Cancer Risks Associated With Germline PALB2 Pathogenic Variants: An International Study of 524 Families - PubMed (original) (raw)
. 2020 Mar 1;38(7):674-685.
doi: 10.1200/JCO.19.01907. Epub 2019 Dec 16.
Goska Leslie 1, Alicja Doroszuk 2, Sandra Schneider 2, Jamie Allen 1, Brennan Decker 1 3 4, Alison M Dunning 5, James Redman 2, James Scarth 2, Inga Plaskocinska 2, Craig Luccarini 5, Mitul Shah 5, Karen Pooley 1, Leila Dorling 1, Andrew Lee 1, Muriel A Adank 6, Julian Adlard 7, Kristiina Aittomäki 8, Irene L Andrulis 9, Peter Ang 10, Julian Barwell 11, Jonine L Bernstein 12, Kristie Bobolis 13, Åke Borg 14, Carl Blomqvist 15, Kathleen B M Claes 16, Patrick Concannon 17, Adeline Cuggia 18 19, Julie O Culver 20, Francesca Damiola 21, Antoine de Pauw 22, Orland Diez 23, Jill S Dolinsky 24, Susan M Domchek 25 26, Christoph Engel 27, D Gareth Evans 28, Florentia Fostira [ 29](#full-view-affiliation-29 "Molecular Diagnostics Laboratory, INRASTES, National Centre for Scientific Research "Demokritos," Athens, Greece."), Judy Garber 26 30, Lisa Golmard 22, Ellen L Goode 31, Stephen B Gruber 32, Eric Hahnen 33 34, Christopher Hake 13, Tuomas Heikkinen 35, Judith E Hurley 36, Ramunas Janavicius 37 38, Zdenek Kleibl 39, Petra Kleiblova 39 40, Irene Konstantopoulou [ 29](#full-view-affiliation-29 "Molecular Diagnostics Laboratory, INRASTES, National Centre for Scientific Research "Demokritos," Athens, Greece."), Anders Kvist 14, Holly Laduca 24, Ann S G Lee 11 41 42, Fabienne Lesueur 43, Eamonn R Maher 2, Arto Mannermaa 44, Siranoush Manoukian 45, Rachel McFarland 24 46, Wendy McKinnon 47, Alfons Meindl 48, Kelly Metcalfe 49, Nur Aishah Mohd Taib 50, Jukka Moilanen 51, Katherine L Nathanson 25, Susan Neuhausen 52, Pei Sze Ng 50 53, Tu Nguyen-Dumont 54 55, Sarah M Nielsen 56, Florian Obermair 57, Kenneth Offit 26 58, Olufunmilayo I Olopade 56, Laura Ottini 59, Judith Penkert 60, Katri Pylkäs 61, Paolo Radice 62, Susan J Ramus 63 64, Vilius Rudaitis 37, Lucy Side 65, Rachel Silva-Smith 66, Valentina Silvestri 59, Anne-Bine Skytte 67, Thomas Slavin 13 68, Jana Soukupova 39, Carlo Tondini 69, Alison H Trainer 70 71, Gary Unzeitig 13, Lydia Usha 13, Thomas van Overeem Hansen 72 73, James Whitworth 2, Marie Wood 47, Cheng Har Yip 53, Sook-Yee Yoon 53, Amal Yussuf 24, George Zogopoulos 18 19, David Goldgar 74, John L Hopper 75, Georgia Chenevix-Trench 76, Paul Pharoah 1, Sophia H L George 77, Judith Balmaña 23 26, Claude Houdayer 22 78, Paul James 70 71, Zaki El-Haffaf 79, Hans Ehrencrona 80 81, Marketa Janatova 39, Paolo Peterlongo 82, Heli Nevanlinna 35, Rita Schmutzler 33 34, Soo-Hwang Teo 50 53, Mark Robson 26 83, Tuya Pal 84, Fergus Couch 26 85, Jeffrey N Weitzel 13 68, Aaron Elliott 24, Melissa Southey 54 55, Robert Winqvist 61, Douglas F Easton 1, William D Foulkes 18 86, Antonis C Antoniou 1, Marc Tischkowitz 2
Affiliations
- PMID: 31841383
- PMCID: PMC7049229
- DOI: 10.1200/JCO.19.01907
Cancer Risks Associated With Germline PALB2 Pathogenic Variants: An International Study of 524 Families
Xin Yang et al. J Clin Oncol. 2020.
Abstract
Purpose: To estimate age-specific relative and absolute cancer risks of breast cancer and to estimate risks of ovarian, pancreatic, male breast, prostate, and colorectal cancers associated with germline PALB2 pathogenic variants (PVs) because these risks have not been extensively characterized.
Methods: We analyzed data from 524 families with PALB2 PVs from 21 countries. Complex segregation analysis was used to estimate relative risks (RRs; relative to country-specific population incidences) and absolute risks of cancers. The models allowed for residual familial aggregation of breast and ovarian cancer and were adjusted for the family-specific ascertainment schemes.
Results: We found associations between PALB2 PVs and risk of female breast cancer (RR, 7.18; 95% CI, 5.82 to 8.85; P = 6.5 × 10-76), ovarian cancer (RR, 2.91; 95% CI, 1.40 to 6.04; P = 4.1 × 10-3), pancreatic cancer (RR, 2.37; 95% CI, 1.24 to 4.50; P = 8.7 × 10-3), and male breast cancer (RR, 7.34; 95% CI, 1.28 to 42.18; P = 2.6 × 10-2). There was no evidence for increased risks of prostate or colorectal cancer. The breast cancer RRs declined with age (P for trend = 2.0 × 10-3). After adjusting for family ascertainment, breast cancer risk estimates on the basis of multiple case families were similar to the estimates from families ascertained through population-based studies (P for difference = .41). On the basis of the combined data, the estimated risks to age 80 years were 53% (95% CI, 44% to 63%) for female breast cancer, 5% (95% CI, 2% to 10%) for ovarian cancer, 2%-3% (95% CI females, 1% to 4%; 95% CI males, 2% to 5%) for pancreatic cancer, and 1% (95% CI, 0.2% to 5%) for male breast cancer.
Conclusion: These results confirm PALB2 as a major breast cancer susceptibility gene and establish substantial associations between germline PALB2 PVs and ovarian, pancreatic, and male breast cancers. These findings will facilitate incorporation of PALB2 into risk prediction models and optimize the clinical cancer risk management of PALB2 PV carriers.
Figures
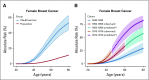
FIG 1.
Estimated absolute risk of developing breast cancer for women with germline PALB2 pathogenic variants (PVs) by age under (A) a model that assumes no cohort effect (blue, the risk for women with PALB2 PVs; red, the risk in the United Kingdom general population, assuming that population incidences are applicable to individuals born between 1950 and 1959) and (B) a model that allows for cohort-specific relative risk parameters. The dotted curves and shaded area show the 95% CI. (*) Assuming that the relative risk estimates apply to the unobserved age ranges for women born in these cohorts.
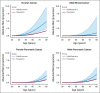
FIG 2.
Estimated absolute risk of developing ovarian, pancreatic, and male breast cancer for individuals with PALB2 pathogenic variants PVs and in the general population by age (assuming that population incidences are applicable to individuals born between 1950 and 1959). The dotted curves and shaded area show the 95% CI.
References
- Xia B, Sheng Q, Nakanishi K, et al. Control of BRCA2 cellular and clinical functions by a nuclear partner, PALB2. Mol Cell. 2006;22:719–729. -PubMed
- Erkko H, Xia B, Nikkilä J, et al. A recurrent mutation in PALB2 in Finnish cancer families. Nature. 2007;446:316–319. -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 203477/Z/16/Z/WT_/Wellcome Trust/United Kingdom
- UM1 CA164920/CA/NCI NIH HHS/United States
- R01 CA097397/CA/NCI NIH HHS/United States
- 20861/CRUK_/Cancer Research UK/United Kingdom
- U01 CA116167/CA/NCI NIH HHS/United States
- R01 CA116167/CA/NCI NIH HHS/United States
- P50 CA116201/CA/NCI NIH HHS/United States
- U01 CA083178/CA/NCI NIH HHS/United States
- R01 CA129639/CA/NCI NIH HHS/United States
- R01 CA114236/CA/NCI NIH HHS/United States
- L60 MD014321/MD/NIMHD NIH HHS/United States
- P30 CA008748/CA/NCI NIH HHS/United States
- R01 CA128978/CA/NCI NIH HHS/United States
- R01 ES027121/ES/NIEHS NIH HHS/United States
- 16563/CRUK_/Cancer Research UK/United Kingdom
- K08 CA234394/CA/NCI NIH HHS/United States
- 10119/CRUK_/Cancer Research UK/United Kingdom
- 16561/CRUK_/Cancer Research UK/United Kingdom
- 10124/CRUK_/Cancer Research UK/United Kingdom
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous