Generation of mucosal cytotoxic T cells against soluble protein by tissue-specific environmental and costimulatory signals - PubMed (original) (raw)
Generation of mucosal cytotoxic T cells against soluble protein by tissue-specific environmental and costimulatory signals
S K Kim et al. Proc Natl Acad Sci U S A. 1998.
Abstract
We compared peripheral and mucosal primary CD8 T cell responses to inflammatory and noninflammatory forms of antigen in a T cell-adoptive transfer system. Immunization with the soluble antigen, ovalbumin (ova), administered i.p. or orally without adjuvant, activated nonmucosal CD8 T cells but did not induce cytotoxic activity. However, after activation, the transferred cells entered the intestinal mucosa and became potent antigen-specific killers. Thus, exogenous intact soluble protein entered the major histocompatibility complex class I antigen presentation pathway and induced mucosal cytotoxic T lymphocytes. Moreover, distinct costimulatory requirements for activation of peripheral versus mucosal T cells were noted in that the CD28 ligand, B7-1, was critical for activated mucosal T cell generation but not for activation of peripheral CD8 T cells. The costimulator, B7-2, was required for optimum activation of both populations. Infection with a new recombinant vesicular stomatitis virus encoding ovalbumin induced lytic activity in mucosal as well as peripheral sites, demonstrating an adjuvant effect of inflammatory mediators produced during virus infection. Generation of antiviral cytotoxic T lymphocytes was also costimulation-dependent. The results indicated that induction of peripheral tolerance via antigen administration may not extend to mucosal sites because of distinct costimulatory and inflammatory signals in the mucosa.
Figures
Figure 1
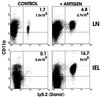
Appearance of CD8 T cells in the intestinal mucosa after activation of transferred T cells. The adoptive transfer method was modified from Kearney et al. (17). C57BL/6-Ly5.2 OT-I T cells (1 × 107) were injected i.v. into C57BL/6-Ly5.1 mice. Two days later 5 mg of ovalbumin (+ antigen) was administered i.p. Three days later cells were isolated and analyzed for the presence of transferred cells (Ly5.2+) and CD11a expression by fluorescence flow cytometry. Total donor cell numbers were calculated by multiplying the number of Ly5.2+ cells by the total number of LN cells isolated. This experiment has been performed at least 10 times with similar results.
Figure 2
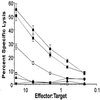
Immunization with soluble antigen induces primary CTL in the intestinal mucosa but not in the periphery. OT-I or OT-I-RAG−/− T cells (1 × 107) were transferred to normal C57BL/6 mice and, 2 days later, were either injected i.p. with 5 mg ova or fed 10 mg ova by intubation. Four days later cells were isolated and cytolytic activity was measured by using [51Cr] sodium chromate-labeled EL4 cells (an H-2b thymoma) with or without the addition of 10 μg/ml of the ova-derived peptide SIINFEKL. Serial dilutions of effector cells were incubated in 96-well round-bottom microtiter plates with 2.5 × 103 target cells for 6 hr at 37°C. Percentage of specific lysis was calculated as: 100 × [(cpm released with effectors) − (cpm released alone)]/[(cpm released by detergent) − (cpm released alone)]. (A) i.p. injection of ova. Open symbols, EL4 no peptide; closed symbols, EL4 with peptide. (B) Open circles, fed antigen; closed circles, i.p. injection; closed squares, LN cells from i.p. injected mouse; all symbols, EL4 with peptide as targets. Lytic activity without peptide added was <5%. Effector-to-target ratios were adjusted to actual donor cell percentages. This experiment has been performed eight times with similar results.
Figure 3
Costimulatory requirements for activation of adoptively transferred CD8 T cells. C57BL/6-Ly5.2 OT-I T cells (1 × 107) were injected i.v. into C57BL/6-Ly5.1 mice. Two days later 5 mg of ovalbumin was administered i.p. One hundred micrograms of the indicated antibody was injected i.p. daily beginning on the day of immunization. Three days after immunization cells were isolated and analyzed for Ly5.2 and CD44 expression by fluorescence flow cytometry. Numbers in parentheses indicate the mean fluorescence intensity of CD44 expression for the donor cells. Numbers above the plots indicate the total number of donor cells. This experiment has been performed three times with similar results.
Figure 4
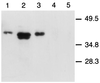
Recombinant VSV containing the ovalbumin gene directs secretion of ovalbumin in infected cells. Western blot analysis of ova expression with purified ova (80 ng) (lane 1), VSV-ova-infected BHK cell lysates (lane 2), supernatant from VSV-ova-infected BHK cells (lane 3), wild-type VSV-infected BHK cell lysates (lane 4), or supernatant (lane 5).
Figure 5
Infection with VSV-ova activates ova-specific T cells and induces CD8 T cell migration to the intestinal mucosa. C57BL/6-Ly5.2 OT-I T cells (2.5 × 106) were transferred to C57BL/6 mice, and 2 days later mice were infected by i.v. injection with 1 × 106 pfu of VSV-ova. One hundred micrograms of the indicated reagent was injected i.p. daily beginning on the day of immunization. Five days later cells were isolated and analyzed for expression of Ly5.2 and CD11a expression by fluorescence flow cytometry.
Figure 6
Infection with recombinant VSV-ova induces anti-ova peripheral and mucosal primary CTL. VSV-ova induces primary anti-ova CTL in LN T cells and IEL. LN cells or IEL from CTLA4-Ig-mut-treated mice (■, LN; •, IEL) or CTLA4-Ig treated mice (□, LN; ○, IEL) were tested for lytic activity against ova-peptide-coated EL4 cells. LN cells and IEL from CTLA4-Ig-mut-treated mice reacted with EL4 cells in the absence of peptide (▴, IEL; ▾, LN). Effector-to-target ratios were adjusted to actual donor cell percentages found in CTLA4-Ig-mut-treated mice. This experiment has been performed three times with similar results. SE bars are shown.
Similar articles
- Soluble antigen and CD40 triggering are sufficient to induce primary and memory cytotoxic T cells.
Lefrançois L, Altman JD, Williams K, Olson S. Lefrançois L, et al. J Immunol. 2000 Jan 15;164(2):725-32. doi: 10.4049/jimmunol.164.2.725. J Immunol. 2000. PMID: 10623816 - The role of B7-1 and B7-2 costimulation for the generation of CTL responses in vivo.
Sigal LJ, Reiser H, Rock KL. Sigal LJ, et al. J Immunol. 1998 Sep 15;161(6):2740-5. J Immunol. 1998. PMID: 9743331 - B7 costimulation is necessary for the activation of the lytic function in cytotoxic T lymphocyte precursors.
Guerder S, Carding SR, Flavell RA. Guerder S, et al. J Immunol. 1995 Dec 1;155(11):5167-74. J Immunol. 1995. PMID: 7594526 - Lentiviral Protein Transfer Vectors Are an Efficient Vaccine Platform and Induce a Strong Antigen-Specific Cytotoxic T Cell Response.
Uhlig KM, Schülke S, Scheuplein VA, Malczyk AH, Reusch J, Kugelmann S, Muth A, Koch V, Hutzler S, Bodmer BS, Schambach A, Buchholz CJ, Waibler Z, Scheurer S, Mühlebach MD. Uhlig KM, et al. J Virol. 2015 Sep;89(17):9044-60. doi: 10.1128/JVI.00844-15. Epub 2015 Jun 17. J Virol. 2015. PMID: 26085166 Free PMC article. - Induction and visualization of mucosal memory CD8 T cells following systemic virus infection.
Kim SK, Schluns KS, Lefrançois L. Kim SK, et al. J Immunol. 1999 Oct 15;163(8):4125-32. J Immunol. 1999. PMID: 10510347
Cited by
- Tissue-resident memory T cells break tolerance to renal autoantigens and orchestrate immune-mediated nephritis.
Arnold F, Kupferschmid L, Weissenborn P, Heldmann L, Hummel JF, Zareba P, Sagar, Rogg M, Schell C, Tanriver Y. Arnold F, et al. Cell Mol Immunol. 2024 Sep;21(9):1066-1081. doi: 10.1038/s41423-024-01197-z. Epub 2024 Jul 3. Cell Mol Immunol. 2024. PMID: 38961265 Free PMC article. - Combined IL-15/IL-15Ralpha immunotherapy maximizes IL-15 activity in vivo.
Stoklasek TA, Schluns KS, Lefrançois L. Stoklasek TA, et al. J Immunol. 2006 Nov 1;177(9):6072-80. doi: 10.4049/jimmunol.177.9.6072. J Immunol. 2006. PMID: 17056533 Free PMC article. - IL-15R alpha expression on CD8+ T cells is dispensable for T cell memory.
Burkett PR, Koka R, Chien M, Chai S, Chan F, Ma A, Boone DL. Burkett PR, et al. Proc Natl Acad Sci U S A. 2003 Apr 15;100(8):4724-9. doi: 10.1073/pnas.0737048100. Epub 2003 Apr 1. Proc Natl Acad Sci U S A. 2003. PMID: 12671073 Free PMC article. - Exploring regulatory mechanisms of CD8+ T cell contraction.
Prlic M, Bevan MJ. Prlic M, et al. Proc Natl Acad Sci U S A. 2008 Oct 28;105(43):16689-94. doi: 10.1073/pnas.0808997105. Epub 2008 Oct 22. Proc Natl Acad Sci U S A. 2008. PMID: 18946035 Free PMC article. - NFκB-Pim-1-Eomesodermin axis is critical for maintaining CD8 T-cell memory quality.
Knudson KM, Pritzl CJ, Saxena V, Altman A, Daniels MA, Teixeiro E. Knudson KM, et al. Proc Natl Acad Sci U S A. 2017 Feb 28;114(9):E1659-E1667. doi: 10.1073/pnas.1608448114. Epub 2017 Feb 13. Proc Natl Acad Sci U S A. 2017. PMID: 28193872 Free PMC article.
References
- Weiner H L, Friedman A, Miller A, Khoury S J, al Sabbagh A, Santos L, Sayegh M, Nussenblatt R B, Trentham D E. Annu Rev Immunol. 1994;12:809–837. - PubMed
- Lefrancois L, Fuller B, Huleatt J W, Olson S, Puddington L. Springer Semin Immunopathol. 1997;18:463–475. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials