Membrane Fas ligand kills human peripheral blood T lymphocytes, and soluble Fas ligand blocks the killing - PubMed (original) (raw)
Membrane Fas ligand kills human peripheral blood T lymphocytes, and soluble Fas ligand blocks the killing
T Suda et al. J Exp Med. 1997.
Abstract
It has been believed that the Fas expressed on human peripheral blood T cells (PBT) is nonfunctional, because these cells are insensitive to agonistic anti-Fas/Apo-1 mAbs that efficiently kill in vitro-activated T cells and many Fas-expressing cell lines. Here, we demonstrate that membrane-bound Fas ligand (FasL) kills both fresh and in vitro-activated PBT, indicating that the Fas expressed on fresh PBT is functional. In contrast, soluble FasL kills only the latter. Naive T cells in umbilical cord blood do not express Fas, but can be induced to express Fas by IFN-gamma or by a combination of IL-2 and anti-CD28 mAb, after which they acquire sensitivity to membrane but not to soluble FasL. Soluble FasL inhibited the killing of fresh PBT by membrane FasL. These results indicate that the shedding of FasL from the membrane is a mechanism for downregulating at least part of its killing activity.
Figures
Figure 1
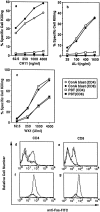
Susceptibility of fresh PBT and ConA blasts to anti-Fas mAb (CH11), soluble human FasL (AL-1), and recombinant soluble mouse FasL (WX1). (a–c), Fresh PBL or ConA blasts were cultured in the presence of the indicated concentrations of CH11 (a), AL-1 (b), and WX1 (c) for 14 h. Cells were then stained with FITC-anti-Fas mAb, PE-conjugated mAb against either CD4 or CD8, and PI. Percent specific cell killing of CD4+ and CD8+ cells was determined as described in Materials and Methods. (d–g) Fresh PBL (d and e) and ConA blasts (f and g) cultured with (solid line) or without (dotted line) 4,000 units/ml of WX1 for 14 h were stained as described above, and 5 × 104 total cells (including both viable and dead cells) were analyzed in a FACScan®. Staining profiles for FITC-anti-Fas mAb of viable (PI−) CD4+ (d and f) or CD8+ cells (e and g) are shown.
Figure 2
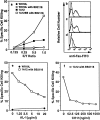
Membrane FasL kills fresh PBT and soluble FasL inhibits the killing. (a) Fresh PBL were cultured with the transfectant expressing human FasL (1A12) or its parental cell line (WR19L) at the indicated effector/target (E/T) ratios for 12 h in the presence or absence of 10 μM BB2116. Cells were then stained with FITC anti-Fas, PE-anti-CD3 mAb and PI, and analyzed in a FACScan®. (b–d) FITC anti-Fas mAb staining profiles for PI−CD3+ cells after culturing with WR19L in the absence of BB2116 (b), or with 1A12 in the absence (c) or presence (d) of BB2116 at an E/T ratio of 1.0 are shown. Dotted profiles indicate negative controls stained with FITC-labeled mouse IgG1. (e and f). Fresh PBL were cultured with 1A12 plus 10 mM BB2116 or with WR19L cells at the E/T ratio of 1.0 in the presence of the indicated concentrations of purified AL-1 (e) or CH11 (f) for 12 h.
Figure 3
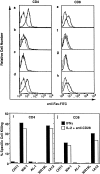
Function of Fas on CBT induced by IFN-γ or a combination of IL-2 and immobilized anti-CD28 mAb. (a–h) Cord blood lymphocytes were cultured for 20 h with medium only (dotted line in each panel), IFN–γ (10 ng/ml, a and e), IL-2 (10 ng/ml, b and f), immobilized anti-CD28 mAb (coated on plate at 10 μg/ml, c and g), or IL-2 plus immobilized anti-CD28 mAb (d and h), respectively. Cells were then stained with FITC–anti-Fas mAb, PE-conjugated mAb against either CD4 or CD8, and PI. FITC–anti-Fas mAb staining profiles of viable (PI−) CD4+ (a–d) or CD8+ cells (e–h) are shown. (i and j) CBT treated with IFN-γ or with IL-2 plus anti-CD28 mAb as described above were cultured with CH11, WX1, AL-1, or 1A12.
Similar articles
- Interferon alpha augments activation-induced T cell death by upregulation of Fas (CD95/APO-1) and Fas ligand expression.
Kaser A, Nagata S, Tilg H. Kaser A, et al. Cytokine. 1999 Oct;11(10):736-43. doi: 10.1006/cyto.1998.0484. Cytokine. 1999. PMID: 10525311 - CD4+ T-cell-mediated cytotoxicity against staphylococcal enterotoxin B-pulsed synovial cells.
Kawakami A, Matsuoka N, Tsuboi M, Urayama S, Nakashima T, Kawabe Y, Koji T, Aoyagi T, Maeda K, Eguchi K. Kawakami A, et al. Immunology. 1998 Sep;95(1):38-46. doi: 10.1046/j.1365-2567.1998.00569.x. Immunology. 1998. PMID: 9767455 Free PMC article. - Crosstalk between keratinocytes and T lymphocytes via Fas/Fas ligand interaction: modulation by cytokines.
Arnold R, Seifert M, Asadullah K, Volk HD. Arnold R, et al. J Immunol. 1999 Jun 15;162(12):7140-7. J Immunol. 1999. PMID: 10358159 - On the role and significance of Fas (Apo-1/CD95) ligand (FasL) expression in immune privileged tissues and cancer cells using multiple myeloma as a model.
Greil R, Egle A, Villunger A. Greil R, et al. Leuk Lymphoma. 1998 Nov;31(5-6):477-90. doi: 10.3109/10428199809057607. Leuk Lymphoma. 1998. PMID: 9922038 Review. - Soluble HLA class I molecules/CD8 ligation trigger apoptosis of CD8+ cells by Fas/Fas-ligand interaction.
Puppo F, Contini P, Ghio M, Indiveri F. Puppo F, et al. ScientificWorldJournal. 2002 Feb 12;2:421-3. doi: 10.1100/tsw.2002.122. ScientificWorldJournal. 2002. PMID: 12806026 Free PMC article. Review.
Cited by
- Killer B lymphocytes and their fas ligand positive exosomes as inducers of immune tolerance.
Lundy SK, Klinker MW, Fox DA. Lundy SK, et al. Front Immunol. 2015 Mar 20;6:122. doi: 10.3389/fimmu.2015.00122. eCollection 2015. Front Immunol. 2015. PMID: 25852690 Free PMC article. - Multimeric soluble CD40 ligand and GITR ligand as adjuvants for human immunodeficiency virus DNA vaccines.
Stone GW, Barzee S, Snarsky V, Kee K, Spina CA, Yu XF, Kornbluth RS. Stone GW, et al. J Virol. 2006 Feb;80(4):1762-72. doi: 10.1128/JVI.80.4.1762-1772.2006. J Virol. 2006. PMID: 16439533 Free PMC article. - Importance of TRAIL Molecular Anatomy in Receptor Oligomerization and Signaling. Implications for Cancer Therapy.
Naval J, de Miguel D, Gallego-Lleyda A, Anel A, Martinez-Lostao L. Naval J, et al. Cancers (Basel). 2019 Mar 29;11(4):444. doi: 10.3390/cancers11040444. Cancers (Basel). 2019. PMID: 30934872 Free PMC article. Review. - Osteoprotegerin Induces Apoptosis of Osteoclasts and Osteoclast Precursor Cells via the Fas/Fas Ligand Pathway.
Liu W, Xu C, Zhao H, Xia P, Song R, Gu J, Liu X, Bian J, Yuan Y, Liu Z. Liu W, et al. PLoS One. 2015 Nov 16;10(11):e0142519. doi: 10.1371/journal.pone.0142519. eCollection 2015. PLoS One. 2015. PMID: 26571489 Free PMC article. - Protein Palmitoylation in Leukocyte Signaling and Function.
Yang X, Chatterjee V, Ma Y, Zheng E, Yuan SY. Yang X, et al. Front Cell Dev Biol. 2020 Oct 28;8:600368. doi: 10.3389/fcell.2020.600368. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33195285 Free PMC article. Review.
References
- Nagata S, Suda T. Fas and Fas ligand: lpr and gldmutations. Immunol Today. 1995;16:39–43. - PubMed
- Bellgrau D, Gold D, Selawry H, Moore J, Franzusoff A, Duke RC. A role for CD95 ligand in preventing graft rejection. Nature. 1995;377:630–632. - PubMed
- Griffith TS, Brunner T, Fletcher SM, Green DR, Ferguson TA. Fas ligand-induced apoptosis as a mechanism of immune privilege. Science. 1995;270:1189–1192. - PubMed
- Nagata S. Apoptosis by death factor. Cell. 1997;88:355–365. - PubMed
- Kondo T, Suda T, Fukuyama H, Adachi M, Nagata S. Essential roles of the Fas ligand in the development of hepatitis. Nat Med. 1997;3:409–413. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous