Treatment interventions to maintain abstinence from alcohol in primary care: systematic review and network meta-analysis - PubMed (original) (raw)
Meta-Analysis
doi: 10.1136/bmj.m3934.
Luke A McGuinness 1, Roy G Elbers 1, Georgina J MacArthur 1, Abigail Taylor 1, Alexandra McAleenan 1, Sarah Dawson 1, José A López-López 1 2, Julian P T Higgins 1 3 4, Sean Cowlishaw 1 5, Anne Lingford-Hughes 6, Matthew Hickman 1 3 4, David Kessler 7 4 8
Affiliations
- PMID: 33239318
- PMCID: PMC7687021
- DOI: 10.1136/bmj.m3934
Meta-Analysis
Treatment interventions to maintain abstinence from alcohol in primary care: systematic review and network meta-analysis
Hung-Yuan Cheng et al. BMJ. 2020.
Abstract
Objective: To determine the most effective interventions in recently detoxified, alcohol dependent patients for implementation in primary care.
Design: Systematic review and network meta-analysis.
Data sources: Medline, Embase, PsycINFO, Cochrane CENTRAL, ClinicalTrials.gov, and the World Health Organization's International Clinical Trials Registry Platform.
Study selection: Randomised controlled trials comparing two or more interventions that could be used in primary care. The population was patients with alcohol dependency diagnosed by standardised clinical tools and who became detoxified within four weeks.
Data extraction: Outcomes of interest were continuous abstinence from alcohol (effectiveness) and all cause dropouts (as a proxy for acceptability) at least 12 weeks after start of intervention.
Results: 64 trials (43 interventions) were included. The median probability of abstinence across placebo arms was 25%. Compared with placebo, the only intervention associated with increased probability of abstinence and moderate certainty evidence was acamprosate (odds ratio 1.86, 95% confidence interval 1.49 to 2.33, corresponding to an absolute probability of 38%). Of the 62 included trials that reported all cause dropouts, interventions associated with a reduced number of dropouts compared with placebo (probability 50%) and moderate certainty of evidence were acamprosate (0.73, 0.62 to 0.86; 42%), naltrexone (0.70, 0.50 to 0.98; 41%), and acamprosate-naltrexone (0.30, 0.13 to 0.67; 17%). Acamprosate was the only intervention associated with moderate confidence in the evidence of effectiveness and acceptability up to 12 months. It is uncertain whether other interventions can help maintain abstinence and reduce dropouts because of low confidence in the evidence.
Conclusions: Evidence is lacking for benefit from interventions that could be implemented in primary care settings for alcohol abstinence, other than for acamprosate. More evidence from high quality randomised controlled trials is needed, as are strategies using combined interventions (combinations of drug interventions or drug and psychosocial interventions) to improve treatment of alcohol dependency in primary care.
Systematic review registration: PROSPERO CRD42016049779.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure from at www.icmje.org/coi\_disclosure.pdf and declare: support from the School for Primary Care Research, National Institute for Health Research for the submitted work; HC, AM, JPTH, and LAM report grants from the National Institute for Health Research (NIHR) School for Primary Care Research during the conduct of the study, LAM reports grants from NIHR doctoral research fellowship during the conduct of the study, ALH reports grants from GSK and Lundbeck and personal fees from Silence Therapeutics during the conduct of the study, JLL reports grants from Economic and Social Research Council outside the submitted work, MH reports personal fess and others from MSD, Gilead, and Abbvie outside the submitted work, ALH reports personal fees from Janssen-Cilag, Pfizer, Sanofi-Aventis, Lundbeck, and others from Opiant and Lightlak (unpaid consultancy) outside the submitted work; ALH led the British Association for Psychopharmacology’s guidelines: Evidence based guidelines for the pharmacological management of substance misuse, addiction, and co-morbidity: recommendations from the British Association for Psychopharmacology. J Psychopharmacol 18(3):293-335; and updated in 2012: Evidence-based guidelines for the pharmacological management of substance misuse, addiction and comorbidity: recommendations from BAP. J Psychopharmacol 26(7):899-952. The original guidelines received support from Archimedes Pharma, Lundbeck, Pfizer, Schering. ALH was a member of the National Institute for Health and Care Excellence clinical guideline group: Alcohol-use disorders: diagnosis, assessment and management of harmful drinking and alcohol dependence. A NICE Clinical guideline (CG115) 2011.
Figures
Fig 1
Study selection flowchart
Fig 2
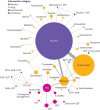
Network plots for alcohol abstinence in relation to treatment for alcohol dependency. Size of circles is proportional to number of randomised patients and width of lines is proportional to number of studies in each direct comparison. A-CHESS=Addiction-Comprehensive Health Enhancement Support System; ACP=acamprosate; CBT=cognitive behavioural therapy; CIT=citalopram; CST=coping skill training; GHB=sodium salt of gamma hydroxybutyric acid (sodium oxybate); MET=motivational enhancement therapy; NTX=naltrexone; TAU=treatment as usual
Fig 3
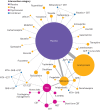
Network plots for all cause dropouts in relation to treatment for alcohol dependency. Size of circles is proportional to number of randomised patients and width of lines is proportional to number of studies in each direct comparison. A-CHESS=Addiction-Comprehensive Health Enhancement Support System; ACP=acamprosate; CBT=cognitive behavioural therapy; CIT=citalopram; CST=coping skill training; GHB=sodium salt of gamma hydroxybutyric acid (sodium oxybate); MET=motivational enhancement therapy; NTX=naltrexone; TAU=treatment as usual
Fig 4
Clustered ranking plot by mean rank values from results of network meta-analyses of abstinence and all cause dropouts. Interventions are coloured according to the confidence of evidence by outline (abstinence) and fill (all cause dropout). The interventions in the white zone were ranked better than placebo based on both outcomes. A-CHESS=Addiction-Comprehensive Health Enhancement Support System; ACP=acamprosate; CBT=cognitive behavioural therapy; CIT=citalopram; EST=escitalopram; CST=coping skill training; GHB=sodium salt of gamma hydroxybutyric acid (sodium oxybate); MET=motivational enhancement therapy; NTX=naltrexone; TAU=treatment as usual
Comment in
- Engagement with Alcoholics Anonymous helps abstinence.
McCartney DJ. McCartney DJ. BMJ. 2021 Jan 5;372:n5. doi: 10.1136/bmj.n5. BMJ. 2021. PMID: 33402357 No abstract available. - Managing alcohol use in primary care.
McCambridge J, Stewart D. McCambridge J, et al. BMJ. 2020 Nov 25;371:m4129. doi: 10.1136/bmj.m4129. BMJ. 2020. PMID: 33481761 No abstract available. - In recently detoxified alcohol-dependent adults, acamprosate increases abstinence maintenance and reduces dropout.
Pal H. Pal H. Ann Intern Med. 2021 May;174(5):JC51. doi: 10.7326/ACPJ202105180-051. Epub 2021 May 4. Ann Intern Med. 2021. PMID: 33939485
Similar articles
- Baclofen for alcohol use disorder.
Agabio R, Saulle R, Rösner S, Minozzi S. Agabio R, et al. Cochrane Database Syst Rev. 2023 Jan 13;1(1):CD012557. doi: 10.1002/14651858.CD012557.pub3. Cochrane Database Syst Rev. 2023. PMID: 36637087 Free PMC article. Review. - Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.
Crider K, Williams J, Qi YP, Gutman J, Yeung L, Mai C, Finkelstain J, Mehta S, Pons-Duran C, Menéndez C, Moraleda C, Rogers L, Daniels K, Green P. Crider K, et al. Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article. - Alcoholics Anonymous and other 12-step programs for alcohol use disorder.
Kelly JF, Humphreys K, Ferri M. Kelly JF, et al. Cochrane Database Syst Rev. 2020 Mar 11;3(3):CD012880. doi: 10.1002/14651858.CD012880.pub2. Cochrane Database Syst Rev. 2020. PMID: 32159228 Free PMC article. - Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.
Sbidian E, Chaimani A, Garcia-Doval I, Do G, Hua C, Mazaud C, Droitcourt C, Hughes C, Ingram JR, Naldi L, Chosidow O, Le Cleach L. Sbidian E, et al. Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. PMID: 29271481 Free PMC article. Updated. Review. - Therapeutic interventions for alcohol dependence in non-inpatient settings: a systematic review and network meta-analysis (protocol).
Cheng HY, Elbers RG, Higgins JP, Taylor A, MacArthur GJ, McGuinness L, Dawson S, López-López JA, Cowlishaw S, Hickman M, Kessler D. Cheng HY, et al. Syst Rev. 2017 Apr 11;6(1):77. doi: 10.1186/s13643-017-0462-2. Syst Rev. 2017. PMID: 28399899 Free PMC article.
Cited by
- IL17RB genetic variants are associated with acamprosate treatment response in patients with alcohol use disorder: A proteomics-informed genomics study.
Ho MF, Zhang C, Cohan JS, Tuncturk M, Heider RM, Coombes BJ, Biernacka J, Moon I, Skime M, Ho AM, Ngo Q, Skillon C, Croarkin PE, Oesterle TS, Karpyak VM, Li H, Weinshilboum RM. Ho MF, et al. Brain Behav Immun. 2024 Aug;120:304-314. doi: 10.1016/j.bbi.2024.06.007. Epub 2024 Jun 8. Brain Behav Immun. 2024. PMID: 38852760 Free PMC article. - Can telehealth expansion boost health care utilization specifically for patients with substance use disorders relative to patients with other types of chronic disease?
Tilhou AS, Dague L, Chachlani P, Burns M. Tilhou AS, et al. PLoS One. 2024 Apr 1;19(4):e0299397. doi: 10.1371/journal.pone.0299397. eCollection 2024. PLoS One. 2024. PMID: 38557607 Free PMC article. - Ligne directrice canadienne pour la prise en charge clinique de la consommation d’alcool à risque élevé et du trouble d’utilisation de l’alcool.
Wood E, Bright J, Hsu K, Goel N, Ross JWG, Hanson A, Teed R, Poulin G, Denning B, Corace K, Chase C, Halpape K, Lim R, Kealey T, Rehm J; pour le comité de rédaction de la Ligne directrice sur le trouble d’utilisation de l’alcool. Wood E, et al. CMAJ. 2024 Mar 10;196(9):E303-E321. doi: 10.1503/cmaj.230715-f. CMAJ. 2024. PMID: 38467412 Free PMC article. French. - Evidence of subgroup differences in meta-analyses evaluating medications for alcohol use disorder: An umbrella review.
Wallach JD, Glick L, Gueorguieva R, O'Malley SS. Wallach JD, et al. Alcohol Clin Exp Res (Hoboken). 2024 Jan;48(1):5-15. doi: 10.1111/acer.15229. Epub 2023 Dec 15. Alcohol Clin Exp Res (Hoboken). 2024. PMID: 38102794 Review. - Automated, tailored adaptive mobile messaging to reduce alcohol consumption in help-seeking adults: A randomized controlled trial.
Muench F, Madden SP, Oommen S, Forthal S, Srinagesh A, Stadler G, Kuerbis A, Leeman RF, Suffoletto B, Baumel A, Haslip C, Vadhan NP, Morgenstern J. Muench F, et al. Addiction. 2024 Mar;119(3):530-543. doi: 10.1111/add.16391. Epub 2023 Nov 27. Addiction. 2024. PMID: 38009576 Clinical Trial.
References
- NHS Digital. Statistics on Alcohol: England, 2018. 2018. digital.nhs.uk/data-and-information/publications/statistical/statistics-.... (Accessed 22 March 2019).
- Williams R, Aspinall R, Bellis M, et al. Addressing liver disease in the UK: a blueprint for attaining excellence in health care and reducing premature mortality from lifestyle issues of excess consumption of alcohol, obesity, and viral hepatitis. Lancet 2014;384:1953-97. 10.1016/S0140-6736(14)61838-9. - DOI - PubMed
- Health Social Care Information Centre Statistics on Alcohol. 2016.
- Public Health England Adult substance misuse statistics from the National Drug Treatment Monitoring System (NDTMS). Public Health England, 2017.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical