Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety (original) (raw)
Abstract
Aging of skin is an intricate biological process consisting of two types. While intrinsic or chronological aging is an inevitable process, photoaging involves the premature aging of skin occurring due to cumulative exposure to ultraviolet radiation. Chronological and photoaging both have clinically differentiable manifestations. Various natural and synthetic retinoids have been explored for the treatment of aging and many of them have shown histological and clinical improvement, but most of the studies have been carried out in patients presenting with photoaged skin. Amongst the retinoids, tretinoin possibly is the most potent and certainly the most widely investigated retinoid for photoaging therapy. Although retinoids show promise in the treatment of skin aging, irritant reactions such as burning, scaling or dermatitis associated with retinoid therapy limit their acceptance by patients. This problem is more prominent with tretinoin and tazarotene whereas other retinoids mainly represented by retinaldehyde and retinol are considerably less irritating. In order to minimize these side effects, various novel drug delivery systems have been developed. In particular, nanoparticles have shown a good potential in improving the stability, tolerability and efficacy of retinoids like tretinoin and retinol. However, more elaborate clinical studies are required to confirm their advantage in the delivery of topical retinoids.
Keywords: photoaging, chronological aging, tretinoin, retinaldehyde, tazarotene, nanoparticles
Introduction
Skin – the largest organ of the body – protects all the other organs from the external environment. The skin is a complex organ with multiple structures and cell types and divided into three layers: epidermis, dermis, and the subcutaneous tissue. The epidermis is mainly composed of keratinocytes, pigment-producing melanocytes, and antigen-presenting Langerhans cells. A basement membrane separates the epidermis from the dermis, which primarily contains extracellular proteins produced by the fibroblasts below. The vascular supply to the skin resides in the dermis. The subcutaneous tissue consists of fat cells that underline the connective tissue network. Type I collagen is the most abundant protein in the skin connective tissue. The other extracellular matrix proteins, which are a part of the skin connective tissue, are collagens (III, V, and VII), elastin, proteoglycans, fibronectin, etc. The newly synthesized type I procollagen is secreted into the dermal extracellular space where it undergoes enzymatic processing to arrange itself into a triple helix configuration (Rittié and Fisher 2002).
Apart from environmental protection against radiation, functions of the skin include heat regulation, immune response, biochemical synthesis, sensory detection, regulation of absorption/loss of water and electrolytes. The stratum corneum formed from nonviable corneocytes plays the major role. Keratin is aligned in the intercrossed disulfidic macrofibres along with filaggrin, the main protein component of the keratolytic granule. The cells develop a cornified involucre resulting from the intercrossing of involucrin and keratohyalin. Lamellar lipids accumulate in the intercellular spaces, which are strongly hydrophobic. The combination of the cornified hydrophilic cells with the hydrophobic intercellular material forms a barrier for the external hydrophilic and hydrophobic substances.
With age the skin’s natural rejuvenation process slows drastically and the skin becomes thinner, drier, and less elastic (Ramos-E-Silva et al 2001).
The aging process
Aging represents a biologic attrition at the cellular level resulting in decreased reserve capacity and ability to perform normal functions occurs throughout an organisms’ life span increasing the likelihood of death. Aging is thus the result of a genetic program or a clock that is implanted in the genetic make-up of each species. One must also remember that cumulative damage to the genes and proteins derived thereof, result in compromised function and homeostatic failure. This leads the organism towards premature aging and death, which in turn shall depend on its repair systems.
The somatic cells have telomeres at the terminal portion of the eukaryotic chromosomes which consist of many hundreds of tandem short sequence repeats (TTAGGG) predetermining the number of times the cell can divide before it senesces. The enzyme DNA polymerase that replicates cellular chromosomes during mitosis cannot replicate the final base pairs of each chromosome, resulting in progressive telomere shortening with each cellular division. A critically short telomere will compromise gene transcription and signal cellular senescence which is otherwise better known as “apoptosis” (Yaar and Gilchrest 2001). Human keratinocytes approach replicative senescence after 50–100 population doublings in culture and remain permanently arrested in the G1 phase of the cell cycle. The telomere is just one of the three molecules which were found to be crucial for replicative senescence. In addition, keratinocytes have an increased resistance to apoptosis, thus giving a time window for DNA and protein damage to accumulate (Rheinwald et al 2002).
The skin, being the ultimate protective barrier between the internal organs and the environment, is exposed to ultraviolet (UV) irradiation and to a lesser extent to other DNA damaging agents such as cigarette smoke, automobile exhaust, and professional exposure. UV irradiation causes formation of pyrimidine dimers and the benzo[a]pyrene from cigarette smoke causes formation of guanine base pair adducts. All this moves hand-in-hand with damage from endogenous agents such as reactive oxygen and nitrogen species (ROS/RNS) generated by all aerobic cell species as part of their routine metabolic processes (Yaar and Gilchrest 2001).
Amino acid racemization and interaction of amino acid groups with reducing sugars (Maillard reaction) result in an altered or total loss of protein functions which does the dermal collagen proteins (Yaar and Gilchrest 2001).
Skin aging
Skin aging is influenced by several factors including genetics, environmental exposure (UV radiation, xenobiotics, and mechanical stress), hormonal changes and metabolic processes (generation of reactive chemical compounds such as activated oxygen species, sugars and aldehydes). All factors together act on the alterations of skin structure, function, and appearance. Yet solar UV radiation unquestionably is the single major factor responsible for skin aging (Rittié and Fisher 2002).
Intrinsic/chronological aging
Intrinisic/chronological aging is defined by the clinical, histological, and physiological decrements that occur in the sun-protected skin, affecting the rate of epidermal turnover, clearance of chemical substances from the dermis, dermal thickness and cellularity, thermoregulation, rate of re-epithelialization after wounding, mechanical protection, immune responsiveness, sensory perception, sweat and sebum production, capacity for vitamin D synthesis and vascular reactivity. Clinically, the intrinsically aged skin is atrophic, which may result in prominence of vasculature and loss of elasticity. The stratum corneum remains relatively unchanged but the epidermis thins with a flattening of the dermo-epidermal junction expressing an increased fragility of the skin. There is considerable decrease in dermal thickness and vascularity as well as a reduction in the number and biosynthetic capacity of the fibroblast resulting in delayed wound healing. With an increasing age, there is a progressive decline in the response of keratinocytes and fibroblasts to growth factors, decreasing the proliferative capacity (Gilchrest 1996). A decreased immune responsiveness is seen with aging since there is a decrease in the number and abnormal morphology seen in the antigen-presenting cells. Another important function that decreases with aging is the formation of vitamin D3 due to decreased formation of 7-dehydrocholesterol in the reduced epidermal cells (Yaar and Gilchrest 2001).
Photoaging
Photoaging is the superimposition of photodamage on intrinsically aged skin generally bringing about premature aging. This specific damage occurs by chronic (multiple) exposure of the skin to UV light. Clinically, the skin becomes coarse; epidermis thickens (hyperplasia) initially and then thins (atrophy), there is laxity, sallowness with wrinkles, irregular hyperpigmentation, lentigines, and telangiectasias (Gilchrest 1996). The pores of the skin are larger, filled with horny material and have a tendency to develop Favre-Racouchot’s syndrome (nodular elastoidosis with cysts and comedones). There is also an increase in development of benign neoplasms (seborrheic keratosis, fibroma, acrochordon, and ruby spots), “premalignant” lesions (actinic keratosis, lentigo maligna), and malignant lesions (basal and squamous cell carcinomas and malignant melanomas) on chronically exposed skin found in the face, hands and neck regions (Torras 1996, Oppel and Korting 2004). In severely damaged skin, there is loss of epidermal polarity (orderly maturation) and individual keratinocytes may show atypia, especially the lower epidermal layers. More profound changes occur in the dermis, where photodamage is characterized by degeneration of collagen and deposition of abnormal elastotic material, reflected by wrinkles, furrows, and yellow discoloration of the skin. The greater the photodamage, the more the accumulation of thickened, tangled and degraded elastic fibers (Gilchrest 1996). The surface roughness is not only attributed to the changes in the stratum corneum but also to the changes in the glycosoaminoglycan (GAG) content of the skin. With increase in age, there is a decrease in the GAG content. Contradictorily, Bernstein and Uitto (1995) found that there is an increase in the GAG content in the photoaged skin. Yet GAG does not deposit in the papillary dermis, instead it accumulates on the abnormal elastotic material, which makes it unavailable as a source of hydration resulting in a dull, leathery appearance of the skin (Kang, Fisher, et al 2001). The microcirculation is also affected by sun exposure. Blood vessels become dilated and twisted (telangiectasia) and finally very sparse, while their walls are initially thickened and later thinned (Gilchrest 1996). UV irradiation of the skin increases the reactive oxygen species and decreases the endogenous antioxidant enzymes. The superoxide anion is produced by energy transfer from several endogenous UV-absorbing chromophores including NADH−/NADPH, tryptophan, riboflavin, or transurocanic acid (Rittié and Fisher 2002) in the presence of molecular water present within the cell. The superoxide anion is then converted to hydrogen peroxide, which in the presence of transition metal ions such as iron and copper undergoes conversion to a highly reactive hydroxyl radical. This increased production of ROS alters gene and protein structure and function leading to skin damage.
Table 1 gives an overview of the various epidermal, dermal, and clinical signs with which one can differentiate between chronological aging and photoaging.
Table 1.
Comparison of chronological aging and photoaging
| Ageing types | Identification characteristics | ||
|---|---|---|---|
| Epidermis | Dermis | Clinical | |
| Chronological aging | Thinner than normal with lower cell growth, minor abnormalities in keratinocyte regularityNormal stratum corneumThere is loss of rete pegs here as well | Elastin fibers appear irregular in their arrangement, whereas collagen fibers begin to lower in number and thickness | Skin is smooth, unblemished, but shows saggy appearance |
| Photoaging | Thick skin, with acanthosis followed by atrophy of the cellsHigh basal keratinocyte irregularity Stratum corneum appears compactThere is loss of rete pegs here as well | Excessive production of elastin fibers in an improper orientation, collagen fibres appear to thicken and then wear out soonAppearance of grenzzone | Smooth, leathery, reddened appearance with initially light wrinkles, which later deepen, thus showing loss of collagen fibers |
Mechanism of collagen degradation
Mature collagen in skin undergoes continuous turnover, which is required for optimal connective tissue function. The unique molecular structure of collagen renders it largely resistant to nonspecific proteolytic attack. The matrix metalloproteinases (MMPs) are a group of enzymes responsible for degradation of collagen. The MMPs are members of a large subfamily of proteinases with certain common structural features. The human family of MMPs is composed of at least 16 members who can be classified into 4 different subfamilies: 1) collagenases, 2) gelatinases, 3) stromelysins, and 4) membrane MMPs. The first three can cleave native, undenatured interstitial helical collagens found in the skin within the triple-helical domain. The cleavage site is specific in type I collagen generating three-quarter and one-quarter length fragments. Following this initial unequal split by collagenase, the resultant denatured collagen called gelatin is further degraded by gelatinases and stromelysins (Kang, Fisher, et al 2001).
Biochemical pathways that are triggered after UV irradiation activating cell surface cytokine and growth factor receptors
Human skin cells respond to UV radiation by activation of multiple cytokine and growth factor receptors. These include epidermal growth factors receptors (EGF-R), tumor necrosis factor (TNF)-α receptors, platelet activating factor (PAF) receptor, interleukin (IL)-1 receptor, insulin receptor and platelet derived growth factor. Amongst these, the EGF-R activation has been the most studied. It is a single chain 180 kDa transmembrane protein. The extracellular domain possesses high affinity binding for EGF and EGF-like ligands (transforming growth factor [TGF]-α, amphiregulin and heparin binding-EGF) (Rittié and Fisher 2002). The intracellular domain possesses intrinsic tyrosine kinase activity. EGF-R also known as ErbB1 undergoes homo- or heterodimerization with either ErbB2 or ErbB3 resulting in the transphosphorylation of specific tyrosine residues. EGF-R tyrosine phosphorylation is a well-characterized marker for receptor activation and occurs within 10 minutes of UV irradiation. Notably, UV fails to induce EGF-R tyrosinase phosphorylation in cells expressing mutant EGF-R lacking tyrosine kinase activity. UV irradiation of EGF-R, like ligand activation, is dependent on EGF-R tyrosine kinase-catalysed trans-phosphorylation. Alternatively, it has been proposed that UV-induced EGF-R tyrosine phosphorylation results from inactivation of protein tyrosine phosphatases (PTPs) that function to maintain EGF-R in a dephosphorylated basal state. Inhibition by specific tyrosine kinase inhibitors results in a very rapid dephosphorylation of EGF-R. Treatment of the cells with UV irradiation substantially prolonged the life of the EGF-R phosphorylated tyrosinases, thus suggesting an inhibitory effect of UV on PTPs. This inhibitory activity by UV was sensitive to N-acetyl cysteine, a scavenger of reactive oxygen intermediates and could be mimicked by treating cells with H2O2. UV-induced inactivation of PTP activity is postulated to result from oxidation of a critical cysteine residue that is present in the catalytic active site of all PTP’s to sulfenic acid. This oxidation occurs by the exposure of the cysteine residue on the PTP to reactive oxygen species which are generated within the cells by UV irradiation (Rittié and Fisher 2002). This inactivation of PTPs may result in the activation of other cell surface receptors and cytokine receptors which in turn leads to activation of small GTP-binding protein families such as the Rac, Ras, and Cdc42. These are either direct or indirect (via other GTP-binding proteins or ROS) upstream regulators of mitogen-activated protein kinases (MAPKs). The UV irradiation causes increased ROS production and simultaneous increase in ceramide levels which may also contribute to the activation of MAPK pathways. A major effector of the MAPK pathways is the transcription factor activator protein-1 (AP-1). AP-1 is constitutively composed of c-Fos and JunD proteins or the other Jun and Fos family proteins (c-Jun, Junb, FosB, Fra1, and Fra2) in the nonirradiated skin. The activation of MAPKs indirectly activates the transcription factors for AP-1 formation ie, transcription of the c-Fos and c-Jun genes. UV irradiation induces c-Jun mRNA and protein in human skin in vivo within 30 min and 1 hour, respectively, and protein levels remain elevated for at least 24 hours post UV irradiation. Increased levels of c-Jun compete with JunD for forming complexes with c-Fos resulting in c-Jun: c-Fos AP-1 complexes (Rittié and Fisher 2002). Transcription of several MMP family members is regulated by this AP-1 complex formed throughout the epidermal and dermal cells. MMPs are a large family of zinc-requiring endoprotreases with a broad range of specificities that together have the capacity to degrade all the extracellular matrix proteins. Initially, MMPs are synthesized as zymogens (proenzymes) which undergo proteolytic degradation to be active. These are inhibited by tissue inhibitors of metalloproteinases (TIMPs). Several MMPs are upregulated by AP-1 including MMP-1 (interstitial collagenase or collagenase1), which intitiates the degradation of types I & III fibrillar collagens, MMP-9 (92 kDa gelatinase or gelatinase B) degrades the collagen fragments (gelatin) generated by collagenases and MMP-3 (stromelysin 1) further degrades collagen type IV of the basement membrane and activates proMMP-1. MMP-1, MMP-3, and MMP-9 transcripts are induced within 8 hours following UV irradiation. Thus, together MMPs have the capacity to completely degrade mature fibrillar collagen in the skin within 24 hours of UV exposure via the induction of transcription factor AP-1. In addition to causing collagen breakdown, UV radiation impairs new type I collagen synthesis and organization of collagen fibrils in skin in vivo. Down-regulation of type I collagen is mediated by down-regulation of the transcription of genes that encode for type I procollagen. Type I procollagen mRNA and protein expression levels are decreased within 8 hours following UV irradiation of the human skin in vivo and become essentially absent in the upper dermis within 24 hours after UV irradiation, consistent with the sustained induction of c-Jun and thus AP-1 activation.
The TGF-β is a major profibrotic cytokine, which regulates multiple cellular functions including differentiation, proliferation and induction of synthesis of major extracellular matrix (ECM) proteins – collagen and elastin (Massague 1998). In human skin, TGF-β inhibits growth of epidermal keratinocytes and stimulates growth of dermal fibroblasts (Massague 2000). TGF-β inhibits the expression of MMP-1 and MMP-3 by binding to a certain cell surface receptor complex (TGF-β receptor proteins: TβR I/II/III), thus preventing the breakdown of collagens. UV irradiation has been shown to impair the TGF-β signaling pathway by reducing TβRII expression and to a lesser extent the inhibitory Smad 7. Moreover, the connective tissue growth factor is down-regulated after UV irradiation (Rittié and Fisher 2002).
In this article, we critically compare the clinical efficacy and safety of various retinoids that have been in the treatment and protection of skin aging.
Retinoids
The importance of retinol (vitamin A) was discovered during World War I and subsequent research showed that its deficiency gives rise to xerosis and follicular hyperkeratosis. The retinoid drug project was launched in 1968 to synthesize compounds similar to vitamin A by chemical manipulation of its molecule to improve clinical efficacy and safety. The use of these substances in therapy dates back some 3000 years to ancient Egypt, where liver was used to treat endemic night blindness. The modern history of retinoids, however, began in 1909 when an essential factor in the viability of an embryo in the fatty extract of the egg yolk, called vitamin A, was discovered. Retinoids finally were introduced into the treatment of dermatoses including photoaging more than two decades ago (Ramos-E-Silva et al 2001).
The retinoid family comprises vitamin A (retinol) and its natural derivatives such as retinaldehyde, retinoic acid, and retinyl esters, as well as a large number of synthetic derivatives (Antille et al 2004). Retinol is a 20-carbon molecule that consists of a cyclohexenyl ring, a side chain with four double bonds (all in trans configuration), and an alcohol end group, hence the name all-trans-retinol. The oxidation of the alcohol end group in retinol results in the formation of an aldehyde (all-trans retinaldehyde or retinal), which can be further oxidized to a carboxylic acid (all-trans retinoic acid or tretinoin). Vitamin A cannot be synthesized by the body; hence it needs to be supplied to the body. Naturally, it is present as retinyl esters and beta-carotene. The retinyl esters are converted to retinol before absorption from the intestine and back to retinyl esters for storage in the liver. In the plasma, retinol is bound to plasma-retinol binding proteins. Retinol is be metabolized to four important products: retinyl esters, all-trans retinoic acid, 14-hydroxy-4, 14-retro retinol, and all-trans 3, 4-didehydroretinol, and its esters. Retinoids are required for a vast number of biological processes. In particular, they are involved in embryogenesis, reproduction, vision, growth, inflammation, differentiation, proliferation, and apoptosis. Retinal is an essential part of the rhodopsin pigment, necessary for vision (Roos et al 1998). Retinoids are found in the keratinocytes in two forms: retinol and retinyl esters – probably the storage form. This esterification is catalysed by two enzymes, acyl CoA: retinol acyltransferase and lecithin: retinol acyltransferase (Törmä and Vahlquist 1990). The metabolism of retinyl esters to retinol is catalysed by retinyl ester hydrolase (Törmä and Vahlquist 1990).
Retinoid classification
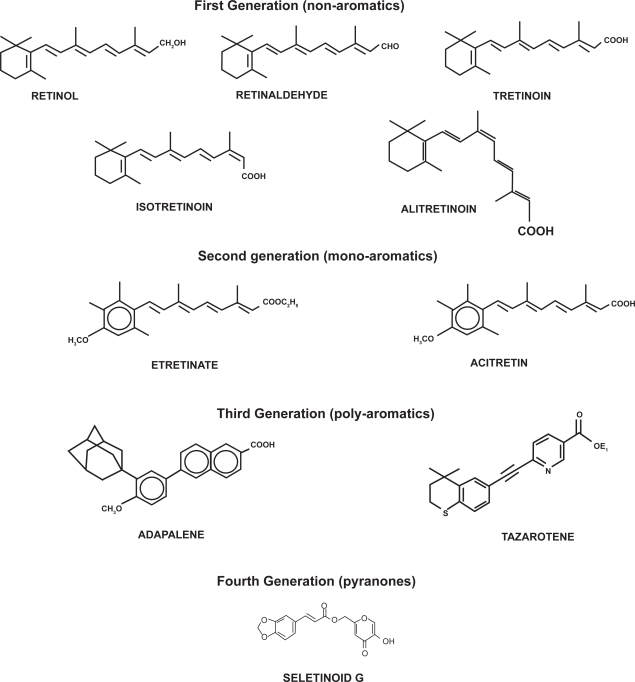
Based on the structural features and reflecting the time of introduction, retinoids can be classified into various generations. The chemical structures of various retinoids are shown in Figure 1.
Figure 1.
Chemical structures of retinoids.
Mechanism of action of topical retinoids
Retinoids are very well known to influence a variety of cellular processes, such as cellular growth and differentiation, cell surface alterations, and immune modulation. Many of their tissue effects are mediated by their interaction with specific cellular and nucleic acid receptors. The cellular or cytoplasmic receptors include the Cellular Retinoic Acid Binding Protein (CRABP) types I and II and the cellular retinol binding protein (Astrom et al 1991). The nucleic acid receptors were discovered in 1987 to reveal the mechanism of action by which tretinoin and several of its analogues would bring about their biological effects. This discovery of the existence of a tretinoin specific gene transcription factor lead to the realization that tretinoin is a hormone. These nuclear receptors are related to a super family of nuclear DNA transcription factors, which include steroid, thyroid hormone, and vitamin D receptors. They comprise two families, each of which are encoded by three genes. The nuclear retinoic acid receptor family called RARs was the first to be described and consists of three forms (RAR-α, RAR-β, RAR-γ) that are activated by RAR specific all-_trans_-retinoic acid (tretinoin). As such the RARs have distinct DNA and retinoid-binding domains and they function in pairs, either pairs of identical receptors called homodimers or pairs of different receptors called heterodimers. In the human skin, RARs partner with retinoid X receptors (RXRs) to form heterodimers (Giguére et al 1987; Petkovich et al 1987; Brand et al 1988; Fisher et al 1994; Xiao et al 1995). The retinoid X receptors or RXRs are the second family of nuclear receptors which interact with 9-cis retinoic acid. Both RARs and RXRs are present in the normal skin providing the necessary machinery for the retinoid repair process of the photodamaged skin. The RAR-γ subtype accounts for nearly 90% of RARs in the human epidermis, whereas the RXR-α subtype accounts for nearly 90% of the RXRs. Therefore, for the most part, the normal human skin is regulated by paired heterodimers composed of RAR-γ and RXR-α. The heterodimer complex binds to specific elements in the DNA known as retinoic acid response elements (RARE) in the promoter region of the genes that are regulated by that specific retinoid thus regulating the transcriptional activity of that retinoid-responsive gene. The heterodimer requires only RAR specific retinoid (tretinoin) to bind to RARE and initiate transcriptional activity; the presence of a RXR binding retinoid (9-cis retinoic acid) does not confer additional trans-activation induced by the RAR retinoid. However, for the heterodimer to function, the RXR protein must be physically present to associate with the RAR protein. This is probably the way topical retinoids improve photoaging by modifying cellular differentiation programs: 1) initiating the increase of epidermal proliferation leading to epidermal thickening; 2) compaction of the stratum corneum; and 3) biosynthesis and deposition of the glycosoaminoglycans (Griffiths et al 1993).
New retinoids are selective for different RAR’s such as the third generation retinoid Adapalene for RAR-β. The newest retinoids are antagonists, which have potent anti-inflammatory activity and look promising as topical treatment for psoriasis (Griffiths et al 1998).
Tretinoin
Tretinoin happens to be the retinoid that is investigated more than any other retinoid implicated in the treatment of intrinsic or photoaging. Although tretinoin has been used in dermatology since the 1960s, its potential in the treatment of aging was realized no earlier than in the 1980s. The efficacy of tretinoin in the treatment of photoaging was first demonstrated by Kligman and colleagues (1984) using an animal model of photoaging. The authors observed that treatment of photoaged mouse skin with tretinoin for 10 weeks resulted in a significant repair zone of new collagen in the papillary dermis, which also correlated with wrinkle effacement. This interesting observation prompted researchers to investigate the potential of tretinoin in the treatment of photoaging. Much later ex-vivo investigations carried out by Fisher and colleagues (1996) helped in understanding the molecular basis of this observation. Fisher and colleagues (1996) found that pretreatment of UV irradiated excised (photoaged) skin with 0.1% tretinoin cream results in complete blockade of interstitial collagenase and gelatinases synthesis thus preventing collagen degradation. Moreover, application of 0.1% tretinoin also blocked UV-induced activation of the nuclear transcription factors AP-1 and NF-κB.
Following the ex-vivo observations, Kligman and colleagues (1986) conducted a vehicle-controlled open study to evaluate the clinical efficacy of 0.05% tretinoin. The study involved application of 0.05% tretinoin on the photoaged facial and forearm skin for the duration of 3–12 months. Interestingly, tretinoin resulted in clinical improvement of the photoaged skin. Moreover, histological examination showed deposition of reticulin fibers and new dermal collagen formation (type I and III) accompanied by angiogenesis in the papillary dermis. Encouraging results obtained from this study stimulated researchers to conduct a vast number of clinical trials to confirm the clinical efficacy of tretinoin in the treatment of photoaging.
Considering the exorbitant number of the reports available in the literature, we have divided this part in several subsections.
Short-term studies on tretinoin
This section should deal with the short-term studies that were carried out immediately after the reports by Kligman and colleagues (1986). Table 2 provides an overview of those studies. In the two double-blind studies a statistically significant clinical improvement of various parameters was observed. Furthermore, the tretinoin treated group had a “rosy glow” not seen in the control group. Moreover, it was observed that the skin condition continued to improve when the follow-up assessment was performed after cessation of treatment. Hence, studies involving longer duration of tretinoin treatment were designed.
Table 2.
Overview of short-term studies on tretinoin
| Reference | Study design | No. of patients | Duration | Observations in tretinoin group |
|---|---|---|---|---|
| Weiss et al (1988) | Randomized, Double-blind 0.1% tretinoin cream vs vehicle | 30 | 4 months | Compaction of stratum corneumIncrease in glycosamine glycans (GAGs)Improvement in fine wrinkles, coarse wrinkles, tactile roughness, sallownessa |
| Lever et al (1990) | Double-blind0.05% tretinoin cream vs placebo control | 20 | 3 months | Epidermal thickeningImprovement in fine wrinklesa |
| Shukuwa et al (1993) | Open-label0.05% tretinoin cream | 5 | 1 month | Compaction of stratum corneum, Disappearance of atypia, dysplasiaNo significant dermal changes |
Long-term studies on tretinoin
Long-term studies on tretinoin were carried out as short-term studies showed that the skin condition continued to improve in appearance over time. Additionally, another objective was to assess the long-term benefit-to-risk ratio of the tretinoin formulations. For suitability of understanding we have divided long-term studies into 6-months studies and studies involving more than 6 months.
Studies involving 6-month tretinoin treatment
Most of the 6-month studies that were carried out used tretinoin emollient cream that is specifically designed for the treatment of photoaging. Additionally, most of these studies compared the efficacy of the various strengths of tretinoin to arrive at the concentration that is optimum for the treatment of photoaging. The various 6-month studies that were carried out are reported in Table 3. All the 6-month studies did show significant improvement in the clinical signs of photoaging, but again the improvement in skin condition continued even after 6 months.
Table 3.
Overview of studies involving 6 months’ tretinoin treatment
| Reference | Study design | Duration | No. of patients | Observations and Inferences |
|---|---|---|---|---|
| Leyden et al (1989) | Randomized, double-blind0.05% tretinoin cream vs vehicle control | 6 months | 30 | Improvement in fine wrinkling, coarse wrinkling, sallowness and hyperpigmentation |
| Caputo et al (1990) | Dose escalating study tretinoin cream 0.01% in the 1st month, 0.025% in the 2nd month, 0.05% for next 4 months | 6 months | 89 | Improvement in fine and coarse wrinkling, mottled hyperpigmentation, skin texture and laxity |
| Weinstein et al (1991) | Double-blind tretinoin emollient cream 0.05% and 0.01% vs vehicle | 6 months | 251 | Significant improvement in fine wrinkling, mottled hyperpigmentation, roughness, laxity, epidermal thickness, in group treated with 0.05% tretinoin as compared with 0.01% and vehicle group Dose-dependant responses were observed No effect was seen in dermal thickness, collagen regeneration, reversal of keratinocytic atypia |
| Bhawan et al (1991) | Randomized, Double-blind tretinoin emollient cream 0.001%, 0.01% and 0.05% vs vehicle | 6 months | 533 | Significant improvement in fine wrinkling, mottled hyperpigmentation, roughness, epidermal thickness, in group treated with 0.05% tretinoin as compared with 0.01%, 0.001% and vehicle group Dose-dependant responses were observed Vehicle-treated group showed some improvement |
| Olsen et al (1992) | Same as in case of Bhawan et al (1991) | 6 months | 296 | Same as in case of Bhawan et al (1991) |
Studies involving tretinoin treatment for more than 6 months
The ability of long-term (more than 6 months) tretinoin treatment to maintain improvement in photoaging was first evaluated by Ellis and colleagues (1990) in a 22-month study carried out in 16 patients with photoaged skin. All the subjects used 0.1% tretinoin for the first 4 months. Thereafter, 3 patients continued this regimen, 8 were changed to alternate day treatment for the last 12 months, and the remaining used 0.05% tretinoin for 5 months and then reduced to alternate day application till the end of therapy. It was observed that the improvement of wrinkling continued up to the 10th month and was maintained thereafter. The stratum corneum and epidermal thickness returned to the normal during the course of treatment. In another trial, Green and colleagues (1993) studied the effect of 0.05% tretinoin emollient cream applied daily for 12 months. Tretinoin treatment showed significant improvement in the clinical signs of photoaging. However, the major degree of changes occurred after 6 months and later on they tended to remain stable as observed in the earlier study. Extension of the study for 6 more months with either weekly or thrice weekly application showed further improvement in overall signs of photoaging.
Thereafter, Bhawan and colleagues (1995) evaluated the changes occurring at the dermal level in Caucasian skin after daily application of 0.05% tretinoin cream for a period of 12 months. Interestingly, no significant changes were observed at 6 months in the papillary dermis in the tretinoin-treated group, which supported the observation made in the initial short-term studies. However, after 12 months, formation of new collagen fibers as well as reduction in nodularly degenerated microfibrillar material was observed in the tretinoin-treated group. This study indicated that for appreciable dermal level improvement, more than 6 months of tretinoin therapy is required. This also provided an explanation why remarkable changes were observed only after 6 months of tretinoin treatment in the study carried out by Green and colleagues (1993). Olsen and colleagues (1997a) evaluated the histological and clinical changes occurring in 298 patients after once daily application of either 0.05% or 0.01% tretinoin emollient cream for a duration of 1 year. Significant improvement in histological and clinical markers was observed in both the 0.05% and the 0.01% tretinoin group as compared with vehicle. In another study, Oslen and colleagues (1997b) evaluated the 6 month effect of once weekly or thrice weekly 0.05% tretinoin emollient or no treatment in 126 individuals who had completed 48 months of 0.05% once daily tretinoin therapy. Thrice weekly tretinoin treatment appeared to be more effective in improving the fine wrinkles than once weekly therapy whereas discontinuation of the therapy resulted in the reversal of beneficial effects to some extent.
Bhawan and colleagues (1996) studied the effect of long-term use (4 years) of tretinoin emollient cream in 27 patients treated with either 0.05% or 0.01% of tretinoin for the first 18 months, followed by 15-month treatment with 0.01% tretinoin and finally 19-month daily treatment with either 0.025% or 0.05% tretinoin. Histological studies indicated that the stratum corneum became compact in the first 3 to 6 months whereas it returned to normal (basket weave pattern) in 12–24 months and remained normal until the end of the therapy. Likewise, granular layer thickness and epidermal thickness were increased in the first 3–6 months, returned to normal in 12–24 months and remained normal until cessation of the therapy. In contrast, epidermal mucin continued to increase and melanin continued to decrease throughout tretinoin treatment. The changes in these 2 components clearly correlated with the observed clinical changes.
Low-strength tretinoin
The concept of low strength tretinoin had gained interest when Griffiths and colleagues (1995) reported observations made in a 48 week, double-blind, vehicle-controlled trial (n = 90) that compared clinical efficacy and tolerability of 0.025% and 0.1% tretinoin cream. The authors observed that both 0.025% and 0.1% tretinoin resulted in statistically significant improvement in all histological and clinical signs of photoaging as compared with vehicle, but there were no clinically or statistically significant differences between the two concentrations of tretinoin. However, the incidences of adverse effects were significantly greater in the 0.1% tretinoin group as compared with the 0.025% tretinoin group. Thus, it was speculated that low strength tretinoin might be a good option for those patients who can not tolerate standard therapy (0.05%). Thereafter, Nykady and colleagues (2001) conducted two 24 weeks, double-blind and vehicle-controlled trials to evaluate the efficacy and tolerability of 0.02% tretinoin cream applied once daily in 328 patients with moderate to severely photodamaged skin. Interestingly, both studies showed that there is significantly greater improvement in clinical signs of photoaging like fine wrinkling, coarse wrinkling, sallowness, and mottled hyperpigmentation (only in one study) as compared with vehicle. Moreover, the treatment was safe and well tolerated in most of the patients. Tretinoin cream 0.02% is now recognized by the FDA for the treatment of photoaging.
High strength tretinoin
High strength tretinoin treatment has been evaluated in the treatment of photoaging as the conventional tretinoin therapy has following disadvantages:
- Beneficial effects of tretinoin are seen slowly and over a long period of time, which often leads to discontinuation of therapy.
- Retinoid related adverse effects like irritation, erythema and dermatitis.
Hence, in order to minimize or avoid these disadvantages, Kligman and colleagues (1998) evaluated the potential of high strength tretinoin (0.25% solution in a fast penetrating vehicle) for the treatment of photoaging in 50 females. The treatment regimen consisted of application of highly concentrated tretinoin solution on alternate nights for 2 weeks and then every night thereafter until the end of the treatment. Interestingly, just 4 to 6 week treatment with high strength tretinoin resulted in improvement in fine wrinkling, mottled hyperpigmentation, elasticity, hydration, angiogenesis, and new collagen deposition above the zone of solar elastosis and the extent was similar to the results observed after 6 to 12 months of standard tretinoin therapy (0.05%). Moreover, the high strength tretinoin treatment was well tolerated in all patients. Subsequently, Cuce and colleagues (2001) evaluated efficacy of the 1% tretinoin solution applied twice a week in 15 women with photodamaged skin. Histological studies carried out after 15 days showed compaction of stratum corneum and increased epidermal thickness. Additionally, surface imaging studies showed improvement in skin texture and appearance.
In another study, Kligman and colleagues (2004) investigated the effect of high strength solution applied every night in 32 women with photodamaged skin. Treatment for 4 weeks resulted in significant improvement in fine wrinkles, mottled hyperpigmentation, and roughness as observed in their earlier study. The most noteworthy, but unexpected observation in this study was the rapid accommodation of the skin to retinoid side effects occurring within just 2 weeks. Hence, although typical retinoid associated side-effects were observed, they diminished very soon resulting in tolerance and better acceptance of the therapy in the patients. Yet, although high strength tretinoin has shown a good potential in photoaging, the reported studies have been carried out in a smaller population. Hence, large scale, multicentric and standard tretinoin therapy (0.05%) controlled studies are required to confirm the efficacy.
Tretinoin in intrinsic aging
To date only one vehicle-controlled clinical study has been undertaken to evaluate the use of topical tretinoin for the treatment of chronologically aged skin. In this study, 0.025% tretinoin cream was applied once daily on chronologically aged inner thigh skin of six women (mean age, 74 years) for a period of 9 months (Kligman et al 1993). The cream was applied to one inner thigh and vehicle to the other. Clinically, the improvement with thigh skin was modest; showing a less scaly, a less wrinkled, and a little firmer skin with a pink hue. In contrast, histological changes associated with tretinoin treatment were much more marked, when compared with the vehicle. Tretinoin resulted in marked increase in epidermal and granular cell layer thickness and a highly undulating dermo-epidermal junction through the development of rete pegs and produced uniformity in keratinocyte density while it decreased melanocyte vacuolization. Ultrastructurally, an increase in anchoring fibrils was noted at the level of the dermo-epidermal junction. In the dermis, development of several new micro vasculatures (angiogenesis) and production of new elastic material and GAGs was observed. These morphological changes suggested that the magnitude of effect of tretinoin might be greater for chronologically aged than for photoaged skin. However, large scale, multicentric clinical trials need to be conducted for confirming the utility of tretinoin for chronological aging.
Isotretinoin
The observed fact that topical isotretinoin (13-cis retinoic acid) results in a dose-dependent effacement of wrinkles with concomitant increase in the formation of normal connective tissues in UVB-irradiated hairless mice caught the interest of dermatologists in isotretinoin. Cunningham evaluated the potential of topical 0.1% isotretinoin cream in a randomized study of 6 months. Isotretinoin-treated patients showed statistically significant improvement in the various symptoms of photoaging like fine wrinkles and pigmentation as compared with placebo-treated subjects (Cunningham 1990). Later, in 2 separate studies, the potential of isotretinoin in treating patients suffering from mild to moderate photodamage was evaluated by Sendagorta and colleagues (1992) (n = 776) and Armstrong and colleagues (1992) (n = 326) in double-blind, vehicle-controlled clinical trials. In both studies, isotretinoin cream 0.05% was applied for 12 weeks, followed by application of higher strength isotretinoin (0.1% cream) during the next 24 weeks. Interestingly, both studies resulted in a statistically significant improvement in overall appearance, fine wrinkling, discrete pigmentation, sallowness, and texture of photoaged skin without causing any significant irritation (Armstrong et al 1992; Sendagorta et al 1992).
Similarly, Maddin et al (2000) conducted a multicentric, double-blind and placebo-controlled trial of 0.1% isotretinoin cream in 800 patients with moderate-to-severe photodamage. After 36 weeks of continuous daily treatment, the isotretinoin-treated group showed statistically significant (p < 0.01) amelioration exceeding the one in the vehicle treated group in overall appearance, fine wrinkles, texture, coarse wrinkling, and hyperpigmented macules after 12 weeks of treatment which was evident up to 36 weeks. Moreover, histological studies indicated a significant increase in epidermal thickness of skins obtained from the isotretinoin-treated group. However, no significant changes were observed in other histological parameters, such as dermal elastosis, thickness of the dermis, epidermal melanin content, number of fibroblasts, and melanocyte dysplasia or keratinocyte atypia. Although five to ten percent patients experienced severe irritation, particularly on facial skin, most of the other patients experienced only mild irritation. Moreover, the plasma level of isotretinoin did not show sustained increase in its concentration over the period of 36 weeks indicating absence of drug accumulation.
Topical isotretinoin has also been evaluated for the treatment of actinic keratoses. Alirezai and colleagues (1994) conducted a vehicle-controlled study which involved the use of 0.1% isotretinoin cream twice daily for 24 weeks. The study resulted in a statistically significant reduction in actinic keratoses and precancerous facial lesions in the isotretinoin group, with 66% of 44 patients achieving a reduction in more than one-third of lesions. However, no significant drug effect was seen for actinic keratoses on the scalp or upper extremities. Mild-to-moderate irritant reactions were observed in the isotretinoin treated group, but symptoms subsided with reduced frequency of the treatment.
Hernandez-Perez and colleagues (2000) conducted a study (n = 60) to evaluate clinical efficacy of oral isotretinoin in the treatment of photoaging. The patients included in the study were divided into 2 groups, one of which received oral isotretinoin 10 mg to 20 mg thrice a week for 2 months in addition to the facial rejuvenative treatment whereas the other group received only facial rejuvenative procedures. Interestingly, the isotretinoin-treated group showed statistically significant improvement exceeding the other group in various parameters such as wrinkles, skin thickness, tone, elasticity, and mottled hyperpigmentation. However, studies in larger populations are needed to prove the beneficial effects of oral isotretinoin therapy.
Griffiths and colleagues (2005) recently conducted a 6-month, multicentric, randomized, double-blind, parallel-group, vehicle-controlled study in 346 subjects with photoaged skin to evaluate the efficacy of 0.05% isotretinoin in combination with sunscreens applied once daily. At the end of the study, patients receiving isotretinoin and sunscreen combination showed significant improvement (p < 0.05) in fine wrinkles compared with the vehicle treated group. Moreover, the incidences of adverse effects were less in the isotretinoin-treated patients.
Retinol
Vitamin A alcohol or all-trans retinol belong to the family of endogenous natural retinoids and is a precursor for synthesis of endogenous retinal and retinoic acid. Although all-trans retinol has been used in OTC cosmetic products since 1984 (Rolewski 2003), its potential in the treatment of photoaging was realized when Kang et al (1995) showed that application of all-_trans-_retinol on normal human skin induces epidermal thickening and enhances the expression of CRABP II and CRBP mRNAs and proteins, as does retinoic acid. Moreover, the authors also observed that retinol showed only minimal signs of erythema and irritation unlike tretinoin. In another study (n = 6; duration = 14 days), Fluhr and colleagues (1999) confirmed that retinol produces considerably less transepidermal water loss, erythema and scaling than retinoic acid. Interestingly, Fisher and colleagues (1996, 1997) further demonstrated that retinol inhibits UV induction of MMP and stimulates collagen synthesis in photoaged skin. However, it was observed that retinol is 20 times less potent than tretinoin and it requires further conversion to retinoic acid (in vivo) to demonstrate its action (Kurlandsky et al 1994; Kang et al 1995). Duell and colleagues (1996) demonstrated that retinol could be as effective as retinoic acid in producing ‘retinoid mediated histological changes’ (like epidermal thickening and keratinocyte proliferation), but with much less irritancy. Pierard-Franchimont and colleagues (1998) first conducted a controlled clinical trial with retinol formulation. They observed that retinol formulation resulted in significant improvement in fine wrinkles after 12 weeks of treatment. Subsequently, Varani and colleagues (2000) studied the effect of topical application of 1% retinol in 53 individuals (80 years or above) with aged skin. The authors observed that retinol application for 7 days reduced MMP (matrix metalloproteinase), collagenase, and gelatinase expression with concomitant increase in fibroblast growth and collagen synthesis in the studied tissue specimens. Thus, it can be concluded that retinol should be effective in the treatment of aging and photoaging. However, the vehicle used for retinol delivery would play a crucial role in eliciting its efficacy, as retinol is extremely unstable and easily gets degraded to biologically inactive forms on exposure to light and air.
Retinol derivatives
Retinol derivatives have been developed in order to improve the chemical stability of retinol. Retinol derivatives like retinyl acetate, retinyl propionate, and retinyl palmitate have been widely used in cosmetic products instead of retinol. In fact, retinol derivatives were thought to be useful for the treatment of photoaging after the observation that retinyl propionate induces epidermal thickening in mouse tail and promoted collagen formation in UV-irradiated mice (Green et al 1998). Based on these encouraging results, Green and colleagues (1998) conducted a double-blind, randomized and placebo-controlled trial for 48 weeks (n = 60). Unfortunately, topical retinyl propionate cream (0.15%) did not demonstrate any statistically significant improvement over placebo in any of the evaluated histopathological or clinical symptoms of photoaging. However, in very few subjects, actinic keratoses were reduced virtually to zero by the end of 48 weeks, but this effect was not statistically significant.
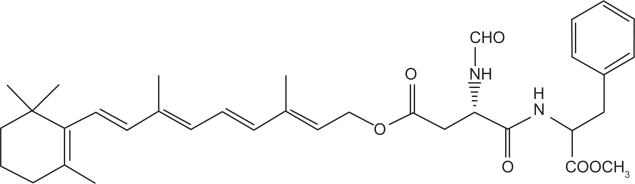
Han and colleagues (2003) have developed various retinol derivatives to improve the photostability of retinol while retaining its anti-aging activity. They found that N-formyl aspartame derivative of retinol has a good potential (Figure 2) to act as anti-aging agent since it exhibited very good photostability. Moreover, it was very well tolerated by human fibroblasts and it suppressed collagenase expression (indication of anti-aging activity) as effectively as retinol. However, elaborate studies are still missing to demonstrate its in vivo efficacy.
Figure 2.
N-formyl aspartame derivative of retinol.
Retinol combinations
Combination therapies are gaining great importance in the treatment of cutaneous disorders like acne and psoriasis as improved therapeutic effects have been observed with combination product compared with the respective monotherapy. Accordingly, researchers have recently attempted the use of retinol in combination with other anti-aging agents.
Seité and colleagues (2005) conducted two double-blind vehicle-controlled clinical studies in postmenopausal women to investigate the effects of a topically applied retinol plus vitamin C combination on epidermal and dermal compartments of aged or photoaged skin.
Study 1 involved a 3-month treatment with a combination of retinol (0.07%) and vitamin C (3.5%) applied twice daily in 8 volunteers. After the 3-month treatment, histological changes such as thinning of stratum corneum, thickening of the viable epidermis, and increase in interdigitation index were observed. In the second study, volunteers with photoaged skin were treated for 6-months with retinol (0.04%) and vitamin C (3%) combination twice daily. After the 6-month topical treatment, the observed histological changes were mainly concentrated at the dermal level. Both treated and control groups showed the same distribution pattern of type I procollagen, however, the high level of type III procollagen originally observed in photoaged skin was reduced in the retinol- and vitamin C-treated group, resulting in a lower type III-to-type I procollagen ratio. Furthermore, a wide band of eosinophilic material just beneath the epidermis, devoid of oxytalan fibers and forming the ‘grenz zone’, appeared more frequently and was larger in the retinol- and vitamin C treated group.
Finally, the authors concluded that repeated topical application of a preparation containing both retinol and vitamin C could reverse, at least in part, skin changes induced by both chronological and photoaging.
Draelos (2005) recently conducted a controlled clinical trial to evaluate the efficacy of retinol (0.3%) and hydroquinone (4%) in the treatment of photoaging in comparison with 0.05% tretinoin emollient cream. Interestingly, the retinol – hydroquinone combination diminished the collective signs of photodamage more effectively than 0.05% tretinoin emollient cream in terms of dyspigmentation, fine wrinkles, and tactile roughness within 16 weeks of treatment. Feinberg and colleagues (2004) evaluated the efficacy of retinol (0.1%) and glycolic acid (8%) combination in comparison with the individual agents in the treatment of photoaging using the photodamaged arm methodology. They observed that retinol-glycolic acid combination offered significant improvement in the appearance of photoaged skin compared with glycolic acid or retinol alone.
Barkovic and colleagues (2005) conducted a randomized, placebo-controlled, double-blind clinical trial in postmenopausal women to evaluate the efficacy of retinol – dimethylenolamine combination in the treatment of chronological aging. The study demonstrated rapid and significant improvements in the appearance of aging skin with daily application of the retinol – dimethylenolamine combination.
Retinaldehyde
Retinaldehyde is a retinoic acid precursor, which is formed as an intermediate metabolite in the transformation of retinol to retinoic acid in human keratinocytes. In the skin retinaldehyde is metabolized to retinoic acid (which is a well known anti-aging agent) as well as to retinol and retinyl esters (which generally get depleted during photoaging), indicating its use in the treatment of photoaging (Sass et al 1996). Moreover, metabolism of retinaldehyde to retinoic acid occurs only by keratinocytes at a pertinent stage of differentiation, leading to a more controlled delivery of retinoic acid and weaker retinoid associated adverse effects as compared to tretinoin and other synthetic retinoids (Saurat et al 1994; Didierjean et al 1999; Sorg et al 1999). Saurat and colleagues (1994) first evaluated the biological activity and tolerability profile of retinaldehyde on human skin. Noteworthy, topical retinaldehyde was well tolerated on human skin and it also resulted in induction of CRABP type II mRNA and protein, increased epidermal thickness, increased keratin-14 expression, and enhanced keratinocyte proliferation. However, later it was shown that retinaldehyde exerts these biological activities only on transformation to retinoic acid (Didierjean et al 1999).
In an open clinical trial, Ochando and colleagues (1994) studied the effect of 0.05% retinaldehyde in 32 female volunteers showing symptoms of mild to moderate photoaging. At the end of 4 months, considerable reduction in the surface roughness and coarse wrinkling was observed. Moreover, retinaldehyde treatment was associated with very few adverse effects. Subsequently, Creidi and colleagues (1998, 1999) conducted a randomized and vehicle-controlled clinical trial (n = 125) to compare the efficacy of 0.05% retinaldehyde with 0.05% retinoic acid for the treatment of photoaging. Optical profilometry studies on the volunteers indicated that both retinaldehyde and retinoic acid were equally effective in reducing wrinkles and skin roughness. However, retinoic acid resulted in higher incidences of local irritation than retinaldehyde thus negatively affecting patient compliance. In another study, Diridollou and colleagues (1999) evaluated the effect of 0.05% retinaldehyde versus vehicle in 40 patients showing symptoms of aging by ultrasound and rheological techniques. Compared with the control group, the retinaldehyde treated group showed a significant increase in epidermal thickness, as well as in cutaneous elasticity (p < 0.01). Similarly, retinaldehyde treatment tended to increase dermal thickness and reduce cutaneous stiffness and was very well tolerated by the patients.
Recently, Mordon and colleagues (2004) conducted a monocentric, comparative, randomized, double-blind clinical trial (n = 16) to evaluate the efficacy of retinaldehyde versus excipient both in combination with nonablative laser remodeling treatment. The study involved daily topical application of 0.05% retinaldehyde (8 patients) immediately after the first laser treatment and up to 3 months after the fifth treatment. The control group (8 patients) was treated under similar conditions, except with a daily application of vehicle instead of retinaldehyde.
At the end of the study, an increase in dermal thickness was observed for all patients treated by laser (both retinaldehyde and control groups) on the forehead and neck. However, the increase was greater for the retinaldehyde group (p < 0.05) when compared with the control group (vehicle). The increase in dermal thickness was 5.27% versus 1.13% for the forehead and 10.54% versus 3.57% for the neck, respectively.
Thus, it can be concluded that retinaldehyde is a useful topical agent for the treatment of aged and photoaged skin, with a lower frequency of irritation.
Tazarotene
Tazarotene is a novel acetylenic retinoid known to be effective in the topical treatment of psoriasis and acne. Tazarotene is a prodrug, rapidly metabolized to its active metabolite tazarotenic acid. Due to its rigid polyaromatic structure, it does not undergo any isomerization or conformational change in the skin. Although tazarotene belongs to the retinoid family, it displays a receptor selectivity pattern different from the one found with tretinoin. Tretinoin directly activates all RAR subtypes and indirectly RXRs whereas tazarotenic acid selectively binds to RAR-β and RAR-γ but not to RXRs. Tazarotenic acid modulates the expression of retinoid-responsive genes, including those that regulate cell proliferation, cell differentiation, and inflammation, corresponding to its binding capacities to various RAR receptors. Tazarotene also down-regulates the abnormal expression of keratinocytes, epidermal growth factor receptor, and hyperproliferative keratins (Nagpal et al 1995; Chandraratna 1996; DiSepio et al 1998; Roeder et al 2004).
Sefton and colleagues (2000) first conducted a pilot, double-blind, randomized trial to evaluate the efficacy of 0.1% tazarotene gel in 10 healthy women with moderate photodamage of the forearm skin. At the end of 12 weeks, significant reduction in pigmentary mottling, fine wrinkling, and skin roughness were observed in the tazarotene treated group as evidenced in silicon skin surface replicas. Moreover, histopathological investigations indicated a reduction of keratinocytic atypia and a restoration of keratinocyte polarity.
Tazarotene was further evaluated by Kang, Leyden, and colleagues (2001) who performed a multicentre, prospective, randomized study in 349 facially photodamaged subjects, to compare the efficacy of four different concentrations of tazarotene (0.01%, 0.025%, 0.05%, and 0.1%) with its vehicle and with tretinoin 0.05% emollient cream. At the end of 24 weeks, treatment success rates based on global responses (>50% improvement compared with baseline) were significantly higher in the tazarotene- and tretinoin-treated groups than in the vehicle-treated group (67% with tazarotene 0.1%, 52% with tazarotene 0.05%, 36% with tazarotene 0.025%, 41% with tazarotene 0.01%, 55% with tretinoin 0.05% and 22% with vehicle). Interestingly, a significantly greater proportion of tazarotene-treated patients showed at least 50% improvement in various clinical parameters than tretinoin-treated patients at weeks 12 and 20 thus indicating a trend towards a quicker response in tazarotene-treated patients. However, at the end of 24 weeks, there was no difference in overall improvement in the photodamage in tazarotene- or tretinoin-treated groups. In this study, both tazarotene 0.1% and tretinoin 0.05% cream showed a similar degree of improvement in epidermal thickness, fine wrinkling, lentigines, elastosis, and mottled hyperpigmentation. The local adverse events observed were generally of mild or moderate severity, and were greater (mainly burning) with higher tazarotene concentrations.
Recently, Lowe and colleagues (2004) again compared the efficacy of tazarotene 0.1% cream and tretinoin 0.05% emollient cream in the treatment of photoaging in a double-blind, randomized, multicentre, 24 week study (n = 173). At week 16, the incidence of treatment success (>50% global improvement) at the study endpoint was 78% in the tazarotene group and 67% in the tretinoin emollient group, with statistical significance in favor of tazarotene. All other significant differences in efficacy measures were also in favor of tazarotene – for the overall integrated assessment of photodamage at week 16, fine wrinkling at week 24, mottled hyperpigmentation at weeks 12 and 16, and coarse wrinkling at week 4. There were no significant between-group differences in the incidence of subjects achieving at least a 1-grade improvement in irregular depigmentation, lentigines, appearance of pore size, elastosis, tactile roughness, telangiectasia, and actinic keratoses. The local adverse events observed were generally of mild or moderate severity and were greater (mainly burning) with higher tazarotene concentrations. Thus, the efficacy and tolerability results obtained from this study are in broad agreement with those reported by Kang, Leyden, and colleagues (2001).
Phillips and colleagues (2002) conducted a multicentre, double-blind, randomized, vehicle-controlled clinical trial that involved treatment of 563 subjects with facial photodamage with tazarotene 0.1% cream for 24-weeks followed by a 28-week open-label extension. At week 24, when compared with vehicle, tazarotene resulted in a significantly greater proportion of patients achieving treatment success (>50% greater improvement) and at least a 1 grade improvement in fine wrinkling, mottled pigmentation, pore size, lentigines, elastosis, irregular depigmentation, tactile roughness, coarse wrinkling, and overall integrated assessment of photodamage. Pigmentary changes were the first to respond to treatment, showing statistically significant improvement over vehicle after 2 weeks of treatment. Fine wrinkling improved in the tazarotene group after 4 weeks of treatment, and coarse wrinkles, elastosis, and pore size were significantly improved over vehicle by week 12. Telangiectasia and actinic keratoses were not significantly improved after 24 weeks of treatment, these two variables not being the primary outcome measures of the study. After the first 24 weeks, an open label extension of tazarotene 0.1% cream was followed for another 28 weeks. Additional clinical improvement was noted, which did not plateau after 52 weeks of treatment, suggesting the value of long-term treatment. Irritation was generally mild or moderate and declined with ongoing treatment, whereas plasma tazarotenic acid concentration did not exceed the plasma levels of endogenous retinoids.
Recently, tazarotene has been evaluated once again against photodamage in two subsequent studies. Machtinger and colleagues (2004) evaluated the histological effects of tazarotene cream in 50 patients with photodamaged facial skin by conducting a multicentre, double-blind, randomized, vehicle-controlled study. Blinded assessments showed that the tazarotene-treated group showed amelioration of keratinocytic and melanocytic atypia compared to the vehicle-treated group. Between-group comparisons in distribution of change from baseline categories of severity were in favour of tazarotene (p = 0.055 for keratinocytic atypia, p = 0.034 for melanocytic atypia, and p < 0.001 for the number of granular cell layers). Compared with vehicle, tazarotene was associated with an increase in epidermal polarity (p = 0.008) and epidermal thickness (p = 0.012), and a tendency for stratum corneum compaction. Tazarotene was also associated with widened intercellular spaces relative to vehicle (p < 0.001).
In another study, Kang and colleagues (2005) evaluated the efficacy and tolerability of tazarotene 0.1% cream in the treatment of 568 patients with facial photodamage. Topical tazarotene offered efficacy in ameliorating multiple effects of photodamage. Tazarotene cream was significantly more effective than vehicle in reducing fine wrinkles, mottled hyperpigmentation, lentigines, irregular depigmentation, apparent pore size, elastosis, tactile roughness, and an overall integrated assessment of photodamage. Significance was achieved as early as at week 2 with mottled hyperpigmentation, lentigines, irregular depigmentation and elastosis and had not plateaued by week 24. The majority of patients reported improvements in their photodamage as early as at week 4. Adverse events were predominantly mild or moderate signs or symptoms of skin irritation. Thus, topical tazarotene has been shown to offer efficacy in ameliorating multiple effects of photodamage.
Adapalene
Adapalene is considered to be a third-generation synthetic retinoid which contains a napthoic acid backbone. Unlike retinoic acid, adapalene shows selectivity for the nuclear retinoic acid receptor (RAR β/γ). It targets abnormal desquamation of the skin, modulates cellular differentiation, and possesses anti-inflammatory properties (Leyden 2001). Moreover, due to its receptor selectivity, it causes less skin irritation. Adapalene is successfully being used for the treatment of acne. However, not much has been done to investigate its potential in aging/photoaging. So far only one study has been carried out to determine the potential of adapalene in photoaging.
Kang and colleagues (2003) conducted a 2-center, randomized, vehicle-controlled, investigator-masked, parallel-group study in 83 patients suffering from actinic keratoses and solar lentigines and other symptoms of photoaging to evaluate the therapeutic potential of adapalene gel (0.1% or 0.3%) or vehicle for 4 weeks, followed by twice-daily applications, if tolerated, for up to 9 months. After 9 months, 0.5 + 0.9% and 2.5 + 0.9%, reduction in actinic keratoses was observed with adapalene gel 0.1% and 0.3%, respectively. Whereas, with the vehicle gel actinic keratoses increased by 1.5 + 1.3% (p < 0.005).
Significant lightening of solar lentigines was observed in patients treated with adapalene gel as compared with the patients treated with vehicle gel (p < 0.05) within 1 month of treatment. At the end of 9 months, 57% and 59% of the patients had brighter lesions in the adapalene 0.1% and 0.3% groups, respectively, in comparison with only 36% in the vehicle group (p < 0.05). Moreover, histological evaluations revealed improved cellular atypia and reduced epidermal melanin content in the adapalene-treated group compared with the vehicle-treated group. A retrospective evaluation of paired clinical photographs (before and after 9-month treatment) revealed significant improvement in wrinkles and other clinical features of photoaged skin with adapalene as compared with its vehicle. As expected, adapalene was very well tolerated by the patients involved in the study. Thus, adapalene can be employed as a second line treatment of photoaging mainly in patients demonstrating extreme intolerance to conventional retinoids. However, large scale clinical trials should be carried out to validate the efficacy of adapalene.
Alitretinoin
Alitretinoin or 9-cis-retinoic acid is a naturally occurring endogenous retinoid that binds to and activates all known intracellular RAR and RXR subtypes (Cheer and Foster 2000). Its use in the topical treatment of AIDS-associated Kaposi’s sarcoma is well documented in the literature (Bodsworth et al 2001). Additionally, its safety and efficacy in the normal treatment of chronic hand dermatitis has also been proved (Ruzicka et al 2004). Considering the RAR binding activity of alitretinoin, Baumann and colleagues (2005) recently conducted an open-label pilot study in 20 patients with photodamaged skin to evaluate the efficacy of 0.1% topical alitretinoin. The topical alitretinoin treatment showed improvement in seborrheic keratoses, actinic keratoses, and other symptoms of photoaging. Moreover, the treatment was well tolerated by the patients. However, larger, blinded, controlled trials are needed to know the role and efficacy of alitretinoin in the treatment of photoaging.
Seletinoid G
Seletinoid G represents a fourth generation of retinoids that might find an important place in the treatment of intrinsic/photo aging. Like other synthetic retinoids, seletinoid G shows receptor selectivity for RAR-γ, which is predominantly expressed in the epidermis as compared with other RARs. Recently, Kim and colleagues (2005) evaluated the safety and efficacy of seletinoid G in comparison with tretinoin for the treatment of intrinsic/photo-aging after topical application in 23 patients belonging to differing age groups. Notably, intrinsically aged skin after topical treatment with seletinoid G showed increase in the expressions of type I procollagen, tropoelastin, and fibrillin-1, and reduced MMP-1 similar to that of tretinoin demonstrating its potential in the treatment of intrinsic aging. Moreover, in the UV-irradiated young skin, topical seletinoid G treatment inhibited UV-induced decrease of type I procollagen and UV-induced increase of MMP-1 and c-Jun protein similar to that seen with tretinoin. Interestingly, topical application of seletinoid G under occlusion induced no skin irritation in contrast to tretinoin, which caused severe erythema. Thus, seletinoid G appears to be as effective as tretinoin in the treatment of intrinsic/photo aging with the added advantage of absence of skin irritation. However, larger, blinded, controlled trials are needed to validate the role and benefit-to-risk ration of seletinoid G in the treatment of intrinsic/photo aging.
Adverse effects of topical retinoids
The most common and frequent adverse effect of topical retinoids are known as ‘retinoid reaction’, characterized by pruritus, burning sensation at the sites of application, erythema, peeling. It is more common with tretinoin and tazarotene than with isotretinoin, adapalene, retinol, and retinaldehyde. The ‘retinoid reaction’ has been found to be due to the free carboxylic acid in the polar end of the retinoid, which is evident from the activity and toxicity experiments done on CHO cells (Oda et al 1996). ‘Retinoid reaction’ manifests itself generally within the first few weeks of treatment and is thought to get initiated by release of proinflammatory cytokines such as IL-1, TNF-α, IL-6, and IL-8 (Torras 1996; Orfanos et al 1997). In order to validate this hypothesis, Kim and colleagues (2003) evaluated the changes in mRNA expression of inflammation-related cytokines such as human monocyte chemoattractant protein-1 (MCP-1), IL-8, TNF-α, interferon-γ (IFN-γ), IL-6, IL-10, after treatment of epidermal cells with retinoic acid and retinol. A minimum 3-fold increase in the mRNA and protein levels of mainly MCP-1 and IL-8 were found, thus validating the hypothesis to some extent.
The other side effect associated with retinoid therapy is photosensitization, which normally occurs at the beginning of the therapy. Patients on retinoid therapy are advised to avoid excessive sun exposure and take precautionary measures (like use of sunscreens) for sun protection. However, after few months of therapy, the skin’s response to UV radiation returns to normal. In certain cases, irritant conjunctivitis has also been reported when the retinoid is applied close to the eye (Torras 1996).
Over the past 30 years, no systemic side-effects on long-term treatment with the topical retinoid have been observed in young adults. Moreover, Lattarino and colleagues (1997) found that topical tretinoin had no detectable effect on endogenous plasma levels of tretinoin or its metabolites. This is mainly because of the limited transdermal uptake of these agents (Krautheim and Gollnick 2003). Pregnant women or women of child-bearing age should use topical retinoids with caution as retinoids on systemic exposure are known to cause teratogenicity/embryotoxicity. However, in over 25 years of topical tretinoin use for acne therapy there have been no cases of related teratogenicity (Kligman 1988). Additionally, in one case-control study of 215 mothers who were exposed to topical tretinoin in the first trimester of pregnancy, the prevalence of major fetal malformations in the tretinoin-exposed group was 1.9% versus 2.6% in the control group, indicating the safety of topical tretinoin (Jick et al 1993). Even then, it is prudent to advise women of childbearing age to avoid pregnancy during treatment or, if pregnant, to discontinue the use of topical retinoids.
Counteracting the adverse effects
To counteract the symptoms of retinoid reaction, reduction in the frequency of application or switching to a less irritating retinoid is normally advised. Moreover, addition of 3% indomethacin or 1% hydrocortisone to the retinoid formulation, or concomitant treatment with a low to medium potency topical corticosteroid has also been suggested to relieve the symptoms (Torras 1996). However, if patients cannot tolerate even the lowest concentration of retinoids, then the treatment should be discontinued.
Kim and colleagues (2003) have demonstrated that concomitant application of natural agents or extracts like β-sitosterol, Magnoliae flos, β-glycyrrhetinic acid, Scleroglucan, Gingko extract, Raspberry extract, Schisandra extract, Cola extract, Enna complex, or Vegetol red grapevine extract could also be useful in counteracting the irritant effects produced by topical retinoids. The abovementioned natural agents or extracts reduced the secretion of MCP-1 and IL-8 from human fibroblasts and also showed a good protection against the retinoid-induced irritation in the rabbit and human patch test. A different approach to reduce adverse events of retinoids was applied by Kambayashi et al (2005). Topical application of a new synthetic retinoid (N-retinoyl-D-glucosamine) to hairless mice showed good efficacy in repair of photoaged skin but did not induce skin irritations compared with retinoic acid.
Although the aforementioned approaches have shown potential in minimizing the side effects, they involve co-administration of other therapeutic agents, which may have its own effects on prolonged duration. The need of hour is to have an approach that can efficiently counteract the adverse effects associated with retinoid therapy and would not require co-administration of any therapeutic agent. In fact, we believe that the same could be achieved by modulating the delivery system/s employed for topical retinoids. The further part of the review describes the potential of delivery systems in optimizing topical retinoids therapy with concomitant minimization in the adverse effects associated with it.
Potential of delivery systems in topical retinoids therapy
Drug delivery strategies are well known in pharmaceutical research for their potential in optimizing efficacy of therapeutic agents by either modulating their physicochemical and biopharmaceutical properties or minimizing/eliminating the side effects associated with them, thus offering better patient compliance. The arrival of controlled release systems, transdermal delivery systems, implants, submicronic emulsions, and vesicular carriers is sufficient to substantiate the aforementioned advantage of delivery systems. Since the last decade, there has been considerable interest in investigating the approaches for improved delivery of retinoids.
The delivery system can be considered to be efficient for topical retinoids if it can:
- Minimize/abolish the adverse effects of topical retinoids.
- Improve the stability (mainly photochemical) of retinoids like retinoic acid and retinol.
- Enhance the anti-aging effect of retinoids by modulating their dermal transport or distribution.
Considering the above points, we believe that nanoparticles can be utilized as an efficient delivery system to optimize the topical retinoid therapy.
Nanoparticles are solid colloidal particles, ranging in size from 1 nm to 1000 nm, consisting of various biocompatible matrices in which a therapeutic moiety can be adsorbed, entrapped, or covalently attached (Lockman et al 2002).
Based on the nanoparticles matrix (or shell), they are be classified as:
- Polymeric nanoparticles,
- Solid lipid nanoparticles,
- Inorganic nanoparticles.
Nanoparticles are extensively being investigated for drug delivery in the pharmaceutical research from the last 3 decades on. Nanoparticles offer immense benefits such as solubilization of hydrophobic actives, improvement in bioavailability, improved (or altered) pharmacokinetics of active pharmaceutical ingredient (API), protection of API from physical, chemical or biological degradation, improved cellular uptake, tissue targeting and controlled release of API. However, most of the initial investigations on nanoparticles dealt with the parenteral or oral delivery of APIs whereas efforts to explore the potential of nanoparticles in topical delivery were initiated in the last decade. Interestingly, nanoparticles were found to improve the distribution characteristics and stability of topically applied APIs such as sunscreens (Wissing and Muller 2002).
In addition, they have been shown to offer localized and targeted delivery of APIs which can be helpful in improving their efficacy with concomitant reduction in systemic side effects associated.
Jenning and colleagues (Jenning, Gylser, et al 2000; Jenning, Schäfer-Korting, et al 2000; Jenning and Gohla 2001) first evaluated the potential of solid lipid nanoparticles (SLNs) for the delivery of retinol. Interestingly, SLNs offered a significant improvement in the stability of retinol as compared with the conventional emulsion. Moreover, highly localized and controlled release of retinol could be observed with the SLNs. Patravale and colleagues (2004) investigated the potential of SLNs in counteracting the irritant effects and in improving the physical stability of the most commonly employed retinoid, ie, tretinoin. In fact, SLN-based tretinoin gels showed drastic improvement in the tolerability of tretinoin as compared with marketed products when evaluated by the Draize patch test in rabbits. Furthermore, photostability of tretinoin was markedly improved in SLNs as compared with methanolic solution. Again as expected, the tretinoin SLNs showed a high degree of localization in skin when evaluated by in vitro skin permeation studies. All these effects were attributed to the encapsulation of tretinoin in nanoparticulate structures.
Recently, Yamaguchi and colleagues (2005) have investigated the potential of inorganic nanoparticles of tretinoin produced by boundary-organized reaction. As observed earlier, tretinoin encapsulation in nanoparticles resulted in significantly less irritation and inflammation as compared with conventional formulation. In addition, nanoparticulate tretinoin showed drastic improvement in photostability as compared with conventional formulation, which was retained even after 46 days of storage. However, the most striking results were observed in the ex-vivo studies.
Histological evaluation in mouse epidermis after daily application of nanoparticulate tretinoin cream for 4 days revealed more than double increase in epidermal thickness than conventional tretinoin vaseline preparation. Additionally, nanoparticulate tretinoin treatment resulted in a significant increase in the mRNA levels of heparin-binding epidermal growth factor. This observation was in good correlation with the increased epidermal thickening observed with nanoparticulate tretinoin. Interestingly, 4 days’ treatment of aged skin of hairless mice with nanoparticulate tretinoin showed significant improvement in fine and coarse wrinkling and texture in the neck area. To our knowledge, this is the first investigation, which clearly demonstrated that nanoparticles could be an ideal approach in optimizing the topical retinoid therapy with concomitant reduction in the side effects associated with this therapy.
Other delivery strategies for retinoids
Literature indicates that apart from nanoparticles, various other formulation approaches like liposomes, microsponges, microemulsions, and inclusion complexes with cyclodextrins could also be employed for improving the topical delivery of retinoids. Their success in improving the stability, tolerability, and efficacy (in acne treatment) of retinoids is well established. However, none of them have been evaluated for improving the efficacy of retinoids in the treatment of aging. The detailed description of their potential in improving the efficacy and tolerability in the treatment of acne has been described by Date and colleagues (2006).
Conclusion and outlook
Aging research is divided into 2 main streams the one being the exploration of various pathophysiological and molecular events responsible for aging and the other being investigation on various anti-aging agents. Although much elaborate mechanistic studies have been carried out for understanding the pathophysiology of aging, they will still continue until the complete cascade of molecular events responsible for intrinsic/photoaging is elucidated. Amongst various anti-aging agents, retinoids are the most promising agents that are available for the treatment of aging. Amongst retinoids, tretinoin is the most potent and best-studied retinoid. However, its irritation potential has prompted dermatologists to switch over to less irritating but comparably effective retinoids like adapalene and to some extent retinol and retinaldehyde. Receptor specific retinoids like seletinoid G have been developed with the same vision and have been found to be successful in small-scale studies.
We believe that future efforts in retinoid research will be directed in following ways
- Development of receptor selective synthetic retinoids (like seletinoid G) or novel retinoid derivatives (like N-formyl aspartamate derivative of retinol) or retinoid co-drugs like retinyl ascorbate (Abdulmajed and Heard 2004) which may be superior in terms of tolerability, stability and efficacy.
- Exploration of natural sources to identify agents or extracts that may have retinoid like activity as in case of PADMA 28 (Aslam et al 2005).
- Evaluation of various combinations of anti-aging agents having synergistic effects (analogous to combination therapy in acne and psoriasis).
- Development and clinical evaluation of nanoparticulate carriers for retinoids.
Considering the developmental cost and the success rates associated with the new chemical entities, there is limited potential for developing novel synthetic retinoids. At the same time, retinoid-like activity as shown by PADMA 28 opens a new era in the identification of natural products for anti-aging treatment. As natural products have the well known benefit of good acceptability we expect stimulation in this area of research. Combination therapy has been well established for cutaneous disorders like acne and psoriasis. As relatively less developmental efforts are required for commercializing new combinations, there is scope for developing retinoid based combination therapies for improved treatment of aging. Finally, in our opinion, there is great scope for development of various drug delivery systems (especially nanoparticulate systems) to optimize the aging treatment with topical retinoids. We believe that among various nanoparticulate carriers, SLNs would have the greatest potential in optimizing the retinoid therapy as apart from their advantage as a carrier they are also known to have a UV-blocking effect, which may help in reducing photosensitization induced by retinoids. Interestingly, in one study, a SLN-based anti-aging product was more effective in reducing the depth of wrinkles (10.3%) as compared with the same product based on conventional vehicle (4.1%) indicating that SLNs themselves may have some effect on improving wrinkling (Muller et al 2002). We believe that future efforts in SLNs should be focused on proving its potential to counteract photosensitivity and to identify the potential of SLNs (blank or in combination with retinoids) in improving the elasticity and wrinkling of intrinsically/photo aged skin. Finally, complementary efforts from clinicians are required to validate the potential of drug delivery strategies in optimizing treatment of aging with topical retinoids.
