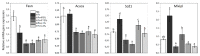High-Fructose Diet-Induced Metabolic Disorders Were Counteracted by the Intake of Fruit and Leaves of Sweet Cherry in Wistar Rats - PubMed (original) (raw)
High-Fructose Diet-Induced Metabolic Disorders Were Counteracted by the Intake of Fruit and Leaves of Sweet Cherry in Wistar Rats
Kinga Dziadek et al. Nutrients. 2019.
Abstract
Numerous studies have indicated that the use of plants rich in bioactive compounds may reduce the risk of non-communicable diseases. The aim of this study was to investigate how the addition of fruit and leaves to high-fructose diet affects lipid metabolism, including the expression of genes involved in fatty acid synthesis and oxidation in the liver and adipose tissue, as well as oxidative stress and inflammation in Wistar rats. The animals were fed with AIN-93G diet, high fructose (HFr) diet, HFr diet with addition of 5% or 10% freeze-dried fruits, and HFr diet with addition of 1% or 3% freeze-dried leaves. The experiment lasted 12 weeks. The results showed that the intake of fruit and leaves of sweet cherry caused the improvement of the liver function, as well as beneficially affected lipid metabolism, among others, by regulating the expression of genes associated with fatty acid synthesis and β-oxidation. Additionally, they exhibited antioxidant and anti-inflammatory properties. In conclusion, the addition of fruit and leaves reduced the adverse changes arising from the consumption of high fructose diet. Therefore, not only commonly consumed fruits, but also leaves can be potentially used as functional foods. These findings may be helpful in prevention and treatment of the obesity-related metabolic diseases, especially cardiovascular diseases.
Keywords: Wistar rats; antioxidant enzymes; high fructose diet; inflammation; lipid metabolism genes; sweet cherry leaves.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures
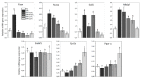
Figure 1
The expression of fatty acid synthesis genes (Fasn, Acaca, Scd1, Mlxipl and Srebf1) and fatty acid oxidation genes (Cpt1a and Ppar-α) in liver of rats. NC—negative control; HFr—high fructose; HFr + F5%—HFr diet with addition of 5% of fruits; HFr + F10%—HFr diet with addition of 10% of fruits; HFr + L1%—HFr diet with addition of 1% of leaves; HFr + L3%—HFr diet with addition of 3% of leaves. Data are expressed as mean ± SD. Values with different letters (a–f) are statistically different (p < 0.05).
Figure 2
The expression of fatty acid synthesis genes (Fasn, Acaca, Scd1, Mlxipl) in adipose tissue of rats. NC—negative control; HFr—high fructose; HFr + F5%—HFr diet with addition of 5% of fruits; HFr + F10%—HFr diet with addition of 10% of fruits; HFr + L1%—HFr diet with addition of 1% of leaves; HFr + L3%—HFr diet with addition of 3% of leaves. Data are expressed as mean ± SD. Values with different letters (a–d) are statistically different (p < 0.05).
Similar articles
- Dioscoreophyllum cumminsii (Stapf) Diels leaves halt high-fructose induced metabolic syndrome: Hyperglycemia, insulin resistance, inflammation and oxidative stress.
Ajiboye TO, Aliyu H, Tanimu MA, Muhammad RM, Ibitoye OB. Ajiboye TO, et al. J Ethnopharmacol. 2016 Nov 4;192:471-479. doi: 10.1016/j.jep.2016.08.024. Epub 2016 Aug 26. J Ethnopharmacol. 2016. PMID: 27568876 - Beneficial effects of cherry consumption as a dietary intervention for metabolic, hepatic and vascular complications in type 2 diabetic rats.
Van der Werf R, Walter C, Bietiger W, Seyfritz E, Mura C, Peronet C, Legrandois J, Werner D, Ennahar S, Digel F, Maillard-Pedracini E, Pinget M, Jeandidier N, Marchioni E, Sigrist S, Dal S. Van der Werf R, et al. Cardiovasc Diabetol. 2018 Jul 20;17(1):104. doi: 10.1186/s12933-018-0744-6. Cardiovasc Diabetol. 2018. PMID: 30029691 Free PMC article. - Fruit Development in Sweet Cherry.
Vignati E, Lipska M, Dunwell JM, Caccamo M, Simkin AJ. Vignati E, et al. Plants (Basel). 2022 Jun 7;11(12):1531. doi: 10.3390/plants11121531. Plants (Basel). 2022. PMID: 35736682 Free PMC article. Review. - Cherry intake as a dietary strategy in sport and diseases: a review of clinical applicability and mechanisms of action.
Santos HO, Genario R, Gomes GK, Schoenfeld BJ. Santos HO, et al. Crit Rev Food Sci Nutr. 2021;61(3):417-430. doi: 10.1080/10408398.2020.1734912. Epub 2020 Mar 4. Crit Rev Food Sci Nutr. 2021. PMID: 32126807 Review.
Cited by
- Valorization of Peels of Eight Peach Varieties: GC-MS Profile, Free and Bound Phenolics and Corresponding Biological Activities.
Mihaylova D, Popova A, Desseva I, Dincheva I, Tumbarski Y. Mihaylova D, et al. Antioxidants (Basel). 2023 Jan 16;12(1):205. doi: 10.3390/antiox12010205. Antioxidants (Basel). 2023. PMID: 36671066 Free PMC article. - Phenolic Compounds from Cherries and Berries for Chronic Disease Management and Cardiovascular Risk Reduction.
Carvalho F, Lahlou RA, Silva LR. Carvalho F, et al. Nutrients. 2024 May 23;16(11):1597. doi: 10.3390/nu16111597. Nutrients. 2024. PMID: 38892529 Free PMC article. Review. - Sweet Cherries as Anti-Cancer Agents: From Bioactive Compounds to Function.
Fonseca LRS, Silva GR, Luís Â, Cardoso HJ, Correia S, Vaz CV, Duarte AP, Socorro S. Fonseca LRS, et al. Molecules. 2021 May 15;26(10):2941. doi: 10.3390/molecules26102941. Molecules. 2021. PMID: 34063349 Free PMC article. Review. - Konjac Glucomannan: An Emerging Specialty Medical Food to Aid in the Treatment of Type 2 Diabetes Mellitus.
Fang Y, Ma J, Lei P, Wang L, Qu J, Zhao J, Liu F, Yan X, Wu W, Jin L, Ji H, Sun D. Fang Y, et al. Foods. 2023 Jan 12;12(2):363. doi: 10.3390/foods12020363. Foods. 2023. PMID: 36673456 Free PMC article. Review. - Coconut oil and medium-chain fatty acids attenuate high-fat diet-induced obesity in mice through increased thermogenesis by activating brown adipose tissue.
Gao Y, Liu Y, Han X, Zhou F, Guo J, Huang W, Zhan J, You Y. Gao Y, et al. Front Nutr. 2022 Oct 28;9:896021. doi: 10.3389/fnut.2022.896021. eCollection 2022. Front Nutr. 2022. PMID: 36386906 Free PMC article.
References
- WHO Obesity and Overweight. [(accessed on 20 August 2019)]; Available online: https://www.who.int/en/news-room/fact-sheets/detail/obesity-and-overweight.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical