Enhanced T-cell immunogenicity and protective efficacy of a human immunodeficiency virus type 1 vaccine regimen consisting of consecutive priming with DNA and boosting with recombinant fowlpox virus - PubMed (original) (raw)
Enhanced T-cell immunogenicity and protective efficacy of a human immunodeficiency virus type 1 vaccine regimen consisting of consecutive priming with DNA and boosting with recombinant fowlpox virus
S J Kent et al. J Virol. 1998 Dec.
Abstract
The induction of human immunodeficiency virus (HIV)-specific T-cell responses is widely seen as critical to the development of effective immunity to HIV type 1 (HIV-1). Plasmid DNA and recombinant fowlpox virus (rFPV) vaccines are among the most promising safe HIV-1 vaccine candidates. However, the immunity induced by either vaccine alone may be insufficient to provide durable protection against HIV-1 infection. We evaluated a consecutive immunization strategy involving priming with DNA and boosting with rFPV vaccines encoding common HIV-1 antigens. In mice, this approach induced greater HIV-1-specific immunity than either vector alone and protected mice from challenge with a recombinant vaccinia virus expressing HIV-1 antigens. In macaques, a dramatic boosting effect on DNA vaccine-primed HIV-1-specific helper and cytotoxic T-lymphocyte responses, but a decline in HIV-1 antibody titers, was observed following rFPV immunization. The vaccine regimen protected macaques from an intravenous HIV-1 challenge, with the resistance most likely mediated by T-cell responses. These studies suggest a safe strategy for the enhanced generation of T-cell-mediated protective immunity to HIV-1.
Figures
FIG. 1
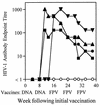
HIV-1 antibodies in DNA-rFPV-vaccinated macaques. Four HIV-1-vaccinated macaques (M2, M3, M4, and M5) (closed symbols) seroconverted to HIV-1 after the second epidermal administration of plasmid DNA pNL4.3dpol as assessed by a particle agglutination assay. HIV-1 antibodies titers declined (>4-fold) thereafter despite three vaccinations with rFPV expressing HIV-1 Env and Gag-Pol antigens. Vaccination times are noted on the x axis. Four control macaques (M7, M8, M9, and M10) (open symbol) received DNA and FPV vaccines not expressing HIV-1 antigens and all had no detectable HIV-1-specific antibodies.
FIG. 2
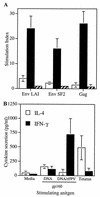
HIV-1-specific Th responses following DNA-rFPV vaccination. (A) rFPV boosting of DNA-primed macaques resulted in a rise in lymphoproliferative responses to both Gag (p24) and homologous (LAI) and heterologous (SF2) subtype B HIV-1 Env antigens. The SIs (means ± standard errors of the means) of PBMC obtained from four macaques receiving HIV-1-expressing vaccines at week 10 following two DNA vaccinations (open bars) and at week 34 following three rFPV boosts (solid bars) are shown in comparison to those for four unvaccinated macaques (hatched bars). (B) PBMC from macaques vaccinated consecutively with HIV-1-expressing DNA and rFPV secrete significant IFN-γ but minimal IL-4 in response to recombinant HIV-1LAI gp160 protein stimulation. The means ± standard errors of the means of triplicate measurements on the four vaccinated macaques (M2 to -5) after two DNA vaccinations alone (week 14) and after three rFPV boosts (week 38) are shown. PBMC from the same animals had an opposite pattern of cytokine secretion in response to stimulation with tetanus toxoid.
FIG. 3
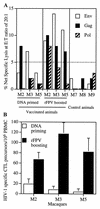
HIV-1-specific CTL responses following DNA-rFPV vaccinations. (A) Net specific lysis of Env-, Gag-, or Pol-expressing autologous BLCL by antigen-stimulated bulk PBMC from three HIV-1-vaccinated macaques and three control macaques was assessed. CTL activities were determined after two DNA vaccinations alone (expressing Env and Gag antigens; week 10 following initial vaccination) or three subsequent rFPV boosting vaccinations (expressing Env, Gag, and Pol antigens; week 34 following initial vaccination). Greater than 5% net specific lysis (dotted line) was considered significant. E:T, effector/target. (B) Quantification of CTL precursors to Env, Gag, and Pol antigens was done by a limiting-dilution assay following DNA and rFPV vaccinations. CTL frequencies were assessed at week 14 (after DNA vaccinations alone) and week 34 (after rFPV boosting). Recognition of control targets expressing VV antigens alone was <5/106 PBMC and has been subtracted. CTL precursor frequencies and 95% confidence intervals were calculated by a maximum-likelihood analysis method.
FIG. 4
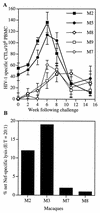
HIV-1-specific CTL responses following HIV-1 challenge. (A) HIV-1 Env, Gag, and Pol CTL precursor frequencies in two HIV-1-vaccinated macaques (closed symbols) and three control macaques (open symbols) were assessed serially over time following HIV-1 challenge by limiting-dilution analysis. Error bars show 95% confidence intervals for CTL precursor estimation. (B) HIV-1 Nef-specific CTL were determined 2 weeks following HIV-1 challenge in bulk PBMC cultures from two vaccinated macaques (M2 and M3) and two control macaques (M8 and M9). E:T, effector/target.
Similar articles
- Evaluation in macaques of HIV-1 DNA vaccines containing primate CpG motifs and fowlpoxvirus vaccines co-expressing IFNgamma or IL-12.
Dale CJ, De Rose R, Wilson KM, Croom HA, Thomson S, Coupar BE, Ramsay A, Purcell DF, Ffrench R, Law M, Emery S, Cooper DA, Ramshaw IA, Boyle DB, Kent SJ; Australian Thai HIV Vaccine Consortium. Dale CJ, et al. Vaccine. 2004 Nov 25;23(2):188-97. doi: 10.1016/j.vaccine.2004.05.024. Vaccine. 2004. PMID: 15531036 - Heterologous prime-boost-boost immunisation of Chinese cynomolgus macaques using DNA and recombinant poxvirus vectors expressing HIV-1 virus-like particles.
Bridge SH, Sharpe SA, Dennis MJ, Dowall SD, Getty B, Anson DS, Skinner MA, Stewart JP, Blanchard TJ. Bridge SH, et al. Virol J. 2011 Sep 7;8:429. doi: 10.1186/1743-422X-8-429. Virol J. 2011. PMID: 21899739 Free PMC article. - Multigene DNA priming-boosting vaccines protect macaques from acute CD4+-T-cell depletion after simian-human immunodeficiency virus SHIV89.6P mucosal challenge.
Doria-Rose NA, Ohlen C, Polacino P, Pierce CC, Hensel MT, Kuller L, Mulvania T, Anderson D, Greenberg PD, Hu SL, Haigwood NL. Doria-Rose NA, et al. J Virol. 2003 Nov;77(21):11563-77. doi: 10.1128/jvi.77.21.11563-11577.2003. J Virol. 2003. PMID: 14557642 Free PMC article. - Prime-boost strategies in DNA vaccines.
Dale CJ, Thomson S, De Rose R, Ranasinghe C, Medveczky CJ, Pamungkas J, Boyle DB, Ramshaw IA, Kent SJ. Dale CJ, et al. Methods Mol Med. 2006;127:171-97. doi: 10.1385/1-59745-168-1:171. Methods Mol Med. 2006. PMID: 16988455 Review. - Immunogenicity and efficacy of DNA/MVA HIV vaccines in rhesus macaque models.
Chea LS, Amara RR. Chea LS, et al. Expert Rev Vaccines. 2017 Oct;16(10):973-985. doi: 10.1080/14760584.2017.1371594. Epub 2017 Sep 4. Expert Rev Vaccines. 2017. PMID: 28838267 Free PMC article. Review.
Cited by
- Improvement of DNA vaccination by adjuvants and sophisticated delivery devices: vaccine-platforms for the battle against infectious diseases.
Grunwald T, Ulbert S. Grunwald T, et al. Clin Exp Vaccine Res. 2015 Jan;4(1):1-10. doi: 10.7774/cevr.2015.4.1.1. Epub 2015 Jan 30. Clin Exp Vaccine Res. 2015. PMID: 25648133 Free PMC article. Review. - Vaccination with attenuated simian immunodeficiency virus by DNA inoculation.
Kent SJ, Dale CJ, Preiss S, Mills J, Campagna D, Purcell DF. Kent SJ, et al. J Virol. 2001 Dec;75(23):11930-4. doi: 10.1128/JVI.75.23.11930-11934.2001. J Virol. 2001. PMID: 11689679 Free PMC article. - Overcoming immunity to a viral vaccine by DNA priming before vector boosting.
Yang ZY, Wyatt LS, Kong WP, Moodie Z, Moss B, Nabel GJ. Yang ZY, et al. J Virol. 2003 Jan;77(1):799-803. doi: 10.1128/jvi.77.1.799-803.2003. J Virol. 2003. PMID: 12477888 Free PMC article. - Heterologous priming-boosting immunization of cattle with Mycobacterium tuberculosis 85A induces antigen-specific T-cell responses.
Taracha EL, Bishop R, Musoke AJ, Hill AV, Gilbert SC. Taracha EL, et al. Infect Immun. 2003 Dec;71(12):6906-14. doi: 10.1128/IAI.71.12.6906-6914.2003. Infect Immun. 2003. PMID: 14638779 Free PMC article. - Priming by DNA immunization augments protective efficacy of Mycobacterium bovis Bacille Calmette-Guerin against tuberculosis.
Feng CG, Palendira U, Demangel C, Spratt JM, Malin AS, Britton WJ. Feng CG, et al. Infect Immun. 2001 Jun;69(6):4174-6. doi: 10.1128/IAI.69.6.4174-4176.2001. Infect Immun. 2001. PMID: 11349095 Free PMC article.
References
- Baba T W, Jeong Y S, Pennick D, Bronson R, Greene M F, Ruprecht R M. Pathogenicity of live, attenuated SIV after mucosal infection of neonatal macaques. Science. 1995;267:1820–1825. - PubMed
- Benveniste R, Agy M, Arthur L, Schmidt A, Corey L, Morton W. Abstracts of the International Conference on AIDS. Vol. 9. 1993. Titration in pig-tailed macaques of the HIV-1 IIIB vaccine challenge stock, abstr. PO-A18-0363; p. 195.
- Boyer J D, Ugen K, Wang B, Agadjanyan M, Gilbert L, Bagarazzi M, Chattergoon M, Frost P, Javadian A, Williams W V, Refaeli Y, Ciccarelli R, McCallus D, Coney L, Weiner D B. Protection of chimpanzees from high-dose heterologous HIV-1 challenge by DNA vaccination. Nat Med. 1997;3:526–532. - PubMed
- Boyer J D, Wang B, Ugen K E, Agadjanyan M, Javadian A, Frost P, Dang K, Carrano R A, Ciccarelli R, Coney L, Williams W V, Weiner D B. In vivo protective anti-HIV immune responses in non-human primates through DNA immunization. J Med Primatol. 1996;25:242–250. - PubMed
- Cohen J. Weakened SIV vaccine still kills. Science. 1997;278:24–25. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources