Very late DNA replication in the human cell cycle - PubMed (original) (raw)
Very late DNA replication in the human cell cycle
R J Widrow et al. Proc Natl Acad Sci U S A. 1998.
Abstract
G2 was defined originally as the temporal gap between the termination of DNA replication and the beginning of mitosis. In human cells, the G2 period was estimated to be 3-4 h. However, the absence of replicative DNA synthesis during this period designated G2 has never been shown conclusively. In this report, we show that, at some autosomal and X linked loci, programmed DNA replication continues within 90 min of mitosis. Furthermore, the major accumulation of cyclin B1, a cell-cycle marker that is usually ascribed to G2, overlaps extensively with very late DNA replication. We conclude that the G2 period is much shorter than previously thought and may, in some cells, be nonexistent.
Figures
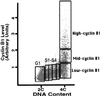
Figure 1
Cell-cycle fractionation by cyclin B1 flow cytometry. Human lymphoblastoid cells were labeled with BrdUrd for 90 min (5), fixed, and separated on the basis of DNA content and cyclin B1 levels. The cyclin B1 units are arbitrary. Equal numbers of cells (between 5,000 and 10,000) were collected from each sorting fraction (G1; S1–S4; and low, mid-, and high cyclin B1) and used for subsequent analysis of replication timing (modified from ref. 9).
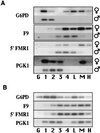
Figure 2
Detection of very late DNA replication for loci on the inactive X chromosome. (A) Newly replicated DNA at four loci on the inactive X chromosome is detected in lymphoblasts recovered from all three cyclin B1 fractions. Replication-timing profiles for several X linked loci in male and female cell lines are shown. The high cyclin B1 fraction contains on average 24% of the replication signal for loci on the inactive X chromosome. (B) In phytohemagglutinin-stimulated lymphocytes, newly replicated DNA at four loci on the inactive X chromosome is detected in cells recovered from all three cyclin B1 fractions. The high cyclin B1 fraction contains on average 22% of the replication signal for loci on the inactive X chromosome. The detection of newly replicated DNA in the high cyclin B1 fraction demonstrates that the replication of sequences close to mitosis in normal human cells is not a consequence of cell transformation.
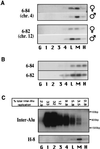
Figure 3
Detection of very late DNA replication for autosomal loci. (A) Replication of two inter-Alu sequences located on chromosomes 4 and 12 is highly restricted and very late in the cell cycle in lymphoblasts. The high cyclin B1 fraction contains on average 13% of the replication signal for these loci. (B) In phytohemagglutinin-stimulated lymphocytes, replication of the same autosomal inter-Alu sequences described in A is also highly restricted and very late in the cell cycle. In this case, the high cyclin B1 fraction contains 26% of the replication signal for these autosomal sequences. (C) Replication timing for a heterogeneous population of inter-Alu sequences is spread throughout the cell cycle in lymphoblasts, with about 1% occurring within the high cyclin B1 fraction. The percentage of the total inter-Alu replication detected by phosphorimager analysis is shown for each cell-cycle fraction. The same membrane was stripped of the labeled primer and reprobed with a specific late replicating inter-Alu sequence found on the X chromosome (H-8); the results demonstrated the specificity of this approach. An alternative quantification method in which radioactive deoxyribonucleoside triphosphates were incorporated during the PCR gave similar results.
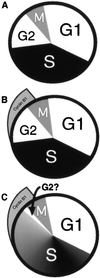
Figure 4
The cell cycle. (A) The textbook view: the cell cycle is divided into four phases; G1, S phase, G2, and mitosis. (B) A more recent view: the accumulation of cyclin B1 identifies a cell as residing in G2 or early mitosis. (C) Our proposed revision: low levels of replicative DNA synthesis (shading) are ongoing much later in the cell cycle than previously suspected, overlapping extensively with the accumulation of cyclin B1. In some cells, the existence of a discrete G2 phase is questioned.
Similar articles
- Separation of cells at different times within G2 and mitosis by cyclin B1 flow cytometry.
Widrow RJ, Rabinovitch PS, Cho K, Laird CD. Widrow RJ, et al. Cytometry. 1997 Mar 1;27(3):250-4. doi: 10.1002/(sici)1097-0320(19970301)27:3<250::aid-cyto6>3.0.co;2-i. Cytometry. 1997. PMID: 9041113 - Estimation of kinetic cell-cycle-related gene expression in G1 and G2 phases from immunofluorescence flow cytometry data.
Jacobberger JW, Sramkoski RM, Wormsley SB, Bolton WE. Jacobberger JW, et al. Cytometry. 1999 Mar 1;35(3):284-9. doi: 10.1002/(sici)1097-0320(19990301)35:3<284::aid-cyto12>3.0.co;2-k. Cytometry. 1999. PMID: 10082310 - Effect of melphalan and hyperthermia on cell cycle progression and cyclin B1 expression in human melanoma cells.
Orlandi L, Zaffaroni N, Bearzatto A, Costa A, Supino R, Vaglini M, Silvestrini R. Orlandi L, et al. Cell Prolif. 1995 Nov;28(11):617-30. doi: 10.1111/j.1365-2184.1995.tb00049.x. Cell Prolif. 1995. PMID: 8555374 - Translational control during mitosis.
Le Breton M, Cormier P, Bellé R, Mulner-Lorillon O, Morales J. Le Breton M, et al. Biochimie. 2005 Sep-Oct;87(9-10):805-11. doi: 10.1016/j.biochi.2005.04.014. Biochimie. 2005. PMID: 15951098 Review. - Regulation of the G2/M transition by p53.
Taylor WR, Stark GR. Taylor WR, et al. Oncogene. 2001 Apr 5;20(15):1803-15. doi: 10.1038/sj.onc.1204252. Oncogene. 2001. PMID: 11313928 Review.
Cited by
- Onset and sequence of RBA-band replication on the inactive X-chromosomes of cattle (Bos taurus L.), river buffalo (Bubalus bubalis L.) and goat (Capra hircus L.).
Di Berardino D, Coppola G, Verdoliva C, Coppola GF, Ramunno L, Enne G, Di Meo GP, Iannuzzi L. Di Berardino D, et al. Chromosome Res. 2002;10(8):637-43. doi: 10.1023/a:1021560225409. Chromosome Res. 2002. PMID: 12575792 - Fragility Extraordinaire: Unsolved Mysteries of Chromosome Fragile Sites.
Feng W, Chakraborty A. Feng W, et al. Adv Exp Med Biol. 2017;1042:489-526. doi: 10.1007/978-981-10-6955-0_21. Adv Exp Med Biol. 2017. PMID: 29357071 Free PMC article. Review. - SIR2 suppresses replication gaps and genome instability by balancing replication between repetitive and unique sequences.
Foss EJ, Lao U, Dalrymple E, Adrianse RL, Loe T, Bedalov A. Foss EJ, et al. Proc Natl Acad Sci U S A. 2017 Jan 17;114(3):552-557. doi: 10.1073/pnas.1614781114. Epub 2017 Jan 3. Proc Natl Acad Sci U S A. 2017. PMID: 28049846 Free PMC article. - Replication Stress, Genomic Instability, and Replication Timing: A Complex Relationship.
Briu LM, Maric C, Cadoret JC. Briu LM, et al. Int J Mol Sci. 2021 Apr 30;22(9):4764. doi: 10.3390/ijms22094764. Int J Mol Sci. 2021. PMID: 33946274 Free PMC article. Review. - Checkpoint Regulation of Nuclear Tos4 Defines S Phase Arrest in Fission Yeast.
Kim SM, Tripathi VP, Shen KF, Forsburg SL. Kim SM, et al. G3 (Bethesda). 2020 Jan 7;10(1):255-266. doi: 10.1534/g3.119.400726. G3 (Bethesda). 2020. PMID: 31719112 Free PMC article.
References
- Howard A, Pelc S R. Heredity. 1953;6:261–273.
- Giulotto E, Mottura A, Carli L D, Nuzzo F. Exp Cell Res. 1978;113:415–420. - PubMed
- Laird C, Jaffe E, Karpen G, Lamb M, Nelson R. Trends Genet. 1987;3:274–281.
- Hansen R S, Canfield T K, Lamb M M, Gartler S M, Laird C D. Cell. 1993;73:1403–1409. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 GM053805/GM/NIGMS NIH HHS/United States
- P30CA1570422/CA/NCI NIH HHS/United States
- R01 HD016659/HD/NICHD NIH HHS/United States
- GM07266/GM/NIGMS NIH HHS/United States
- GM53805/GM/NIGMS NIH HHS/United States
- T32 GM007266/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Miscellaneous