EAT-4, a homolog of a mammalian sodium-dependent inorganic phosphate cotransporter, is necessary for glutamatergic neurotransmission in caenorhabditis elegans - PubMed (original) (raw)
EAT-4, a homolog of a mammalian sodium-dependent inorganic phosphate cotransporter, is necessary for glutamatergic neurotransmission in caenorhabditis elegans
R Y Lee et al. J Neurosci. 1999.
Abstract
The Caenorhabditis elegans gene eat-4 affects multiple glutamatergic neurotransmission pathways. We find that eat-4 encodes a protein similar in sequence to a mammalian brain-specific sodium-dependent inorganic phosphate cotransporter I (BNPI). Like BNPI in the rat CNS, eat-4 is expressed predominantly in a specific subset of neurons, including several proposed to be glutamatergic. Loss-of-function mutations in eat-4 cause defective glutamatergic chemical transmission but appear to have little effect on other functions of neurons. Our data suggest that phosphate ions imported into glutamatergic neurons through transporters such as EAT-4 and BNPI are required specifically for glutamatergic neurotransmission.
Figures
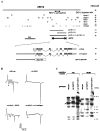
Fig. 1.
eat-4 cloning. This figure summarizes our strategy for and the results from cloning_eat-4_, starting with the cosmid ZK512. A, The abilities of ZK512 and its derivatives to rescue_eat-4_ mutants were tested by germline transformation rescue experiments (right column; for details, see Materials and Methods). A 2.4 kb region of ZK512 in which a restriction-fragment length polymorphism (RFLP) was found in eat-4(ad572) (see_C_) is marked by ↔. Circles are used to mark the sites of restriction enzymes that did not interfere with the ability of ZK512 to rescue, whereas crosses are used to mark the sites of enzymes that did appear to interfere. pRE4-4 and pRE4-8 are two plasmid subclones of ZK512. The deduced extent (∼6.9 kb) and location of the minimal eat-4 rescuing activity are indicated. At the bottom, drawn to an expanded scale, the relationships among the cDNA, the minigene construct, the_eat-4:: reporter_ constructs, and the cosmid are illustrated. The GenBank accession number for eat-4 cDNA is AF095787. B, Examples of M3 neurotransmission phenotypes, assayed by electropharyngeogram (EPG) recordings, observed in wild-type, eat-4 mutant, and transgene-carrying eat-4 mutant animals. Examples of M3 transients [one in each EPG record, except that of the eat-4(ky5) animal] are marked by asterisks. C, A Southern blot analysis of genomic DNA isolated from animals of three different genotypes: wild type, eat-4(ad572), and a spontaneous intragenic revertant eat-4(ad572 ad613), digested with the restriction enzymes_Pst_I or Hin_dIII and probed with labeled ZK512 cosmid. In the set of lanes for each restriction enzyme a band (marked by >), apparent in both the wild-type and the revertant lanes, disappears in the eat-4(ad572) lane where a novel, apparently 1.3 kb larger, band (marked by an_asterisk) is visible. This apparent insertion found in_eat-4_(ad572) was deduced by restriction patterns to be in a 2.4 kb region of ZK512, as indicated by ↔ in_A_.
Fig. 2.
Comparisons between EAT-4 and rat brain-specific sodium inorganic phosphate cotransporter I (BNPI). A, Protein sequence alignment of EAT-4 and BNPI. Identical amino acid residues are highlighted by the gray boxes. The overall identity between these two proteins is 48%. B, Kyte and Doolittle (1982) hydrophobicity profiles of EAT-4 and BNPI. The calculation is based on a window size of 20 residues.
Fig. 3.
Expression of_eat-4:: reporters_. The reporter constructs schematically shown in Figure 1_A_ were used to generate transgenic animals. A, Expression of_eat-4:: lacZ_(nls) in the pharynx, observed with Nomarski optics. β-Galactosidase activity was found in the nuclei of two types (each consists of a bilaterally symmetric pair) of pharyngeal neurons, M3 and_NSM_. Also seen in this panel is the strong signal in the nucleus of an extrapharyngeal cell posterior to M3. This extrapharyngeal cell is probably a neuron in the IL1 class.B, C, Expression of_eat-4:: gfp_ in extrapharyngeal neurons. The identity of these cells is marked either in the figure (B) or above the photograph (C). The identity of one cell (marked as_AVJ_ or AIN) is uncertain. In_B_, the entire animal is shown, whereas in_C_ only the head is shown. Only the right half of the animal is shown in C, because these classes of cells are bilaterally symmetric. C, Shown is an overlay (with Adobe Photoshop software) of nine serial confocal images with an intersection spacing of 0.9 μm.
Fig. 4.
eat-4 function in the anterior touch cells. A, Schematic circuit diagram of the anterior touch response. The diagram is modified from Chalfie et al. (1985). The arrows represent chemical synapses, and the bars represent gap junctions. Dashed lines represent the chemical synapses affected by_eat-4_, as suggested by the data in B.B, Quantitation of the anterior touch response. For each gentle touch to the anterior body the animal’s response was scored as sensitive if backing occurred immediately, partially sensitive if backing occurred with a delay, and insensitive if backing did not occur within several seconds. The response of each animal was tested at least seven times, and the bars represent the average percentage of trials in which the animals responded as sensitive (black portion of the bar) or partially sensitive (gray portion of the_bar_). Error bars represent the SEM for the percentages of responses that were either sensitive or partially sensitive. Cases in which the behavior of the operated animals was significantly different from that of the mock-operated animals of the same genotype are indicated by an asterisk(p < 0.05); n, number of animals tested.
Similar articles
- The localization of the brain-specific inorganic phosphate transporter suggests a specific presynaptic role in glutamatergic transmission.
Bellocchio EE, Hu H, Pohorille A, Chan J, Pickel VM, Edwards RH. Bellocchio EE, et al. J Neurosci. 1998 Nov 1;18(21):8648-59. doi: 10.1523/JNEUROSCI.18-21-08648.1998. J Neurosci. 1998. PMID: 9786972 Free PMC article. - Identification of a vesicular glutamate transporter that defines a glutamatergic phenotype in neurons.
Takamori S, Rhee JS, Rosenmund C, Jahn R. Takamori S, et al. Nature. 2000 Sep 14;407(6801):189-94. doi: 10.1038/35025070. Nature. 2000. PMID: 11001057 - Cloning and expression of a cDNA encoding a brain-specific Na(+)-dependent inorganic phosphate cotransporter.
Ni B, Rosteck PR Jr, Nadi NS, Paul SM. Ni B, et al. Proc Natl Acad Sci U S A. 1994 Jun 7;91(12):5607-11. doi: 10.1073/pnas.91.12.5607. Proc Natl Acad Sci U S A. 1994. PMID: 8202535 Free PMC article. - [The inorganic phosphate transport system in the brain and the potency as vesicular glutamate transporters].
Hisano S. Hisano S. Kaibogaku Zasshi. 2002 Sep;77(3):51-8. Kaibogaku Zasshi. 2002. PMID: 12382430 Review. Japanese. - Evolution of the Na-P(i) cotransport systems.
Werner A, Kinne RK. Werner A, et al. Am J Physiol Regul Integr Comp Physiol. 2001 Feb;280(2):R301-12. doi: 10.1152/ajpregu.2001.280.2.R301. Am J Physiol Regul Integr Comp Physiol. 2001. PMID: 11208556 Review.
Cited by
- A neurotransmitter atlas of C. elegans males and hermaphrodites.
Wang C, Vidal B, Sural S, Loer C, Aguilar GR, Merritt DM, Toker IA, Vogt MC, Cros CC, Hobert O. Wang C, et al. Elife. 2024 Oct 18;13:RP95402. doi: 10.7554/eLife.95402. Elife. 2024. PMID: 39422452 Free PMC article. - A neurotransmitter atlas of C. elegans males and hermaphrodites.
Wang C, Vidal B, Sural S, Loer C, Aguilar GR, Merritt DM, Toker IA, Vogt MC, Cros C, Hobert O. Wang C, et al. bioRxiv [Preprint]. 2024 Jun 7:2023.12.24.573258. doi: 10.1101/2023.12.24.573258. bioRxiv. 2024. PMID: 38895397 Free PMC article. Updated. Preprint. - Distinct mechanisms of non-autonomous UPRER mediated by GABAergic, glutamatergic, and octopaminergic neurons.
Coakley AJ, Hruby A, Wang J, Bong A, Nair T, Ramos CM, Alcala A, Averbukh M, Dutta N, Moaddeli D, Hicks D, de Los Rios Rogers M, Sahay A, Curran SP, Mullen PJ, Benayoun BA, Garcia G, Higuchi-Sanabria R. Coakley AJ, et al. bioRxiv [Preprint]. 2024 May 30:2024.05.27.595950. doi: 10.1101/2024.05.27.595950. bioRxiv. 2024. PMID: 38854121 Free PMC article. Preprint. - A hyperpolarizing neuron recruits undocked innexin hemichannels to transmit neural information in Caenorhabditis elegans.
Nakayama A, Watanabe M, Yamashiro R, Kuroyanagi H, Matsuyama HJ, Oshima A, Mori I, Nakano S. Nakayama A, et al. Proc Natl Acad Sci U S A. 2024 May 21;121(21):e2406565121. doi: 10.1073/pnas.2406565121. Epub 2024 May 16. Proc Natl Acad Sci U S A. 2024. PMID: 38753507 - Neuronal basis and diverse mechanisms of pathogen avoidance in Caenorhabditis elegans.
Lei M, Tan Y, Tu H, Tan W. Lei M, et al. Front Immunol. 2024 May 1;15:1353747. doi: 10.3389/fimmu.2024.1353747. eCollection 2024. Front Immunol. 2024. PMID: 38751431 Free PMC article. Review.
References
- Albertson DG, Thomson JN. The pharynx of Caenorhabditis elegans. Philos Trans R Soc Lond [Biol] 1976;275:299–325. - PubMed
- Avery L. Motor-neuron M3 controls pharyngeal muscle relaxation timing in Caenorhabditis elegans. J Exp Biol. 1993b;175:283–297. - PubMed
- Avery L, Horvitz HR. Pharyngeal pumping continues after laser killing of the pharyngeal nervous system of C. elegans. Neuron. 1989;3:473–485. - PubMed
- Avery L, Horvitz HR. Effects of starvation and neuroactive drugs on feeding in Caenorhabditis elegans. J Exp Zool. 1990;253:263–270. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 GM024663/GM/NIGMS NIH HHS/United States
- HL46154/HL/NHLBI NIH HHS/United States
- R01 HL046154/HL/NHLBI NIH HHS/United States
- R37 GM024663/GM/NIGMS NIH HHS/United States
- R37 HL046154/HL/NHLBI NIH HHS/United States
- GM24663/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Molecular Biology Databases