References
- Abdulmajed K, Heard C. Topical delivery of retinyl-ascorbate co-drug 1. Synthesis, penetration, into and permeation across human skin. Int J Pharm. 2004;280:113–24. doi: 10.1016/j.ijpharm.2004.05.008. [DOI] [PubMed] [Google Scholar]
- Alirezai M, Dupuy R, Amblard P, et al. Clinical evaluation of topical isotretinoin in the treatment of actinic keratoses. J Am Acad Dermatol. 1994;30:447–51. doi: 10.1016/s0190-9622(94)70054-0. [DOI] [PubMed] [Google Scholar]
- Antille C, Tran C, Sorg O, et al. Penetration and metabolism of topical retinoids in ex-vivo organ-cultured full-thickness human skin explants. Skin Pharmacol Physiol. 2004;17:124–8. doi: 10.1159/000077238. [DOI] [PubMed] [Google Scholar]
- Armstrong RB, Lesiewicz J, Harvey G, et al. Clinical panel assessment of photodamaged skin treated with isotretinoin using photographs. Arch Dermatol. 1992;128:352–6. [PubMed] [Google Scholar]
- Aslam MN, Fligiel H, Lateef H, et al. PADMA 28: A multi-component herbal preparation with retinoid like dermal activity but without epidermal effects. J Invest Dermatol. 2005;124:524–9. doi: 10.1111/j.0022-202X.2004.23596.x. [DOI] [PubMed] [Google Scholar]
- Astrom A, Tavakkol A, Pettersson U, et al. Molecular cloning of two human cellular retinoic acid-binding proteins (CRABP) J Biol Chem. 1991;266:17662–6. [PubMed] [Google Scholar]
- Barkovic S, Leyden JJ, Wu IT, et al. Clinical evaluation of topical retinol-dimethylenolamine (DEMA) treatments for aging skin. J Am Acad Dermatol. 2005;52(3:S1):P27. [Google Scholar]
- Baumann L, Vujevich J, Halem M, et al. Open-label pilot study of 0.1% alitretinoin gel in the treatment of photoaging. Cutis. 2005;76:69–73. [PubMed] [Google Scholar]
- Bernstein EF, Uitto J. Connective tissue alterations in photoaged skin and the effects of alphahydroxy acids. J Geriatr Dermatol. 1995;3(Suppl. A):7–18A. [Google Scholar]
- Bhawan J, Gonzalez-Serva A, Nehal K, et al. Effects of tretinoin on photodamaged skin. A histologic study. Arch Dermatol. 1991;127:666–72. [PubMed] [Google Scholar]
- Bhawan J, Palco MJ, Lee J, et al. Reversible histologic effects of tretinoin on photodamaged skin. J Geriatr Dermatol. 1995;3:62–7. [Google Scholar]
- Bhawan J, Olsen E, Lufrano L, et al. Histologic evaluation of the long-term effects of tretinoin on photodamaged skin. J Dermatol Sci. 1996;11:177–82. doi: 10.1016/0923-1811(95)00432-7. [DOI] [PubMed] [Google Scholar]
- Bodsworth NJ, Bloch M, Bower M, et al. Phase III vehicle-controlled, multi-centered study of topical alitretinoin gel 0.1% in cutaneous AIDS-related Kaposi’s sarcoma. Am J Clin Dermatol. 2001;2:77–87. doi: 10.2165/00128071-200102020-00004. [DOI] [PubMed] [Google Scholar]
- Brand MJ, Petkovich M, Krust A, et al. Identification of a second human retinoic acid receptor. Nature. 1988;332:850–3. doi: 10.1038/332850a0. [DOI] [PubMed] [Google Scholar]
- Caputo R, Monti M, Motta S, et al. The treatment of visible signs of senescence: the Italian experience. Br J Dermatol. 1990;122(Suppl 35):97–103. doi: 10.1111/j.1365-2133.1990.tb16133.x. [DOI] [PubMed] [Google Scholar]
- Chandraratna RA. Tazarotene-first of a new generation of receptor-selective retinoids. Br J Dermatol. 1996;135(Suppl. 49):18–25. doi: 10.1111/j.1365-2133.1996.tb15662.x. [DOI] [PubMed] [Google Scholar]
- Cheer SM, Foster RH. Alitretinoin. Am J Clin Dermatol. 2000;1:307–16. doi: 10.2165/00128071-200001050-00005. [DOI] [PubMed] [Google Scholar]
- Creidi P, Vienne MP, Ochonisky S, et al. Profilometric evaluation of photodamage after topical retinaldehyde and retinoic acid treatment. J Am Acad Dermatol. 1998;39:960–5. doi: 10.1016/s0190-9622(98)70270-1. [DOI] [PubMed] [Google Scholar]
- Creidi P, Humbert P. Clinical use of topical retinaldehyde on photoaged skin. Dermatology. 1999;199(Suppl 1):49–52. doi: 10.1159/000051379. [DOI] [PubMed] [Google Scholar]
- Cuce LC, Bertino MC, Scattone L, et al. Tretinoin peeling. Dermatol Surg. 2001;27:12–4. [PubMed] [Google Scholar]
- Cunningham WJ. Topical isotretinoin and photodamage. Dermatologica. 1990;181:350–1. [Google Scholar]
- Date AA, Naik B, Nagarsenker MS. Novel drug delivery systems: potential in improving topical delivery of antiacne agents. Skin Pharmacol Physiol. 2006;19:2–16. doi: 10.1159/000089138. [DOI] [PubMed] [Google Scholar]
- Didierjean I, Tran C, Sorg O, et al. Biological activities of topical retinaldehyde. Dermatology. 1999;1(Suppl 1):19–24. doi: 10.1159/000051373. [DOI] [PubMed] [Google Scholar]
- Diridollou S, Vienne MP, Alibert M, et al. Efficacy of topical 0.05% retinaldehyde in skin aging by ultrasound and rheological techniques. Dermatology. 1999;199(Suppl 1):37–41. doi: 10.1159/000051377. [DOI] [PubMed] [Google Scholar]
- DiSepio D, Ghosn C, Eckert RL, et al. Identification and characterization of a retinoid-induced class II tumor suppressor/growth regulatory gene. Proc Natl Acad Sci U S A. 1998;95:14811–15. doi: 10.1073/pnas.95.25.14811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Draelos S. Novel approach to the treatment of hyperpigmented photodamaged skin: 4% hydroquinone/0.3% retinol versus tretinoin 0.05% emollient cream. Dermatol Surg. 2005;31:799–804. doi: 10.1111/j.1524-4725.2005.31723. [DOI] [PubMed] [Google Scholar]
- Duell EA, Derguini F, Kang S, et al. Extraction of human epidermis treated with retinol yields retro-retinoids in addition to free retinol and retinyl esters. J Invest Dermatol. 1996;107:178–82. doi: 10.1111/1523-1747.ep12329576. [DOI] [PubMed] [Google Scholar]
- Ellis CN, Weiss JJ, Hamilton TA, et al. Sustained improvement with prolonged topical tretinoin (retinoic acid) for photoaged skin. J Am Acad Dermatol. 1990;23:629–37. doi: 10.1016/0190-9622(90)70265-j. [DOI] [PubMed] [Google Scholar]
- Feinberg C, Hawkins S, Battaglia A, et al. Comparison of anti-aging efficacy from cosmetic ingredients on photoaged skin. J Am Acad Dermatol. 2004;50(3:S1):P27. [Google Scholar]
- Fisher GJ, Talwar HS, Xiao JH, et al. Immunological identification and functional quantification of retinoic acid and retinoic X receptor proteins in the human skin. J Biol Chem. 1994;269:20629–35. [PubMed] [Google Scholar]
- Fisher GJ, Datta SC, Talwar HS, et al. The molecular basis of sun induced premature ageing and retinoid antagonism. Nature. 1996;379:335–8. doi: 10.1038/379335a0. [DOI] [PubMed] [Google Scholar]
- Fisher GJ, Wang ZQ, Datta SC, et al. Pathophysiology of premature skin aging induced by ultraviolet light. N Engl J Med. 1997;337:1419–28. doi: 10.1056/NEJM199711133372003. [DOI] [PubMed] [Google Scholar]
- Fluhr JW, Vienne MP, Lauze C, et al. Tolerance profile of retinol, retinaldehyde and retinoic acid under maximized and long-term clinical conditions. Dermatology. 1999;199(Suppl 1):57–60. doi: 10.1159/000051381. [DOI] [PubMed] [Google Scholar]
- Giguère V, Ong ES, Segui P, et al. Identification of a receptor for the morphogen retinoic acid. Nature. 1987;330:624–9. doi: 10.1038/330624a0. [DOI] [PubMed] [Google Scholar]
- Gilchrest BA. A review of skin aging and its medical therapy. Br J Dermatol. 1996;135:867–75. doi: 10.1046/j.1365-2133.1996.d01-1088.x. [DOI] [PubMed] [Google Scholar]
- Green LJ, McCormick A, Weinstein GD. Photoaging and the skin: the effects of tretinoin. Dermatol Clin. 1993;11:97–105. [PubMed] [Google Scholar]
- Green C, Orchard G, Cerio R, et al. A clinicopathological study of the effects of topical retinyl propionate cream in skin photoaging. Clin Exp Dermatol. 1998;23:162–7. doi: 10.1046/j.1365-2230.1998.00331.x. [DOI] [PubMed] [Google Scholar]
- Griffiths CEM, Finkel IJ, Tranfaglia MG, et al. An in-vivo experimental model for topical retinoid effects on human skin. Br J Dermatol. 1993;29:389–99. doi: 10.1111/j.1365-2133.1993.tb03163.x. [DOI] [PubMed] [Google Scholar]
- Griffiths CEM, Kang S, Ellis CN, et al. Two concentrations of topical tretinoin (retinoic acid) cause similar improvement of photoaging but different degrees of irritation. Arch Dermatol. 1995;131:1037–44. [PubMed] [Google Scholar]
- Griffiths CEM. Dowling Oration delivered at the Royal College of Physicians, London, Retinoids: renaissance and reformation. Clin Exp Dermatol. 1998;24:329–35. doi: 10.1046/j.1365-2230.1999.00491.x. [DOI] [PubMed] [Google Scholar]
- Griffiths CE, Maddin S, Wiedow O, et al. Treatment of photoaged skin with a cream containing 0.05% isotretinoin and sunscreens. J Dermatol Treat. 2005;16:79–86. doi: 10.1080/09546630510027732. [DOI] [PubMed] [Google Scholar]
- Han HS, Kwon YJ, Park MS, et al. Efficacy validation of synthesized retinol derivatives in vitro: stability, toxicity, and activity. Bioorg Med Chem. 2003;11:3839–45. doi: 10.1016/s0968-0896(03)00334-1. [DOI] [PubMed] [Google Scholar]
- Hernandez-Perez E, Khawaja H, Alvarez T. Oral isotretinoin as part of the treatment of cutaneous aging. Dermatol Surg. 2000;26:649–52. doi: 10.1046/j.1524-4725.2000.99210.x. [DOI] [PubMed] [Google Scholar]
- Jenning V, Gysler A, Schäfer-Korting M, et al. Vitamin A loaded solid lipid nanoparticles (SLN) for topical use: occlusive properties and drug targeting to the upper skin. Eur J Pharm Biopharm. 2000a;49:211–18. doi: 10.1016/s0939-6411(99)00075-2. [DOI] [PubMed] [Google Scholar]
- Jenning V, Schäfer-Korting M, Gohla S. Vitamin A loaded solid lipid nanoparticles (SLN) for topical use: drug release properties. J Control Rel. 2000b;66:115–26. doi: 10.1016/s0168-3659(99)00223-0. [DOI] [PubMed] [Google Scholar]
- Jenning V, Gohla S. Encapsulation of retinoids in solid lipid nanoparticles (SLN) J Microencapsul. 2001;18:149–58. doi: 10.1080/02652040010000361. [DOI] [PubMed] [Google Scholar]
- Jick SJ, Terris BZ, Jick H. First trimester topical tretinoin and congenital disorders. Lancet. 1993;341:1181–2. doi: 10.1016/0140-6736(93)91004-6. [DOI] [PubMed] [Google Scholar]
- Kambayashi H, Odake Y, Takada K, et al. N-retinoyl-D-glucosamine, a new retinoic acid agonist, mediates topical retinoid efficacy with no irritation on photoaged skin. Br J Dermatol. 2005;153:30–36. doi: 10.1111/j.1365-2133.2005.06967.x. [DOI] [PubMed] [Google Scholar]
- Kang S, Duell EA, Fisher GJ, et al. Application of retinol to human skin in vivo induces epidermal hyperplasia and cellular retinoid binding proteins characteristic of retinoic acid but without measurable retinoic acid levels or irritation. J Invest Dermatol. 1995;105:549–56. doi: 10.1111/1523-1747.ep12323445. [DOI] [PubMed] [Google Scholar]
- Kang S, Fisher G, Voorhees JJ. Photoaging: Pathogenesis, prevention and treatment. Clin Ger Med. 2001;17:643–59. doi: 10.1016/s0749-0690(05)70091-4. [DOI] [PubMed] [Google Scholar]
- Kang S, Leyden JJ, Lowe NJ, et al. Tazarotene cream for the treatment of facial photodamage. Arch Dermatol. 2001;137:1597–604. doi: 10.1001/archderm.137.12.1597. [DOI] [PubMed] [Google Scholar]
- Kang S, Goldfarb M, Weiss J, et al. Assessment of adapalene gel for the treatment of actinic keratoses and lentigines: A randomized trial. J Am Acad Dermatol. 2003;49:83–90. doi: 10.1067/mjd.2003.451. [DOI] [PubMed] [Google Scholar]
- Kang S, Krueger GG, Tanghetti EA, et al. A multicenter, randomized, double-blind trial of tazarotene 0.1% cream in the treatment of photodamage. J Am Acad Dermatol. 2005;52:268–74. doi: 10.1016/j.jaad.2004.06.021. [DOI] [PubMed] [Google Scholar]
- Kim BH, Lee YS, Kang KS. The mechanism of retinol-induced irritation and its application to anti-irritant development. Toxicol Lett. 2003;146:65–73. doi: 10.1016/j.toxlet.2003.09.001. [DOI] [PubMed] [Google Scholar]
- Kim MS, Lee SR, Rho HS, et al. The effects of a novel synthetic retinoid, seletinoid G, on the expression of extracellular matrix proteins in aged human skin in vivo. Clinica Chimica Acta. 2005;362:161–9. doi: 10.1016/j.cccn.2005.06.016. [DOI] [PubMed] [Google Scholar]
- Kligman LH, Chen HD, Kligman AM. Topical retinoic acid enhances the repair of ultraviolet damaged dermal connective tissue. Connect Tissue Res. 1984;12:139–50. doi: 10.3109/03008208408992779. [DOI] [PubMed] [Google Scholar]
- Kligman AM, Grove GL, Hirose R, et al. Topical tretinoin for photoaged skin. J Am Acad Dermatol. 1986;15:836–59. doi: 10.1016/s0190-9622(86)70242-9. [DOI] [PubMed] [Google Scholar]
- Kligman AM. Is topical tretinoin teratogenic? [letter] JAMA. 1988;259:2918. [Google Scholar]
- Kligman AM, Dogadkina D, Lavker RM. Effects of topical tretinoin on the non-sun exposed protected skin of the elderly. J Am Acad Dermatol. 1993;29:25–33. doi: 10.1016/0190-9622(93)70147-l. [DOI] [PubMed] [Google Scholar]
- Kligman DE, Sadiq I, Pagnoni A, et al. High-strength tretinoin: a method for rapid retinization of facial skin. J Am Acad Dermatol. 1998;39:S93–7. doi: 10.1016/s0190-9622(98)70454-2. [DOI] [PubMed] [Google Scholar]
- Kligman DE, Draelos ZD. High-strength tretinoin for rapid retinization of photoaged skin. Dermatol Surg. 2004;30:864–6. doi: 10.1111/j.1524-4725.2004.30254.x. [DOI] [PubMed] [Google Scholar]
- Krautheim A, Gollnick H. Transdermal penetration of topical drugs used in the treatment of acne. Clin Pharmacokinet. 2003;42:1287–304. doi: 10.2165/00003088-200342140-00005. [DOI] [PubMed] [Google Scholar]
- Kurlandsky SB, Xiao JH, Duell EA, et al. Biological activity of all-trans retinol requires metabolic conversion to all-trans-retinoic acid and is mediated through activation of nuclear retinoid receptors in human kertinocytes. J Biol Chem. 1994;269:32821–7. [PubMed] [Google Scholar]
- Latriano L, Tzimas G, Wong F, et al. The percutaneous absorption of topically applied tretinoin and its effect on endogenous plasma tretinoin concentrations following single doses or long-term use. J Am Acad Dermatol. 1997;36:S37–46. doi: 10.1016/s0190-9622(97)70059-8. [DOI] [PubMed] [Google Scholar]
- Lever I, Kumar P, Marks R. Topical retinoic acid for treatment of solar damage. Br J Dermatol. 1990;122:91–8. doi: 10.1111/j.1365-2133.1990.tb08244.x. [DOI] [PubMed] [Google Scholar]
- Leyden JJ, Grove GL, Grove MJ, et al. Treatment of photodamaged facial skin with topical tretinoin. J Am Acad Dermatol. 1989;21:638–44. doi: 10.1016/s0190-9622(89)70231-0. [DOI] [PubMed] [Google Scholar]
- Leyden J. Adapalene in clincal practice. Cutis. 2001;68(4S):7–9. [PubMed] [Google Scholar]
- Lockman PR, Mumper RJ, Khan MA, et al. Nanoparticle technology for drug delivery across blood brain barrier. Drug Dev Ind Pharm. 2002;28:1–13. doi: 10.1081/ddc-120001481. [DOI] [PubMed] [Google Scholar]
- Lowe N, Gifford M, Tanghetti E, et al. Tarazotene 0.1% cream versus tretinoin 0.05% emollient cream in the treatment of photodamaged facial skin: a multicenter, double-blind, randomized, parallel-group study. J Cosmet Laser Ther. 2004;6:79–85. doi: 10.1080/14764170410032406. [DOI] [PubMed] [Google Scholar]
- Machtinger LA, Kaidbey K, Lim J, et al. Histological effects of tazarotene 0.1% cream vs. vehicle on photodamaged skin: a 6-month, multicentre, double-blind, randomized, vehicle-controlled study in patients with photodamaged facial skin. Br J Dermatol. 2004;151:1245–52. doi: 10.1111/j.1365-2133.2004.06186.x. [DOI] [PubMed] [Google Scholar]
- Maddin S, Lauharanta J, Agache P, et al. Isotretinoin improves the appearance of photodamaged skin: results of a 36-week, multicenter, double-blind, placebo-controlled trial. J Am Acad Dermatol. 2000;42:56–63. doi: 10.1016/s0190-9622(00)90009-4. [DOI] [PubMed] [Google Scholar]
- Massague J. TGF-β signal transduction. Ann Rev Biochem. 1998;67:753–91. doi: 10.1146/annurev.biochem.67.1.753. [DOI] [PubMed] [Google Scholar]
- Massague J. How cells read TGF-β signals. Nat Rev. 2000;1:169–78. doi: 10.1038/35043051. [DOI] [PubMed] [Google Scholar]
- Mordon S, Lagarde JM, Vienne MP, et al. Ultrasound imaging demonstration of the improvement of non-ablative laser remodeling by concomitant daily topical application of 0.05% retinaldehyde. J Cosmet Laser Ther. 2004;6:5–9. doi: 10.1080/14764170310016388. [DOI] [PubMed] [Google Scholar]
- Müller RH, Radtke S, Wissing SA. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 2002;54(S1):S131–55. doi: 10.1016/s0169-409x(02)00118-7. [DOI] [PubMed] [Google Scholar]
- Nagpal S, Athanikar J, Chandraratna RA. Separation of transactivation and AP1 antagonism functions of retinoic acid receptor alpha. J Biol Chem. 1995;270:923–7. doi: 10.1074/jbc.270.2.923. [DOI] [PubMed] [Google Scholar]
- Nyirady J, Bergfeld W, Ellis C, et al. Tretinoin cream 0.02% for the treatment of photodamaged facial skin: A review of 2 double-blind clinical trials. Cutis. 2001;68:135–42. [PubMed] [Google Scholar]
- Ochando N, LaGarde JM, Couval E, et al. Evaluation clinique et paraclinique des effets du rétinaldéhyde topique dans le photovieillissement cutané. Nouv Dermatol. 1994;13:525–35. [Google Scholar]
- Oda RM, Shimizu RW, Sabatine SC, et al. Effects of structural changes on retinoid cytotoxity in the CHO clonal assay. In vitro. Toxicol. 1996;9:173–81. [Google Scholar]
- Olsen EA, Katz I, Levine N, et al. Tretinoin emollient cream: a new therapy for photodamaged skin. J Am Acad Dermatol. 1992;26:215–24. doi: 10.1016/0190-9622(92)70030-j. [DOI] [PubMed] [Google Scholar]
- Olsen EA, Katz HI, Levine N, et al. Tretinoin emollient cream for photodamaged skin: Results of 48-week, multicenter, double-blind studies. J Am Acad Dermatol. 1997a;37:217–6. doi: 10.1016/s0190-9622(97)80128-4. [DOI] [PubMed] [Google Scholar]
- Olsen EA, Katz HI, Levine N, et al. Sustained improvement in photodamaged skin with reduced tretinoin emollient cream treatment regimen: Effect of once-weekly and three times-weekly applications. J Am Acad Dermatol. 1997b;37:227–30. doi: 10.1016/s0190-9622(97)80129-6. [DOI] [PubMed] [Google Scholar]
- Oppel T, Korting HC. Actinic keratosis: the key event in the evolution from photoaged skin to squamous cell carcinoma. Therapy based on pathogenetic and clinical aspects. Skin Pharmacol Physiol. 2004;17:67–76. doi: 10.1159/000076016. [DOI] [PubMed] [Google Scholar]
- Orfanos CE, Zouboulis CC, Almond-Roesler B, et al. Current use and future potential role of retinoids in dermatology. Drugs. 1997;53:358–88. doi: 10.2165/00003495-199753030-00003. [DOI] [PubMed] [Google Scholar]
- Patravale VB, Shah K. Solid lipid nanoparticles based tretinoin gels using novel solvent emulsification-diffusion approach (abstract P-39); Proceedings of the International Symposium on Advances in Technology and Business Potential of Novel Drug Delivery Systems; 2005. p. 55. [Google Scholar]
- Petkovich M, Brand MJ, Krust A, et al. A human retinoic acid receptor which belongs to the family of nuclear receptors. Nature. 1987;330:444–50. doi: 10.1038/330444a0. [DOI] [PubMed] [Google Scholar]
- Phillips TJ, Gottlieb AB, Leyden JJ, et al. Efficacy of 0.1% tazarotene cream for the treatment of photodamage. A 12-month multicenter, randomized trial. Arch Dermatol. 2002;138:1486–93. doi: 10.1001/archderm.138.11.1486. [DOI] [PubMed] [Google Scholar]
- Piérard-Franchimont C, Castelli D, Cromphaut IV. Tensile properties and contours of aging facial skin, A controlled double-blind comparative study of the effects of retinol, melibose-lactose and their association. Skin Res Technol. 1998;4:237–43. doi: 10.1111/j.1600-0846.1998.tb00116.x. [DOI] [PubMed] [Google Scholar]
- Ramos-E-Silva M, Hexsel DM, Rutowitsch MS, et al. Hydroxy acids and retinoids in cosmetics. Clin Dermatol. 2001;19:460–6. doi: 10.1016/s0738-081x(01)00189-4. [DOI] [PubMed] [Google Scholar]
- Rheinwald JG, Hahn WC, Ramsey MR, et al. A two-stage p16 (INK4A) and p53–dependent keratinocyte senescence mechanism that limits replicative potential independent telomere status. Mol Cell Bio. 2002;22:5157–72. doi: 10.1128/MCB.22.14.5157-5172.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rittié L, Fisher G. UV-light-induced signal cascades and skin aging. Aging Res Rev. 2002;1:705–20. doi: 10.1016/s1568-1637(02)00024-7. [DOI] [PubMed] [Google Scholar]
- Roeder A, Schaller M, Schäfer-Korting M, et al. Tazarotene: Therapeutic strategies in the treatment of psoriasis, acne and photoaging. Skin Pharmacol Physiol. 2004;17:111–18. doi: 10.1159/000077236. [DOI] [PubMed] [Google Scholar]
- Rolewski SL. Clinical review: Topical retinoids. Dermatol Nurs. 2003;15:447–65. [PubMed] [Google Scholar]
- Roos TC, Jugert FK, Merk HF, et al. Retinoid metabolism in the skin. Pharmacol Rev. 1998;50:315–33. [PubMed] [Google Scholar]
- Ruzicka T, Larsen FG, Galewicz D, et al. Oral alitretinoin (9-cis-retinoic acid) therapy for chronic hand dermatitis in patients refractory to standard therapy: results of a randomized, double-blind, placebo-controlled, multicenter trial. Arch Dermatol. 2004;140:1453–9. doi: 10.1001/archderm.140.12.1453. [DOI] [PubMed] [Google Scholar]
- Sass Jo, Didierjean I, Carraux P, et al. Metabolism of topical retinaldehyde and retinol by mouse skin in vivo: predominant formation of retinyl esters and identification of 14-hydroxy-4, 14-retro-retinol. Exp Dermatol. 1996;5:267–71. doi: 10.1111/j.1600-0625.1996.tb00128.x. [DOI] [PubMed] [Google Scholar]
- Saurat JH, Didierjean L, Masgrau E, et al. Topical retinaldehyde on human skin: biologic effects and tolerance. J Invest Dermatol. 1994;103:770–4. doi: 10.1111/1523-1747.ep12412861. [DOI] [PubMed] [Google Scholar]
- Sefton J, Kligman AM, Kopper SC, et al. Photodamage pilot study: A double-blind, vehicle-controlled study to assess the efficacy and safety of tazarotene 0.1% gel. J Am Acad Dermatol. 2000;43:656–63. doi: 10.1067/mjd.2000.107938. [DOI] [PubMed] [Google Scholar]
- Seité S, Bredoux C, Compan D, et al. Histological evaluation of a topically applied Retinol-Vitamin C combination. Skin Pharmacol Physiol. 2005;18:81–7. doi: 10.1159/000083708. [DOI] [PubMed] [Google Scholar]
- Sendagorta E, Lesiewicz J, Armstrong RB. Topical isotretinoin for photodamaged skin. J Am Acad Dermatol. 1992;27(Suppl):S15–8. doi: 10.1016/s0190-9622(08)80254-x. [DOI] [PubMed] [Google Scholar]
- Shukuwa T, Kligman AM, Stoudemayer T, et al. The effect of Short-term (1 month) topical tretinoin on photodamaged forearm skin. J Dermatol Treat. 1993;4:139–43. [Google Scholar]
- Sorg O, Didierjean L, Saurat J. Metabolism of topical retinaldehyde. Dermatology. 1999;199(S 1):13–7. doi: 10.1159/000051372. [DOI] [PubMed] [Google Scholar]
- Törmä H, Vahlquist A. Vitamin A esterification in human epidermis: A relation to keratinocytes differentiation. J Invest Dermatol. 1990;94:132–8. doi: 10.1111/1523-1747.ep12873990. [DOI] [PubMed] [Google Scholar]
- Torras H. Retinoids in aging. Clin Dermatol. 1996;74:207–15. doi: 10.1016/0738-081x(95)00156-a. [DOI] [PubMed] [Google Scholar]
- Varani J, Warner RL, Gharaee-Kermani M, et al. Vitamin A antagonizes decreased cell growth and elevated collagen-degrading matrix metalloproteinases and stimulates collagen accumulation in naturally aged human skin. J Invest Dermatol. 2000;114:480–6. doi: 10.1046/j.1523-1747.2000.00902.x. [DOI] [PubMed] [Google Scholar]
- Weinstein GD, Nigra TP, Pochi PE, et al. Topical tretinoin for treatment of photodamaged skin. Arch Dermatol. 1991;127:659–65. [PubMed] [Google Scholar]
- Weiss JS, Ellis CN, Headington JT, et al. Topical tretinoin improves photoaged skin: a double-blind vehicle-controlled study. JAMA. 1988;259:527–32. [PubMed] [Google Scholar]
- Wissing SA, Muller RH. Solid lipid nanoparticles as carrier for sunscreens: in vitro release and in vivo skin penetration. J Control Rel. 2002;81:225–33. doi: 10.1016/s0168-3659(02)00056-1. [DOI] [PubMed] [Google Scholar]
- Xiao JH, Durand B, Chambon P, et al. Endogenous retinoic acid receptor-retinoid X receptor heterodimers are the major functional forms regulating retinoid-responsive elements in adult human keratinocytes. J Biol Chem. 1995;270:3001–11. doi: 10.1074/jbc.270.7.3001. [DOI] [PubMed] [Google Scholar]
- Yaar M, Gilchrest BA. Skin Aging: Postulated mechanisms and consequent changes in structure and function. Clin Ger Med. 2001;17:617–30. doi: 10.1016/s0749-0690(05)70089-6. [DOI] [PubMed] [Google Scholar]
- Yamaguchi Y, Nagasawa T, Nakamura N, et al. Successful treatment of photo-damaged skin of nano-scale atRA particles using a novel transdermal delivery. J Control Rel. 2005;104:29–40. doi: 10.1016/j.jconrel.2004.11.036. [DOI] [PubMed] [Google Scholar]