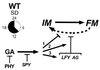Flowers into shoots: photo and hormonal control of a meristem identity switch in Arabidopsis - PubMed (original) (raw)
Flowers into shoots: photo and hormonal control of a meristem identity switch in Arabidopsis
J K Okamuro et al. Proc Natl Acad Sci U S A. 1996.
Abstract
Little is known about the signals that govern the network of meristem and organ identity genes that control flower development. In Arabidopsis, we can induce a heterochronic switch from flower to shoot development, a process known as floral meristem reversion, by manipulating photo-period in the floral homeotic mutant agamous and in plants heterozygous for the meristem identity gene leafy. The transformation from flower to shoot meristem is suppressed by hy1, a mutation blocking phytochrome activity, by spindly, a mutation that activates basal gibberellin signal transduction in a hormone independent manner, or by the exogenous application of gibberellins. We propose that LFY and AG play an important role in the maintenance of flower meristem identity and that floral meristem reversion in heterozygous lfy and in ag flowers is regulated by a phytochrome and gibberellin signal transduction cascade.
Figures
Figure 1
Photoperiod controls floral meristem activity in_lfy-6_ and ag-1 flowers. (A) LD homozygous lfy-6 flower. lfy-6 flowers show a basipetal to acropetal gradient of phenotypes (16). A typical acropetal LD lfy-6 flower is characterized by the production of sepals (s) and carpels (c) and the absence of petals and stamens. (B) SD homozygous lfy-6 flower.lfy flower development is suppressed under SDs (12, 15). A typical SD “flower” produces an indeterminate number of leaf-like organs (l). (C_–_F) SD heterozygous lfy-6 flower development. Heterozygous_lfy-6_ flowers produce a normal complement of organs (C) and are initially indistinguishable from wild type. Later, basipetal flowers are distinguished by a dramatically swollen ovary (so) (D). The swollen ovary often ruptures and a single flower-bearing ectopic shoot (es) emerges (E). The resulting shoot (es) produces several leaf-like organs, numerous flowers, displays internode elongation, and a spiral pattern of phyllotaxis (F). Flowers produced by the ectopic shoot appear either wild type or resemble ap2 mutant flowers. (G) LD ag-1 flower. ag-1 flowers are characterized by the transformation of stamens into petals and an indeterminate pattern of floral organogenesis (sepal, petal, petal)n. (H and I) SD_ag-1_ flower development. Basipetal SD_ag-1_ flowers initially produce several whorls of sepals and petals (H). Organogenesis then ceases and is replaced by the production of an ectopic shoot (es) that bears numerous flowers and displays internode elongation (I). (J_–_L) Ectopic shoot formation in basipetal SD heterozygous lfy-6 flowers. Longitudinal sections (2 μm) were prepared as described in Materials and Methods. An ectopic shoot (es) arises directly from the apical dome of the floral meristem (J). A magnified view of the ectopic shoot is shown in K. At this stage of development the ectopic shoot produces leaf-like organs (l). The ectopic shoot continues to grow and eventually fills the ovary (L). ov, The ovary wall. (Bar = 0.1 mm.) (M) Wild-type flower. Shown is a longitudinal section (2 μm) through a SD wild-type flower. Floral meristem activity is suppressed after carpel primordia are established. (Bar = 0.1 mm.) (N and_O_) Ectopic shoot formation in SD ag-1 flowers. A 2-μm longitudinal section through a SD basipetal_ag-1_ flower shows floral buds (fb) emerging from the apex of the floral meristem (N). A LD ag-1 flower is shown for comparison (O). (Bar = 0.1 mm.) (P_–_R) AG gene expression in SD heterozygous lfy-6 flowers. The spatial pattern of_AG_ gene expression in SD heterozygous lfy-6(P and Q) and wild-type flowers (R) was determined by in situ hybridization as described. The presence of AG transcripts is revealed by the orange staining. The flowers in P,Q, and R are at stages 3, 8, and 10, respectively (28). es, The ectopic shoot primordium. (Bar = 0.1 mm.)
Figure 2
Frequency of floral meristem reversion exhibits a basipetal-acropetal gradient. (A) Floral meristem reversion in SD heterozygous lfy-6 flowers. (B) Floral meristem reversion in SD ag-1 flowers.
Figure 3
A speculative model for the photo and hormonal control of the maintenance of floral meristem identity by_LFY_ and AG. In wild-type plants cells produced by the inflorescence meristem (IM) are committed to flower meristem development (FM) by a network of genes including_AP1_, AP2, CAL, CLV1, CLV3, LFY, and others. Under SDs floral meristem reversion, represented by the broken arrow from FM to IM, is strongly but not totally repressed by_LFY_ and AG (see Table 1). GA signal transduction is negatively controlled by SPY. GA synthesis or activity is negatively regulated by phytochrome (PHY). This model suggests three possible pathways for the maintenance of flower meristem identity by gibberellin (GA) signal transduction as shown by arrows 1–3 (see text).
Similar articles
- Photo and hormonal control of meristem identity in the Arabidopsis flower mutants apetala2 and apetala1.
Okamuro JK, Szeto W, Lotys-Prass C, Jofuku KD. Okamuro JK, et al. Plant Cell. 1997 Jan;9(1):37-47. doi: 10.1105/tpc.9.1.37. Plant Cell. 1997. PMID: 9014363 Free PMC article. - ULTRAPETALA1 and LEAFY pathways function independently in specifying identity and determinacy at the Arabidopsis floral meristem.
Engelhorn J, Moreau F, Fletcher JC, Carles CC. Engelhorn J, et al. Ann Bot. 2014 Nov;114(7):1497-505. doi: 10.1093/aob/mcu185. Epub 2014 Oct 6. Ann Bot. 2014. PMID: 25288633 Free PMC article. - Regulatory networks that function to specify flower meristems require the function of homeobox genes PENNYWISE and POUND-FOOLISH in Arabidopsis.
Kanrar S, Bhattacharya M, Arthur B, Courtier J, Smith HM. Kanrar S, et al. Plant J. 2008 Jun;54(5):924-37. doi: 10.1111/j.1365-313X.2008.03458.x. Epub 2008 Feb 23. Plant J. 2008. PMID: 18298668 - Genetic regulation of time to flower in Arabidopsis thaliana.
Komeda Y. Komeda Y. Annu Rev Plant Biol. 2004;55:521-35. doi: 10.1146/annurev.arplant.55.031903.141644. Annu Rev Plant Biol. 2004. PMID: 15377230 Review. - The poetry of reproduction: the role of LEAFY in Arabidopsis thaliana flower formation.
Siriwardana NS, Lamb RS. Siriwardana NS, et al. Int J Dev Biol. 2012;56(4):207-21. doi: 10.1387/ijdb.113450ns. Int J Dev Biol. 2012. PMID: 22451042 Review.
Cited by
- Floral meristem initiation and emergence in plants.
Chandler JW. Chandler JW. Cell Mol Life Sci. 2012 Nov;69(22):3807-18. doi: 10.1007/s00018-012-0999-0. Epub 2012 May 10. Cell Mol Life Sci. 2012. PMID: 22573183 Free PMC article. Review. - FPF1 promotes flowering in Arabidopsis.
Kania T, Russenberger D, Peng S, Apel K, Melzer S. Kania T, et al. Plant Cell. 1997 Aug;9(8):1327-38. doi: 10.1105/tpc.9.8.1327. Plant Cell. 1997. PMID: 9286110 Free PMC article. - Control of flowering and cell fate by LIF2, an RNA binding partner of the polycomb complex component LHP1.
Latrasse D, Germann S, Houba-Hérin N, Dubois E, Bui-Prodhomme D, Hourcade D, Juul-Jensen T, Le Roux C, Majira A, Simoncello N, Granier F, Taconnat L, Renou JP, Gaudin V. Latrasse D, et al. PLoS One. 2011 Jan 31;6(1):e16592. doi: 10.1371/journal.pone.0016592. PLoS One. 2011. PMID: 21304947 Free PMC article. - Characterization of MdMADS2, a member of the SQUAMOSA subfamily of genes, in apple.
Sung SK, Yu GH, An G. Sung SK, et al. Plant Physiol. 1999 Aug;120(4):969-78. doi: 10.1104/pp.120.4.969. Plant Physiol. 1999. PMID: 10444080 Free PMC article. - Molecular and genetic mechanisms of floral control.
Jack T. Jack T. Plant Cell. 2004;16 Suppl(Suppl):S1-17. doi: 10.1105/tpc.017038. Epub 2004 Mar 12. Plant Cell. 2004. PMID: 15020744 Free PMC article. Review. No abstract available.
References
- Medford J I, Callos J D, Behringer F J, Link B M. In: Arabidopsis. Meyerowitz E M, Somerville C R, editors. Plainview, NY: Cold Spring Harbor Lab. Press; 1994. pp. 355–378.
- Sussex I M. Cell. 1989;56:225–229. - PubMed
- Clark S E, Running M P, Meyerowitz E M. Development (Cambridge, UK) 1993;119:397–418. - PubMed
- Laibach F. Beitr Biol Pflanz. 1951;28:173–210.
- Gregory F G, Hussey G G. Proc Linn Soc London. 1953;164:137–139.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases