 Jul 2:12:1895-1915.
doi: 10.2147/IDR.S171352. eCollection 2019.
Affiliations
- PMID: 31308710
- PMCID: PMC6613460
- DOI: 10.2147/IDR.S171352
Review
Omadacycline: a novel aminomethylcycline
Rodrigo M Burgos et al. Infect Drug Resist. 2019.
Abstract
Tetracyclines have come a long way since they became available almost seven decades ago, with numerous enhancements allowing new agents to overcome bacterial mechanisms of resistance. However, these enhancements come with toxicities and pharmacokinetic disadvantages such as the gastrointestinal side-effects and poor oral bioavailability seen with the glycylcylcines. Omadacycline, a new and improved tetracycline, has demonstrated a broad spectrum of in vitro activity, has oral and intravenous formulations, improved safety compared to glycylcyclines, as well as clinical efficacy and safety for two types of infections: acute bacterial skin and skin structure infections and community-acquired bacterial pneumonia. This review will summarize salient points about its pharmacologic properties, available clinical efficacy, and safety data and omadacycline's place in therapy.
Keywords: community-acquired pneumonia; skin infections; tetracyclines.
Conflict of interest statement
Dr Rodvold is a consultant for Paratek Pharmaceuticals, Inc. and reports personal fees from Paratek Pharmaceuticals, outside the submitted work. The authors report no other conflicts of interest in this work.
Figures
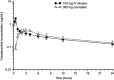
Figure 1
Mean±SD plasma concentration-time curve of omadacycline after administration of 300 mg oral dose (open triangles) and 100 mg intravenous dose (closed circles) in healthy subjects.Note: Data from Sun et al.
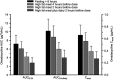
Figure 2
Mean±SD area under the plasma concentration-time curve (AUC) and maximum plasma concentration (Cmax) of omadacycline following oral administration of a 300 mg oral dose under fasting condition (solid black bar), when a standard high-fat non-dairy meal was ingested 4 hours pre-dose (solid gray bar), when a standard high-fat non-dairy meal was ingested 2 hours pre-dose (crossed line bar), and when a standard high-fat meal including dairy was ingested 2 hours pre-dose (confetti bar).Note: Data from Tzanis et al.
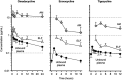
Figure 3
Mean±SD unbound plasma, epithelial lining fluid (ELF), and alveolar macrophages (AM) concentration-time curve of omadacycline, eravacycline, and tigecycline after multiple intravenous doses of 100 mg, 1 mg/kg, and 50 mg, respectively. Note that the duration of time represents the dosing interval of 24 hours for omadacycline and 12 hours for eravacycline and tigecycline.Note: Data from these studies,
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases