Safety and efficacy of avapritinib in advanced systemic mastocytosis: the phase 1 EXPLORER trial - PubMed (original) (raw)
Clinical Trial
. 2021 Dec;27(12):2183-2191.
doi: 10.1038/s41591-021-01538-9. Epub 2021 Dec 6.
Deepti H Radia 2, Tracy I George 3, William A Robinson 4, Albert T Quiery 5, Mark W Drummond 6, Prithviraj Bose 7, Elizabeth O Hexner 8, Elliott F Winton 9, Hans-Peter Horny 10, Meera Tugnait 11, Oleg Schmidt-Kittler 11, Erica K Evans 11, Hui-Min Lin 11, Brenton G Mar 11, Srdan Verstovsek 7, Michael W Deininger # 12, Jason Gotlib # 13
Affiliations
- PMID: 34873347
- PMCID: PMC8674134
- DOI: 10.1038/s41591-021-01538-9
Clinical Trial
Safety and efficacy of avapritinib in advanced systemic mastocytosis: the phase 1 EXPLORER trial
Daniel J DeAngelo et al. Nat Med. 2021 Dec.
Abstract
Advanced systemic mastocytosis (AdvSM) is a rare hematologic neoplasm driven by the KIT D816V mutation and associated with poor survival. This phase 1 study ( NCT02561988 ) evaluated avapritinib (BLU-285), a selective KIT D816V inhibitor, in patients with AdvSM. The primary endpoints were the maximum tolerated dose, recommended phase 2 dose and safety of avapritinib. Secondary endpoints included overall response rate and changes in measures of mast cell burden. Avapritinib was evaluated at doses of 30-400 mg once daily in 86 patients, 69 with centrally confirmed AdvSM. Maximum tolerated dose was not reached, and 200 mg and 300 mg daily were studied in dose-expansion cohorts. The most frequent adverse events observed were periorbital edema (69%), anemia (55%), diarrhea (45%), thrombocytopenia (44%) and nausea (44%). Intracranial bleeding occurred in 13% overall, but in only 1% of patients without severe thrombocytopenia (platelets <50 × 109/l). In 53 response-evaluable patients, the overall response rate was 75%. The complete remission rate was 36%. Avapritinib elicited ≥50% reductions in marrow mast cells and serum tryptase in 92% and 99% of patients, respectively. Avapritinib induced deep and durable responses, including molecular remission of KIT D816V in patients with AdvSM, and was well tolerated at the recommended phase 2 dose of 200 mg daily.
© 2021. The Author(s).
Conflict of interest statement
The authors declare the following competing interests: D.J.D. has served as a consultant for Amgen, Agios, Autolus, Blueprint Medicines Corporation, Forty-Seven, Incyte Corporation, Jazz, Novartis, Pfizer, Shire and Takeda, and received research funding from AbbVie, GlycoMimetics and Novartis. D.H.R. is a paid consultant for and has received research support from Blueprint Medicines Corporation, is a member of the RACs for the EXPLORER and PATHFINDER studies and has received honoraria from Novartis for educational events and consultancy. T.I.G. is a paid consultant and has served as a study steering group member for Blueprint Medicines Corporation, and is employed by ARUP Laboratories/University of Utah, which received funding from Blueprint Medicines Corporation. W.A.R. has received research funding from Blueprint Medicines Corporation. A.T.Q. has no competing interests to declare. M.W.D. received research support from Blueprint Medicines Corporation and Novartis. P.B. has received research honoraria/consulting fees from Blueprint Medicines Corporation, Incyte Corporation, Celgene Corporation (now BMS), CTI BioPharma, Kartos Therapeutics and Sierra Oncology, and research support from Blueprint Medicines Corporation, Celgene Corporation (now BMS), Kartos Therapeutics, Incyte Corporation, Constellation Pharmaceuticals, CTI BioPharma, Astellas Pharmaceuticals, Pfizer, Inc., NS Pharma and Promedior. E.O.H. has received research support and has served on a Data and Safety Monitoring Board for Blueprint Medicines Corporation, has received research support from Novartis Pharmaceuticals and Tmunity Therapeutics, and serves on the hematology exam committee for the American Board of Internal Medicine. E.F.W. has received research support for conduct of clinical studies from Blueprint Medicines Corporation, Samus Therapeutics and Incyte Corporation. H.-P.H. has served as a consultant for Novartis and Blueprint Medicines Corporation. M.T., O.S.-K. and E.K.E. are shareholders and former employees of Blueprint Medicines Corporation. H-M.L. and B.G.M. are both current employees and shareholders of Blueprint Medicines Corporation. S.V. declares research support for the conduct of clinical studies from Incyte, Roche, NS Pharma, Celgene, Gilead, Promedior, CTI BioPharma, AbbVie, Blueprint Medicines Corporation, Novartis, Sierra Oncology, PharmaEssentia, Constellation Pharmaceuticals, Ital Pharma, Protagonist and Kartos Therapeutics. M.W.D. is a paid consultant for Fusion Pharma, Takeda, Medscape, Novartis, Incyte, Sangamo, SPARC, Pfizer and DisperSol, has received research support from Blueprint Medicines Corporation and SPARC, and has served on study management committees for clinical studies sponsored by Pfizer, Takeda and Blueprint Medicines Corporation. J.G. is a paid consultant for, and has received research funding from, Blueprint Medicines Corporation, Deciphera, Incyte and Kartos Therapeutics, and has served as chair of the RAC for Blueprint Medicines Corporation’ phase 1 EXPLORER study, and for the phase 2 PATHFINDER study, and as co-chair for the Deciphera Study Steering Committee for ripretinib in AdvSM and chair of the Central Response Review Committee for the phase 2 study of bezuclastinib in AdvSM.
Figures
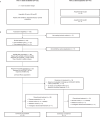
Fig. 1. Study design and patient disposition.
a, Study design showing patients with confirmed AdvSM. b, Patient disposition. AdvSM, advanced systemic mastocytosis; mIWG-MRT-ECNM, modified International Working Group-Myeloproliferative Neoplasms Research and Treatment and European Competence Network on Mastocytosis; MCL, mast cell leukemia; QD, once daily.
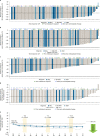
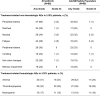
Fig. 2. Clinicopathologic measures of response and change in patient-reported measures of symptom burden by AdvSM-SAF score.
a, BM mast cell burden. b, Serum tryptase level. c, Spleen volume. d, KIT D816V VAF in BM, assessed by central ddPCR assay. e, Change from baseline in systemic mastocytosis symptom burden, evaluated by AdvSM-SAF TSS. For the change in TSS by AdvSM-SAF, a two-sided paired _t_-test was performed for the change from baseline at C3 (P < 0.001), C7 (P = 0.022) and C11 (P = 0.002). BL, baseline; CR, complete remission; VAF, variant allele fraction. AdvSM-SAF, advanced systemic mastocytosis symptom assessment form; ASM, aggressive systemic mastocytosis; BM, bone marrow; C, cycle; CIs, confidence intervals; ddPCR, droplet digital polymerase chain reaction; MCL, mast cell leukemia; SM-AHN, systemic mastocytosis with an associated hematologic neoplasm; TSS, total symptom score.
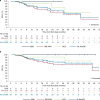
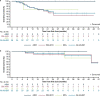
Fig. 3. Kaplan–Meier estimates of overall survival and progression-free survival.
a, PFS in the response-evaluable population. b, OS in the AdvSM safety population. AdvSM, advanced systemic mastocytosis; ASM, aggressive systemic mastocytosis; MCL, mast cell leukemia; SM-AHN, systemic mastocytosis with an associated hematologic neoplasm.
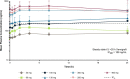
Extended Data Fig. 1. Steady State Mean Plasma Concentration–Time Profiles of Avapritinib in Patients with Systemic Mastocytosis.
The figure shows the mean steady state plasma concentrations of avapritinib assessed over time (Cycle 1 Day 15) in patients who received different doses of avapritinib. At most time points, avapritinib plasma concentrations at doses ≥130 mg exceeded the IC90 of 189 ng/mL for inhibition of KIT D816V measured by immunoblot in a xenograft model. Data are presented as mean values + standard deviation. The number of patients who received each dose are as follows: 30 mg, n = 3; 60 mg, n = 5; 100 mg, n = 3; 130 mg, n = 3; 200 mg, n = 21; 300 mg, n = 43; 400 mg, n = 7. h, hours; IC90, 90% inhibitory concentration; t1/2 terminal half-life.
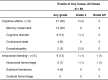
Extended Data Fig. 2. Treatment-Related Adverse Events Reported in ≥15% of Patients.
AEs, adverse events.
Extended Data Fig. 3. Summary of Cognitive Effects and Intracranial Bleeding Events by Grade Occurring at Any Dose (AdvSM Safety Population).
*Two events were associated with head trauma and one occurred in the context of progressive mast cell leukemia. AdvSM, advanced systemic mastocytosis.
Extended Data Fig. 4. Antecedent Dose, Platelet Count, Prothrombin International Normalized Ratio (INR), Activated Partial Thromboplastin Time (aPTT) and Platelet Transfusions in Patients Who had Intracranial Bleeding Events (AdvSM Safety Population).
*Nadir platelet count presented as value during screening/nadir value within 56 days prior to event onset. †Baseline INR and aPTT presented as values during screening/maximum values within 56 days prior to event onset. ‡Total number of units of platelets given by transfusion, after first dose of avapritinib and prior to the onset of a first intracranial bleeding event. AdvSM, advanced systemic mastocytosis; aPTT, activated partial thromboplastin time; ASM, aggressive systemic mastocytosis; CTCAE, Common Terminology Criteria for Adverse Events; INR, prothrombin international normalized ratio; MCL, mast cell leukemia; NR, not recorded; RAC, Response Adjudication Committee; SM-AHN, systemic mastocytosis with an associated hematologic neoplasm.
Extended Data Fig. 5. Adverse Events Leading to Treatment Discontinuation (AdvSM Safety Population, N = 69).
*Events determined to be probably or possibly related to the study medication. †Acute myeloid leukemia is a progressive disease event that was also reported as an AE in these 3 cases. ‡Cognitive disorder and encephalopathy occurred in the same patient. §Staphylococcal sepsis and thrombocytopenia occurred in the same patient. AdvSM, advanced systemic mastocytosis.
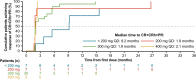
Extended Data Fig. 6. Impact of Starting Dose on Time to Response (CR/CRh/PR) per Modified IWG-MRT-ECNM Criteria Among Responders in the Response-Evaluable Population.
CR, complete remission; CRh, CR with partial recovery of peripheral blood counts; IWG-MRT-ECNM, International Working Group-Myeloproliferative Neoplasms Research and Treatment and European Competence Network on Mastocytosis; QD, once daily; PR, partial remission.
Extended Data Fig. 7. Summary of Clinical Improvement in Measures of Mast Cell Burden (AdvSM Safety Population).
*n values indicate number of patients with neoplastic mast cell aggregates at baseline. †n values indicate number of patients with baseline serum tryptase assessment. ‡Images by CT or MRI centrally assessed by independent radiologist. §Based on central laboratory testing. AdvSM, advanced systemic mastocytosis; ASM, aggressive systemic mastocytosis; CR, complete remission; CT, computed tomography; MCL, mast cell leukemia; MRI, magnetic resonance imaging; PR, partial remission; SM-AHN, systemic mastocytosis with an associated hematologic neoplasm; VAF, variant allele fraction.
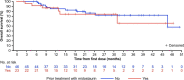
Extended Data Fig. 8. Overall Survival by Prior Midostaurin in the AdvSM Safety Population.
AdvSM, advanced systemic mastocytosis.
Extended Data Fig. 9. Overall Survival by S/A/R genotype in the AdvSM Safety Population.
a, _S/A/R_-positive patients. b, _S/A/R_-negative patients. AdvSM, advanced systemic mastocytosis; ASM, aggressive systemic mastocytosis; MCL, mast cell leukemia; SM-AHN, systemic mastocytosis with an associated hematologic neoplasm; S/A/R, SRSF2, ASXL1, or RUNX1.
Comment in
- Finding the right KIT inhibitor for advanced systemic mastocytosis.
Castells M, Akin C. Castells M, et al. Nat Med. 2021 Dec;27(12):2081-2082. doi: 10.1038/s41591-021-01588-z. Nat Med. 2021. PMID: 34873346 No abstract available. - A new standard for mastocytosis.
Killock D. Killock D. Nat Rev Clin Oncol. 2022 Feb;19(2):71. doi: 10.1038/s41571-021-00592-z. Nat Rev Clin Oncol. 2022. PMID: 34912050 No abstract available.
Similar articles
- Efficacy and safety of avapritinib in previously treated patients with advanced systemic mastocytosis.
Reiter A, Schwaab J, DeAngelo DJ, Gotlib J, Deininger MW, Pettit KM, Alvarez-Twose I, Vannucchi AM, Panse J, Platzbecker U, Hermine O, Dybedal I, Lin HM, Rylova SN, Ehlert K, Dimitrijević S, Radia DH. Reiter A, et al. Blood Adv. 2022 Nov 8;6(21):5750-5762. doi: 10.1182/bloodadvances.2022007539. Blood Adv. 2022. PMID: 35640224 Free PMC article. Clinical Trial. - Efficacy and safety of avapritinib in advanced systemic mastocytosis: interim analysis of the phase 2 PATHFINDER trial.
Gotlib J, Reiter A, Radia DH, Deininger MW, George TI, Panse J, Vannucchi AM, Platzbecker U, Alvarez-Twose I, Mital A, Hermine O, Dybedal I, Hexner EO, Hicks LK, Span L, Mesa R, Bose P, Pettit KM, Heaney ML, Oh ST, Sen J, Lin HM, Mar BG, DeAngelo DJ. Gotlib J, et al. Nat Med. 2021 Dec;27(12):2192-2199. doi: 10.1038/s41591-021-01539-8. Epub 2021 Dec 6. Nat Med. 2021. PMID: 34873345 Free PMC article. - Avapritinib for advanced systemic mastocytosis.
Gotlib J, Reiter A, DeAngelo DJ. Gotlib J, et al. Blood. 2022 Oct 13;140(15):1667-1673. doi: 10.1182/blood.2021014612. Blood. 2022. PMID: 35877999 - Response Criteria in Advanced Systemic Mastocytosis: Evolution in the Era of KIT Inhibitors.
Shomali W, Gotlib J. Shomali W, et al. Int J Mol Sci. 2021 Mar 15;22(6):2983. doi: 10.3390/ijms22062983. Int J Mol Sci. 2021. PMID: 33804174 Free PMC article. Review. - Indirect treatment comparisons of avapritinib versus midostaurin for patients with advanced systemic mastocytosis.
Pilkington H, Smith S, Roskell N, Iannazzo S. Pilkington H, et al. Future Oncol. 2022 Apr;18(13):1583-1594. doi: 10.2217/fon-2021-1509. Epub 2022 Feb 4. Future Oncol. 2022. PMID: 35114819
Cited by
- Drugs in the GIST Field (Therapeutic Targets and Clinical Trial Staging).
Huang C, Ma X, Wang M, Cao H. Huang C, et al. Curr Drug Deliv. 2024;21(1):80-90. doi: 10.2174/1567201820666221122120657. Curr Drug Deliv. 2024. PMID: 36415101 Free PMC article. - Efficacy and safety of avapritinib in previously treated patients with advanced systemic mastocytosis.
Reiter A, Schwaab J, DeAngelo DJ, Gotlib J, Deininger MW, Pettit KM, Alvarez-Twose I, Vannucchi AM, Panse J, Platzbecker U, Hermine O, Dybedal I, Lin HM, Rylova SN, Ehlert K, Dimitrijević S, Radia DH. Reiter A, et al. Blood Adv. 2022 Nov 8;6(21):5750-5762. doi: 10.1182/bloodadvances.2022007539. Blood Adv. 2022. PMID: 35640224 Free PMC article. Clinical Trial. - Allogeneic haematopoietic cell transplantation for advanced systemic mastocytosis: Best practice recommendations on behalf of the EBMT Practice Harmonisation and Guidelines Committee.
McLornan DP, Czerw T, Damaj G, Ethell M, Gurnari C, Hernández-Boluda JC, Polverelli N, Schwaab J, Sockel K, Raffaella G, Onida F, Sánchez-Ortega I, Battipaglia G, Elena C, Gotlib J, Reiter A, Rossignol J, Ustun C, Valent P, Yakoub-Agha I, Radia DH. McLornan DP, et al. Leukemia. 2024 Apr;38(4):699-711. doi: 10.1038/s41375-024-02182-1. Epub 2024 Mar 12. Leukemia. 2024. PMID: 38472477 Review. - CDK4/CDK6 Inhibitors Synergize with Midostaurin, Avapritinib, and Nintedanib in Inducing Growth Inhibition in KIT D816V+ Neoplastic Mast Cells.
Schneeweiss-Gleixner M, Filik Y, Stefanzl G, Berger D, Sadovnik I, Bauer K, Smiljkovic D, Eisenwort G, Witzeneder N, Greiner G, Hoermann G, Schiefer AI, Schwaab J, Jawhar M, Reiter A, Sperr WR, Arock M, Valent P, Gleixner KV. Schneeweiss-Gleixner M, et al. Cancers (Basel). 2022 Jun 23;14(13):3070. doi: 10.3390/cancers14133070. Cancers (Basel). 2022. PMID: 35804842 Free PMC article. - Avapritinib treatment of aggressive systemic mastocytosis with a novel KIT exon 17 mutation.
Sandow L, Town A, Heinrich MC. Sandow L, et al. Leuk Res Rep. 2023 Dec 30;21:100409. doi: 10.1016/j.lrr.2023.100409. eCollection 2024. Leuk Res Rep. 2023. PMID: 38273969 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- P01 CA214278/CA/NCI NIH HHS/United States
- P30 CA016672/CA/NCI NIH HHS/United States
- UL1 TR001878/TR/NCATS NIH HHS/United States
- R01 HL148014/HL/NHLBI NIH HHS/United States
- UE5 CA246744/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous