An Integrated Network of Androgen Receptor, Polycomb, and TMPRSS2-ERG Gene Fusions in Prostate Cancer Progression (original) (raw)
. Author manuscript; available in PMC: 2011 May 18.
Published in final edited form as: Cancer Cell. 2010 May 18;17(5):443–454. doi: 10.1016/j.ccr.2010.03.018
SUMMARY
While chromosomal rearrangements fusing the androgen-regulated gene TMPRSS2 to the oncogenic ETS transcription factor ERG occur in approximately 50% of prostate cancers, how the fusion products regulate prostate cancer remains unclear. Using chromatin immunoprecipitation coupled with massively parallel sequencing (ChIP-Seq), we found that ERG disrupts androgen receptor (AR) signaling by inhibiting AR expression, binding to and inhibiting AR activity at gene-specific loci, and inducing repressive epigenetic programs via direct activation of the H3K27 methyltransferase EZH2, a Polycomb group protein. These findings provide a working model in which TMPRSS2-ERG plays a critical role in cancer progression by disrupting lineage-specific differentiation of the prostate and potentiating the EZH2-mediated de-differentiation program.
INTRODUCTION
Prostate cancer is the most common non-skin cancer and a leading cause of cancer-related deaths in American men. We and others have previously characterized chromosomal rearrangements fusing the androgen-regulated gene TMPRSS2 to ETS transcription factors such as ERG and ETV1 in a majority of prostate cancers (Perner et al., 2006; Soller et al., 2006; Tomlins et al., 2005; Yoshimoto et al., 2006). Among these, fusions of TMPRSS2 to the oncogenic ETS transcription factor ERG occur most frequently, accounting for 40–80% of prostate cancers (Clark et al., 2007; Hermans et al., 2006). ERG has also been implicated in recurrent gene fusions found in Ewing’s sarcoma and acute myeloid leukemia (Hsu et al., 2004; Oikawa and Yamada, 2003). Knock-down of ERG in VCaP prostate cancer cells inhibits cell growth, cell invasion, and xenograft tumor growth (Tomlins et al., 2008; Wang et al., 2008). Over-expression of ERG increases cell invasion in vitro (Tomlins et al., 2008; Wang et al., 2008), and induces prostate cancer precursor-like lesions in mice (Klezovitch et al., 2008; Tomlins et al., 2008). Furthermore, ERG collaborates with genetic lesions of the PI3K pathway to promote prostate cancer progression in mouse models (Carver et al., 2009; King et al., 2009; Zong et al., 2009). While it is clear that ERG may possess oncogenic properties, it has been much less clear as to how ERG promotes prostate cancer progression. Given the prevalence and recurrence of TMPRSS2-ERG in prostate cancer we hypothesized that it plays an essential role in prostate tumorigenesis.
We have earlier demonstrated that ERG induces metalloproteinase and plasminogen activator pathway genes such as MMP3, PLAT, and PLAU (Tomlins et al., 2008). ERG is a member of the ETS family transcription factors that specifically bind to genomic regions containing the core GGA(A/T) motifs (Nye et al., 1992). Interestingly, ETS motifs were found enriched in the Androgen Receptor (AR) binding sites determined by ChIP-on-chip analysis and ETS1, another member of the ETS family transcription factors, was shown to physically interact with AR (Massie et al., 2007). Moreover, recent studies revealed that ERG represses the expression of the prostate-specific antigen PSA (Sun et al., 2008), while ETV1 induces PSA expression (Shin et al., 2009). The molecular crosstalk between ERG and AR, however, remains largely uncharacterized.
AR belongs to a family of nuclear transcription factors that mediate the action of steroid hormones. Cytoplasmic AR, when bound by androgen, translocates to the nucleus and binds to the 15-bp palindromic Androgen Response Elements (ARE) on target genes (Heemers and Tindall, 2007). AR is paramount for the lineage-specific differentiation of the prostate, inducing the expression of prostate-specific genes such as PSA and TMPRSS2, and maintaining the differentiated prostate epithelial phenotype (Wright et al., 2003). Cellular de-differentiation, by contrast, is a hallmark of malignant transformation. Previous studies have shown that a majority of metastatic prostate tumors have up-regulated expression of the Polycomb group (PcG) protein EZH2, which plays critical roles in maintaining the undifferentiated state of embryonic stem (ES) cells via catalyzing H3K27 tri-methylation (Lee et al., 2006; Varambally et al., 2002). EZH2 over-expression in advanced prostate cancer leads to epigenetic silencing of developmental regulators and tumor suppressor genes, subverting cancer cells to a stem cell-like epigenetic state (Yu et al., 2007c).
Here we employed chromatin immunoprecipitation coupled with massively parallel sequencing (ChIP-Seq) (Barski et al., 2007; Johnson et al., 2007; Mikkelsen et al., 2007) to systematically map the genomic landscape of 4 transcription factors and 8 histone marks across multiple prostate cancer cell lines as well as tissues (summarized in Table S1). Integrative genomic analysis was undertaken to delineate the interaction between TMPRSS2-ERG, AR and EZH2. In addition, we examined the mechanisms of TMPRSS2-ERG in prostate tumorigenesis in the context of AR-induced prostatic differentiation and EZH2-mediated cellular dedifferentiation.
RESULTS
Genomic landscape of AR in prostate cancer
To determine AR binding sites across the human genome, we performed ChIP-Seq analysis of the LNCaP prostate cancer cells treated with either vehicle or saturating amounts of synthetic androgen R1881 as previously reported (Wang et al., 2007). To evaluate the reproducibility of the ChIP-Seq assay, we compared the distribution of sequencing reads mapped in every 25bp bin across the entire genome and observed substantial overlap between technical as well as biological replicates (Figure S1A). Using the HPeak program (http://www.sph.umich.edu/csg/qin/HPeak/), we identified enriched binding peaks from mappable sequencing reads. In LNCaP cells our study revealed 37193 AR binding peaks, which include 82% of the AR-bound sites previously reported by ChIP-on-chip assay (Wang et al., 2009). Importantly, known AR binding sites, such as the enhancer upstream to the FKBP5 gene, showed marked ChIP-Seq enrichment comprised of thousands of sequencing reads. By sorting the binding peaks by height, the top AR-bound genes included FKBP5, C6ORF81, TACC2, CUTL2 and SLC43A1, all of which showed “prostate-specific” expression based on the International Genomics Consortium’s expO microarray dataset profiling 28 tumor types (www.oncomine.com) (Figure S1B). These genes also ranked among the top 10 AR-bound genes in the VCaP prostate cancer cells that harbor the TMPRSS2-ERG gene fusions (Tomlins et al., 2005). Approximately 61% of the AR binding sites in the VCaP cells physically overlapped with those in LNCaP, suggesting shared as well as cell-type specific AR recruitment (Figure 1A). The shared AR binding targets were further confirmed by the inclusion of previously reported androgen-sensitive 5′ partners of gene fusions in prostate cancer such as TMPRSS2, SLC45A3 and C15ORF21 (Tomlins et al., 2007) (Figure S1C). By contrast, no AR binding was detected on the androgen-insensitive 5′ fusion partner gene HNPRA2B1 that is constitutively active, supported by the enrichment of two markers of active transcription, histone H3 lysine 4 trimethylation (H3K4me3) and RNA Polymerase II (RNA PolII), on its promoter. AR recruitment to these genetic loci was further confirmed by ChIP-PCR using gene-specific primers. Analysis of cell type-specific AR binding revealed much higher false negative rate of ChIP-Seq in VCaP than LNCaP, partially accounting for the lesser amount of AR binding sites detected in the former (Figure S1D). We next examined the presence of consensus sequence motifs in the AR binding sites. Out of all 508 pre-defined motifs of vertebrate transcription factors (www.genomatix.de), the 15-bp canonical ARE, as expected, was the most enriched. In addition, the occurrence of the ARE motif was positively correlated with the height of ChIP-Seq binding peaks (r = 0.87, P < 0.001) and some binding peaks contained multiple AREs (Figure S1E). By categorizing all AR binding sites based on whether they contained a full or half or no ARE motifs, we found that the binding sites containing full ARE motifs had significantly (p<0.001 by t-test) higher enrichment peaks than those with half ARE motifs, which had higher peaks than those without any ARE motifs, supporting the role of ARE in recruiting AR. Furthermore, de novo motif search using the MEME program (Bailey and Elkan, 1994) identified a refined ARE motif that was markedly similar to the canonical ARE (Figure 1B). Surprisingly, the ETS family motifs were the 2nd most enriched motifs suggesting potential colocalization of ERG with AR (Table S2). Approximately 40% of all AR binding sites contained at least one ARE motif and about 29% contained an ETS motif, both being significantly (p<0.001 by Fisher Exact Test) more than their respective occurrence in control sequences (Table S3).
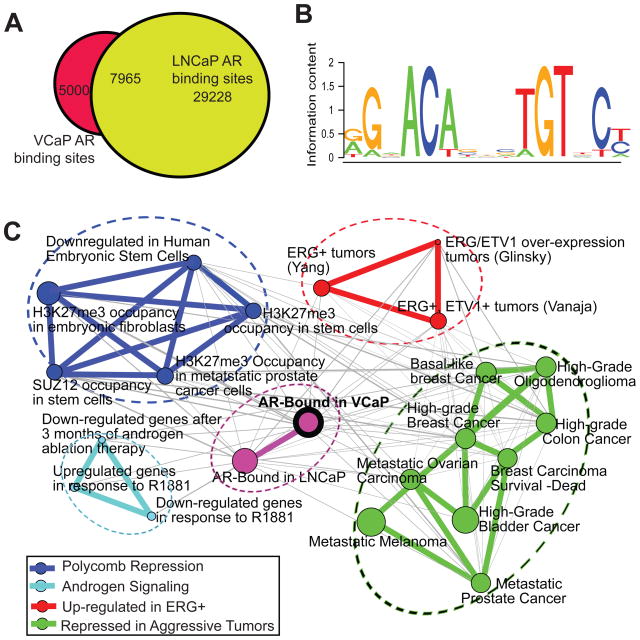
Figure 1. Genome-wide location analysis of AR in prostate cancer.
(A) Venn diagram showing overlap of the ChIP-Seq AR binding sites in the LNCaP and VCaP prostate cancer cells.
(B) The consensus motif identified in the AR binding sites. De novo motif search was performed using the MEME program (Bailey and Elkan, 1994).
(C) An enrichment network linking AR, TMPRSS2-ERG pathways and the polycomb-mediated de-differentiation program. AR-bound genes (purple node with black ring) were derived by ChIP-Seq in the VCaP cells and analyzed by the Molecular Concept Map in OncomineTM. Each node represents one molecular concept or gene set with node size proportional to the number of genes. Each edge represents a statistically significant (P < 1 × 10−10) overlap of genes in the two linked nodes. Molecular concepts were grouped into 5 major clusters indicated by oval rings of distinct color. See also Figure S1 and Tables S1-4.
To obtain a functional taxonomy of the AR-bound genes, we performed Molecular Concept Map (MCM) analysis for enrichment of the AR-bound genes in thousands of pre-defined molecular concepts/gene sets in the Oncomine database (www.oncomine.com). Out of approximately 20,000 molecular concepts, a total of 1462 (7%) showed significant (P < 0.001) enrichment (Table S4). Not surprisingly, AR-bound genes in the VCaP cells significantly overlapped with those in the LNCaP cells (P < 1.0 × 10−100) and they both related to genes that are regulated by androgen in vitro or in vivo (P < 1.0 × 10−10) (Figure 1C). In addition, there was significantly enriched AR binding on “genes under-expressed in metastatic or high-grade tumors” (P < 4.0 × 10−15), confirming reduced androgen signaling in aggressive prostate tumors. Importantly, AR-bound genes were significantly enriched in a number of ERG-regulated gene sets (P < 4.0 × 10−13), indicating an interesting link between ERG regulation and androgen signaling. Notably, a group of most significantly enriched (P < 1.0 × 10−20) concepts related to stem cell gene signatures, including target genes of Polycomb group proteins and H3K27me3 in embryonic stem cells and metastatic prostate cancers (Boyer et al., 2006; Lee et al., 2006; Yu et al., 2007c), further connecting the AR and ERG pathways to PcG-mediated epigenetic silencing and cellular de-differentiation.
ERG occupancy of AR target genes in prostate cancer
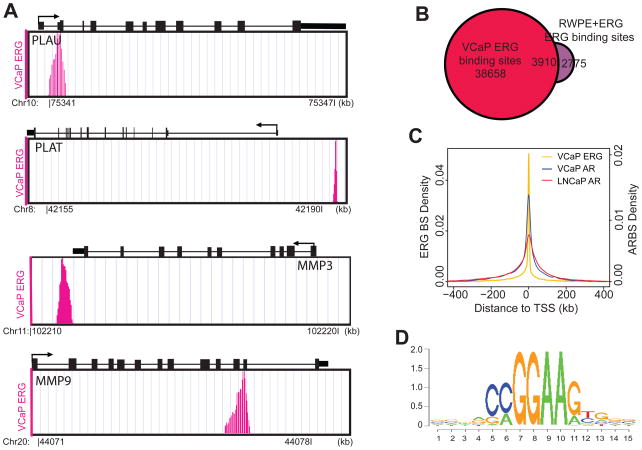
To characterize the potential link between the transcriptional pathways of AR, ERG, and epigenetic modifications, we performed ChIP-Seq analysis of ERG and a number of key histone marks in prostate cancer (Table S1). Approximately 42,000 ERG binding sites were identified in the VCaP cells including known ERG target genes such as PLAU, PLAT, MMP3 and MMP9 (Tomlins et al., 2008) (Figure 2A). By contrast, only 608 binding sites were detected in the ERG-negative LNCaP cells. Interestingly, ChIP-Seq analysis of the RWPE benign prostate epithelial cells with stable ERG overexpression (RWPE+ERG) identified 6,685 ERG binding sites, 58% of which overlapped with those detected in the VCaP cells (Figure 2B). Unlike AR, which was mostly enriched at distal enhancers, ERG primarily bound to the promoter regions of target genes (Figure 2C & S2). Not surprisingly, the ETS family motifs were the most significantly enriched motifs in the ERG binding sites (Table S2) and MEME analysis discovered a de novo ERG motif that was strikingly similar to the consensus ETS motifs (Figure 2D).
Figure 2. ChIP-Seq analyses of ERG binding sites in prostate cancer.
(A) ChIP-Seq analysis of the VCaP cells detected ERG binding to previously reported target genes (Tomlins et al., 2008).
(B) Venn diagram showing overlap between the ERG binding sites identified in the VCaP cells and the RWPE+ERG cells. The RWPE cells were infected with lentivirus overexpressing ERG or lacZ and selected for stable clones. ChIP-Seq of ERG was performed in the stable RWPE+ERG cells using the RWPE+lacZ as control.
(C) Distribution of AR or ERG binding sites relative to the transcriptional start sites (TSS) of all RefSeq genes. The Y-axis on the left and right represents the density of ERG (ERG BS) and AR binding sites (ARBS), respectively. See also Figure S2.
(D) The consensus sequence motif identified in the ERG binding sites by the MEME program.
To test the hypothesis that AR and ERG co-occupy target genes, we analyzed the overlap in their binding sites in every 25bp bin across the entire genome. While 61% of the AR-bound regions in the VCaP cells overlapped with those in LNCaP, approximately 44% of these sites, surprisingly, also recruited ERG (Figure 3A). This overlap between AR and ERG binding sites is remarkable, as AR and ERG each bound to only 0.1% and 0.7% sequences of the entire genome, respectively. To evaluate this overlap statistically, we compared AR and ERG binding sites with those of NRSF, a neural specific transcription factor that has not been associated with either AR or ERG (Johnson et al., 2007). Importantly, there was less than 2% of overlap. The overlap between the AR and ERG binding sites was significantly more than their overlap with NRSF (P < 0.0001 by Fisher’s exact test). Further, we compared the binding sites of AR and ERG with other transcription factors and epigenetic marks analyzed by ChIP-Seq in both LNCaP and VCaP cells (Figure S3A & Table S5). As expected, we observed substantial overlap of the same factor across cell types. Remarkably, the overlap between AR and ERG in the VCaP cells was amongst the highest in all overlaps between different regulators, including those between AR and FoxA1, PolII, H3K4me1, H3K4me3, H3K9me3 and H3K27me3. The next strongest overlap was of AR with FoxA1and Ace-H3, both of which have been previously reported (Jia et al., 2008; Lupien et al., 2008).
Figure 3. ERG and AR co-occupancy of target genes in prostate cancer.
(A) Overlap between the binding sites of different regulators. Significance of overlap was measured relative to their respective overlap with the NRSF binding sites. *P<0.05, ** P<0.01, and ***P<0.001 by hypergeometric test.
(B) ERG binds to a majority of the AR-bound genes. AR or ERG ChIP-Seq binding sites were each assigned to the nearest gene on the genome to derive the list of bound genes.
(C) Overlap between the ERG-bound genes and the genes that are bound by both AR and ERG as determined by re-ChIP-Seq using an anti-ERG and an anti-AR antibody.
(D) Representative genes co-occupied by AR and ERG. On the Y-axes are the number of reads of AR (left, in blue) and ERG (right, in red) binding sites. Above the plot are the TSS and direction denoted by arrows, exons by black box, and introns by horizontal lines, respectively. The genomic coordinates are indicated below the plot.
(E) ChIP-PCR confirms AR and ERG co-occupancy on selected genes. The Y-axis on the left represents AR ChIP enrichment in VCaP cells treated with R1881 normalized to the ethanol (Ethl) treated cells. The Y-axis on the right represents ChIP enrichment (in log scale) by an anti-ERG antibody normalized to IgG. Error bars: n=3, mean ± SEM. The 3′ intronic region of the KIAA0066 gene was used as a negative control.
(F) AR and ERG co-occupancy in a prostate cancer tissue. A prostate tumor tissue that expresses both AR and TMPRSS2-ERG were analyzed by ChIP-Seq.
(G) Physical interaction between the AR and ERG proteins. VCaP cells were immunoprecipitated by various antibodies and immunoblotted for AR. To deplete DNA, VCaP cell lysates were pre-incubated with ethidium bromide for 30 min before immunoprecipitation. Representative experiment of 4 independent co-immunoprecipitation assessments is shown.
(H) Interaction of ERG with AR in vitro via the ETS domain. HaloTag-ERG protein and mutants were generated by in vitro transcription/translation in wheat germ extracts. Recombinant GST-AR was incubated with the in vitro translated protein products and glutathione beads used in pull down assays. The interaction between AR and various domains of ERG are summarized and indicated as + or -. See also Figure S3 and Table S5.
By assigning ChIP-Seq binding sites to their nearest genes, we found that ERG is in fact recruited to over 90% of the AR-bound genes (Figure 3B). This co-binding was further confirmed by re-ChIP-Seq experiment first using an antibody against ERG followed by an antibody against AR (Figure 3C). To further validate this, we randomly selected a set of 5 genes that bound both AR and ERG, including NDRG1, C60rf81, CUTL2, LOC400451, and ZBTB16 (Figure 3D & S3B). Importantly, using gene-specific primers, we confirmed AR and ERG co-occupancy on all selected genes by conventional ChIP-PCR assays (Figure 3E). In addition, the expression of these genes was found to be differentially regulated by both androgen stimulation and ERG RNA interference (Figure S3C).
To confirm the interconnected pathways of ERG and AR in vivo, we performed ChIP-Seq analysis of a human prostate tumor that expressed both ERG and AR, and identified 6,967 ERG and 12,036 AR binding sites, respectively. Not surprisingly, they both overlapped significantly with their respective binding sites identified in prostate cancer cell lines (P < 0.001 by Fisher’s exact test). Importantly, a comparison of the AR- and ERG-binding sites in the prostate tumor specimen revealed a remarkable overlap (44%) that is comparable to that (43.97%) observed in the VCaP cell line model (Figure 3F). MCM analysis of the 1,534 tissue ERG-bound genes revealed a core transcriptional regulatory circuitry composed of biological correlates with highly significant overlaps (P < 1.0 × 10−90); these include ERG-bound and AR-bound genes in tumors, ERG-bound and AR-bound genes in the VCaP cells, AR-bound genes in the LNCaP cells, and ERG-bound genes in the stable RWPE+ERG cells (Figure S3D).
Next, we determined if ERG and AR physically interact. Co-immunoprecipitation assays demonstrated an endogenous interaction between the AR and ERG proteins in cell line as well as prostate cancer tissues (Figure 3G & S3E). Using ethidium bromide pre-incubation to deplete DNA, we demonstrated that the interaction between AR and ERG was DNA independent. Further, in vitro protein-protein interaction assays revealed that AR directly bound to the ETS domain of the ERG protein (Figure 3H & S3F). This interaction was further pinpointed to the C-terminal half (ETS-3) of the ETS domain.
The TMPRSS2-ERG gene fusion product disrupts androgen signaling
Interestingly, one of the top genomic loci bound by ERG was the promoter of the AR gene as illustrated by ChIP-Seq and confirmed by ChIP-PCR (Figure 4A-B). By over-expressing ERG we observed significantly decreased AR transcript in multiple prostate cell lines, including the VCaP and LNCaP prostate cancer cells and the immortalized RWPE prostate epithelial cells (Figure 4C). Concordantly, knockdown of ERG in VCaP cells led to AR upregulation, further supporting an inhibitory role of ERG on the AR gene (Figure S4A-B). In addition, we observed a marked decrease of AR protein following ERG overexpression in the LNCaP and VCaP prostate cancer cells as well as in the 22RV1 cells that are free of ETS family gene fusions, whereas AR protein was expectedly not detected in the RWPE cells (Figure 4D & S4C). Consistent with this, RNA interference of ERG in the hormone-starved VCaP cells greatly increased AR protein (Figure 4E). Adding androgen to these cells dramatically increased ERG expression, as expected. AR levels, however, were decreased, further demonstrating a negative regulation. Moreover, we showed that ERG overexpression greatly reduced AR promoter reporter activity in LNCaP cells (Figure S4D). In addition, AR and ERG expression were significantly anti-correlated (r = −0.35, P = 0.001) in vivo in localized prostate tumors (Figure S4E).
Figure 4. ERG negatively regulates the AR gene.
(A) ChIP-Seq showing AR and ERG co-binding to the regulatory regions of the AR gene. The Y-axes are as denoted as in Figure 3D.
(B) ChIP-PCR confirms ERG binding to the AR promoter.
(C) Ectopic ERG overexpression represses the AR transcript. VCaP and LNCaP prostate cancer cells and RWPE benign prostate epithelial cells were infected by LacZ or ERG adenovirus. QRT-PCR was used to assay the ERG and AR transcript normalized to GAPDH. Error bars: n=3, mean ± SEM.
(D) ERG represses the AR protein. VCaP (in the presence or absence of androgen R1881), LNCaP, RWPE and 22RV1 cells were infected with ERG for 48hr before immunoblotting.
(E) ERG knockdown de-represses the AR protein. RNA interference of ERG was done in VCaP cells in the presence or absence of androgen. See also Figure S4.
Besides directly regulating AR itself, ERG was recruited to a majority of AR target genes at gene-specific loci, for instance KLK3, KLK2, SLC45A3, and FKBP5 (Figure 5A). We thus analyzed AR target gene expression in 67 localized prostate tumors that were classified into either ERG-positive (ERG+) or ETS-negative (ETS−) depending on the status of ETS family gene fusions. ETS- tumors were used as a negative control for comparison so as to preclude any effects from non-ERG ETS family gene fusions. Importantly, GSEA analysis demonstrated significant enrichment (P < 0.001) of “genes induced by androgen” in the gene set that is “repressed in ERG+ relative to ETS- prostate tumors”, thus linking androgen induction with ERG repression (Figure 5B). For example, KLK3, TMPRSS2, KLK2 and KLK4 were all expressed at significantly lower levels in the ERG+ than the ETS- prostate tumors (Figure S5A). This is an interesting finding that is consistent with our overall hypothesis that ERG functions to repress androgen regulation of a lineage-specific differentiation program. To further confirm this, we overexpressed ERG in LNCaP cells and carried out qRT-PCR analysis of a set of known androgen-induced genes, many of which are markers of prostate epithelial differentiation. Importantly, our results showed marked inhibition of these genes, while ERG expectedly activated other targets such as PLAT and PLAU (Tomlins et al., 2008) (Figure 5C). Similar suppression of androgen signaling by ERG was also observed in the 22RV1 cells that are negative for ETS family gene fusions (Figure S5B). We next investigated whether RNA interference of ERG may rescue this inhibition using the TMPRSS2-ERG-positive VCaP cells. We first treated hormone-starved VCaP cells with androgen and confirmed androgen-induced expression of AR target genes. Remarkably, the expression of these genes can be further increased by RNA interference of ERG, confirming endogenous ERG suppression of androgen response in the VCaP cells that can be de-repressed by removing ERG (Figure 5D).
Figure 5. ERG attenuates AR transcriptional activity.
(A) ChIP-Seq showing AR and ERG co-binding to the regulatory regions of representative AR target genes. The Y-axes are as denoted in Figure 3D.
(B) Androgen-induced genes are significantly enriched for repression by ERG. Androgen-induced genes were obtained from microarray analysis of time-course androgen treatment of LNCaP cells. ERG-repressed genes are down-regulated in the ERG+ (n=35) relative to the ETS- (n=31) prostate tumors based on cancer gene expression microarray profiling.
(C) ERG overexpression represses AR target genes. LNCaP cells were infected with ERG or lacZ for 48 hrs prior to qRT-PCR analysis. ERG, PLAT and PLAU were used as positive controls for ERG overexpression.
(D) ERG knockdown de-represses androgen–induced genes. VCaP cells were hormone starved for 2 days and treated with either ethanol, synthetic androgen R1881, or R1881 with concurrent RNA interference targeting ERG for 48hrs before qRT-PCR analysis. The level of ERG knockdown is shown in Figure S4B.
(E) Suppression of the KLK3 and TMPRSS2 promoters by ERG. LNCaP cells were co-transfected with various promoter reporter constructs along with pRL-TK (the internal control) at 24hrs post-infection of ERG or LacZ, incubated for another 24hrs and then monitored for luciferase activity. Error bars: n=3, mean ± SEM. See also Figure S5.
ChIP-PCR analysis of LNCaP cells following ERG titration revealed gradually increased ERG binding, but decreased AR binding, on these gene-specific genomic loci, supporting direct regulation by ERG independent of AR (Figure S5C-D). Consistent with this, ChIP-Seq analysis of VCaP cells following RNA interference of ERG revealed a 24% decrease in the number of ERG binding sites and a 26% increase in AR binding sites. Further, ERG overexpression in cells with either AR knockdown or AR overexpression continued to inhibit AR target genes (Figure S5E–F). Furthermore, luciferase reporter assays demonstrated substantially reduced (~5 fold) promoter activities of the KLK3 and TMPRSS2 gene by ERG overexpression (Figure 5E). Therefore, ERG disrupts androgen signaling through multiple mechanisms including by inhibiting AR expression, binding to and repressing AR downstream targets at gene-specific loci.
We next examined the oncogenic role of TMPRSS2-ERG gene fusions in the context of androgen signaling. Interestingly, ectopic overexpression of ERG in VCaP and LNCaP prostate cancer cells remarkably increased cell invasion in the presence or absence of androgen, independent of AR (Figure 6A-B). Similarly, ERG overexpression dramatically induced cell proliferation, independent of androgen as well as AR protein expression (Figure 6C-E). We therefore tested if ERG is able to drive androgen-independent prostate cancer cell growth. Stable clones of VCaP cells expressing ERG (VCaP+ERG) or the GUS control gene (VCaP+GUS) were selected and assayed for cell proliferation. Remarkably, we observed that the VCaP+ERG cells grow significantly faster than the VCaP+GUS cells (Figure 6F). In addition, the VCaP+ERG cells were able to continuously proliferate in the absence of androgen, while the VCaP+GUS cells failed to grow (Figure 6G). Therefore, by inhibiting AR-mediated pro-differentiation and regulating AR-independent oncogenesis TMPRSS2-ERG may provide a potential mechanism for androgen resistance.
Figure 6. ERG regulates the neoplastic properties of prostate cancer cells independent of androgen signaling.
(A) Ectopic ERG overexpression induces prostate cancer cell invasion in the absence of androgen. The VCaP and LNCaP prostate cancer were hormone-starved for 1 day, infected with ERG or control for another 2 days in the absence of androgen. Error bars: n=3, mean ± SEM.
(B) Ectopic ERG overexpression induces prostate cancer cell invasion independent of AR. VCaP and LNCaP cells were subjected to RNA interference against AR or control for 1 day before adenovirus infection of ERG or LacZ. Error bars: n=3, mean ± SEM.
(C) Ectopic ERG overexpression induces VCaP cell growth independent of androgen. VCaP cells were hormone-deprived for 48 hrs before infection with LacZ or ERG adenovirus in the presence or absence of androgen. Error bars: n=3, mean ± SEM.
(D–E) Ectopic ERG overexpression induces prostate cancer cell growth independent of AR expression. VCaP and LNCaP cells were subjected to RNA interference targeting AR or control for 1 day before adenovirus infection by ERG or LacZ. Cell proliferation was assayed at 48hrs or 96hrs after infection.
(F) Ectopic ERG overexpression in prostate cancer cells increases cell growth. VCaP cells were infected with lentivirus expressing ERG or GUS (control). Stable clones expressing ERG (VCaP+ERG) or control (VCaP+GUS) were plated equally, and assayed for cell proliferation in regular medium.
(G) Ectopic ERG overexpression confers cell growth in the absence of AR signaling. Equal numbers of stable VCaP+ERG and control cells were hormone-deprived for 48 hrs and assayed for cell proliferation in hormone-deprived medium.
TMPRSS2-ERG induces EZH2-mediated epigenetic silencing
By MCM analysis we have earlier shown a link of ERG and androgen signaling with Polycomb-mediated H3K27 methylation, which has been shown to prevent the differentiation of embryonic stem cells and contribute to tumor cell de-differentiation (Boyer et al., 2006; Lee et al., 2006; Yu et al., 2007c) (Figure 1C). We thus hypothesized that, besides inhibiting AR-mediated pro-differentiation, ERG may potentiate pathways involving H3K27me3 to control cell de-differentiation. Indeed, while AR and H3K27me3 binding sites rarely overlap (<2%) in LNCaP, there was substantially more overlap (13%) in VCaP, probably due to the recruitment by ERG (Figure S6A-B). To test this, we investigated ERG regulation of the H3K27 methyltransferase EZH2. Interestingly, ChIP-Seq analysis revealed ERG binding to the EZH2 promoter, which was confirmed by ChIP-PCR (Figure 7A-B). In addition, RNA interference of ERG greatly decreased EZH2 expression (Figure 7C). Concordant with this, ERG overexpression in LNCaP and RWPE cells markedly induced EZH2 (Figure 7D). Further, EZH2 and ERG expression were positively correlated (r = 0.23, p = 0.043) in clinically localized prostate tumors. EZH2 expression was significantly higher (P < 0.001) in the ERG+ (n=35) than the ETS- (n=31) tumors, thus supporting a model in which ERG activates EZH2 (Figure 7E).
Figure 7. ERG induces EZH2–mediated epigenetic silencing.
(A–B) ChIP-Seq and ChIP-PCR shows ERG binding to the EZH2 promoter in VCaP cells.
(C) ERG knockdown in VCaP cells decreases EZH2 protein.
(D) ERG overexpression increases EZH2 protein. LNCaP and RWPE cells were infected with ERG or LacZ adenovirus for 48hrs before immunoblot analysis.
(E) EZH2 is expressed at significantly (P<0.001) higher levels in the ERG+ (n=35) than the ETS- (n=31) prostate tumors.
(F) ERG overexpression in the LNCaP cells activates EZH2 and represses known EZH2 target genes. LNCaP cells were infected with LacZ or ERG adenovirus for 48 hrs and analyzed by qRT-PCR. Error bars: n=3, mean ± SEM. See also Figure S6.
(G) Polycomb-related signatures and androgen-induced genes predict the status of TMPRSS2-ERG gene fusion in localized prostate tumors. The target genes of PcG proteins in embryonic stem cells were derived from a previous study (Ben-Porath et al., 2008), while the control gene signatures were taken from the ChIP-Seq experiments. Androgen-induced or–repressed genes were from microarray profiling of the LNCaP cells following androgen treatment. A random signature not related to ERG (by removing ERG-regulated genes) was used as an absolute negative control (rand_nonERG). The Polycomb signatures and the androgen-induced genes have prediction scores significantly (P<0.001) better than the other gene sets.
(H) A model of TMPRSS2-ERG in prostate cancer by disrupting prostate-specific differentiation and potentiating a stem cell-like de-differentiation program. (i) androgen signaling leads to normal prostate differentiation. (ii) formation of the TMPRSS2-ERG somatic mutation. (iii) inhibition of AR expression and direct interaction with AR by the TMPRSS2-ERG fusion product. (iv) ERG binding to AR target gene loci for negative regulation. (v,viii) activation of epigenetic silencing, stem cell like state and oncogenesis. (vi) induction of EZH2. (vii) induction of H3K27 marks and epigenetic silencing of pro-differentiation genes.
We next investigated if ERG regulates EZH2-mediated epigenetic silencing. ChIP-Seq analysis demonstrated ERG binding to a number of previously reported EZH2 target genes such as ADRB2, CDH1, DAB2IP, SNCA and SOCS (Cao et al., 2008; Chen et al., 2005; Yu et al., 2007c) (Figure S6B). Remarkably, while ERG activated EZH2 expression, it strongly repressed the expression of EZH2 target genes, thus supporting ERG activation of EZH2-mediated epigenetic silencing in prostate cancer (Figure 7F & S6C). In addition, silencing of EZH2 restored the expression of EZH2 target genes such as DAB2IP, loss of which was recently shown leading to epithelial-to-mesenchymal transition and cellular mal-differentiation (Figure S6D) (Min et al.; Xie et al.). To further test a genome-wide association between ERG and EZH2-mediated de-differentiation pathways, we investigated whether the expression pattern of PcG target genes are able to predict the status of TMPRSS2-ERG gene fusions in prostate tumors. We first derived previously reported Polycomb gene signatures associated with ES cells and poorly differentiated tumors, including target genes of Polycomb repressive complex 2 (PRC2), of PcG proteins SUZ12 and EED, and of H3K27me3 (Ben-Porath et al., 2008). We then analyzed the power of these gene signatures in predicting the status of TMPRSS2-ERG gene fusions in clinically localized prostate tumors. Importantly, the expression pattern of all PcG-related gene signatures accurately predicted the ERG status, whereas irrelevant control gene sets, including the target genes of histone H3, PolII, H3K36me3 and H3K4me3, were significantly (P < 0.001) less predictive (Figure 7G). Notably, androgen-induced genes (presumably repressed by ERG) also successfully predicted ERG status in prostate tumors. Therefore, ERG may have more global effects by disrupting androgen-mediated prostatic differentiation and inducing EZH2-mediated cellular de-differentiation (Figure 7H).
DISCUSSION
In the present study, we systematically mapped the genomic landscape of AR, ERG, FoxA1 and RNA PolII, along with 8 critical histone marks in multiple prostate cancer cell lines as well as in one prostate tumor specimen. These studies not only reveal important biological findings regarding the mechanisms of TMPRSS2-ERG gene fusions in prostate cancer but they provide a compendium of 57 genome-wide ChIP-Seq experiments and a large set of paired microarray expression profiling data that will be useful for the investigation of biological mechanisms of cancer and steroid hormone receptor signaling.
By analyzing these genome-wide maps we provide a working model of how TMPRSS2-ERG gene fusions regulate prostate cancer progression. In the context of an androgen-regulated gene fusion such as TMPRSS2-ERG, this fusion product can attenuate androgen signaling by multiple, cooperative mechanisms including direct inhibition of AR expression and attenuation of AR signaling at gene-specific loci (Figure 7H). Furthermore, our study reveals an additional pathway of ERG in perturbing cell differentiation through the Polycomb group proteins. Enrichment of H3K27me3-marked genes silenced in ES cells and aggressive tumors was first apparent by MCM analysis of AR-occupied genes in prostate cancer, linking both AR and ERG to repressive epigenetic signatures (Figure 1C). This was further substantiated in prostate cancer tissues harboring ERG gene fusions being distinguishable by H3K27me3-containing and/or Polycomb-occupied genes. ERG was found to be a direct activator of EZH2 and the level of EZH2 expression was associated with the ERG status in a cohort of prostate tumors (Figure 7). Thus, TMPRSS2-ERG plays a central role as a “malignant regulatory switch” that shuts down androgen signaling, inhibiting normal prostate differentiation, and turning on EZH2 expression which induces an ES cell-like de-differentiation program.
As TMPRSS2-ERG gene fusions are androgen-responsive, they were thought to merely represent one of many mutated pathways emanating from AR signaling. Our results, however, suggest that TMPRSS2-ERG plays a much more fundamental role. As an early-onset genetic lesion, TMPRSS2-ERG gene fusion may provide a mechanism for AR over-expression and mutation in advanced prostate cancer. Antiandrogen treatments such as bicalutamide (also called Casodex) or flutamide are currently being used to treat advanced disease (Anderson, 2003). Unfortunately, patients treated with these AR antagonists often develop recurrent disease that is resistant to this therapy. Tumors from men with castration resistant metastatic prostate cancer (CRMPC) often overexpress AR through multiple mechanisms including AR amplification (Scher and Sawyers, 2005). Repression of AR by TMPRSS2-ERG may provide a malignant selection pressure contributing to recurrent tumors with AR amplification. This is supported by our observation of a negative correlation (r = −0.35, p = 0.0014) between AR and ERG expression in localized prostate tumors, but a positive correlation (r = 0.30, p = 0.058) in metastatic prostate cancers. Further, while AR amplification on its own is not sufficient to induce hyperplastic lesions, overexpression of a CMV-promoter driven AR (thus not susceptible to ERG repression, mimicking a hormone-refractory state with AR amplification), together with forced ERG overexpression, has recently been shown to promote the development of a more poorly differentiated, invasive adenocarcinoma (Zong et al., 2009). This may also suggest that therapies targeting AR may not produce a durable response in prostate cancer patients when the underlying mutation may in fact be TMPRSS2-ERG. Paradoxically, therapies employing high dose testosterone may have a beneficial effect transiently by favoring a normal differentiation state. Consistent with this, preclinical models suggest that high doses of exogenous testosterone inhibit prostate cancer growth while lows levels of testosterone promote tumor growth (Koivisto et al., 1997; Kokontis et al., 1998). Recently, high doses of exogenous testosterone have been shown to be safe in patients with CRMPC (Morris et al., 2009). Bicalutamide and flutamide exhibit a partial agonistic effect which may also promote normal prostate differentiation which is eventually overcome by TMPRSS2-ERG expression and consequent resistant disease.
Taken together, our findings provide a working model in which TMPRSS2-ERG plays a critical role in cancer progression by disrupting the AR lineage-specific differentiation program of the prostate and favoring EZH2-mediated cellular de-differentiation. In addition, by inhibiting AR signaling, TMPRSS2-ERG may exert a selective pressure for the development of prostate cancer that is resistance to hormone deprivation therapies. Furthermore, our study provides a compendium of 57 ChIP-Seq experiments of key transcription factors and histone modifications in prostate cancer, which will be invaluable for prostate cancer and steroid hormone research.
EXPERIMENTAL PROCEDURES
Cell Culture and In vitro overexpression, inhibition and function assays
LNCaP, VCaP, RWPE and 22RV1 cell lines were obtained from ATCC and cultured accordingly. Adenoviral and lentiviral constructs expressing ERG or GUS control were generated as previously described by recombining pCR8-ERG and a control entry clone (pENTR-GUS) (Invitrogen) with pAD/CMV/V5 and pLenti6/CMV/V5, respectively, using LR Clonase II (Invitrogen) (Tomlins et al., 2008). The pENTR-GUS serves as a positive control for the LR recombination reaction and GUS refers to beta-Glucuronidase, a protein detectable by either a fluorescent or blue substrate in the cells. VCaP cells were infected with ERG or GUS lentiviruses, and stable clones expressing ERG (VCaP+ERG) or GUS (VCaP+GUS) were selected.
For RNA interference, cells were treated with non-targeting siRNA (D-001210-01), or siRNA specific to ERG (D-003886-01) or AR (J-003400) from Dharmacon. For androgen treatment, cells were hormone starved for 2–3 days before treated with 1nM R1881 for 48 hrs (for expression assay) or 10nM R1881 for 16 hr (for ChIP). Cell proliferation and invasion assays were carried out as previously described (Yu et al., 2007a).
ChIP and ChIP-Seq Assays
ChIP was carried out as previously described (Yu et al., 2007a) and detailed in the Supplemental Text. Antibodies used include AR (#06-680), H3K27me3 (#07-449), and Ace-H3 (#06-599) from Millipore, ERG (SC354X, Santa Cruz), and H3K4me3 (ab8580), H3K9me3 (ab8898), H3K4me1 (ab8895), H3K4me2 (ab7766), H3K36me3 (ab9050), Pan-H3 (ab1791), FoxA1 (ab23738) and RNA PolII (ab817) from Abcam. ChIP DNA was prepared into libraries and sequenced using the Genome Analyzer (Illumina) following manufacturer’s protocols. The raw sequencing image data was analyzed by the Illumina analysis pipeline, aligned to the unmasked human reference genome (NCBI v36, hg18) using ELAND (Illumina), which is further analyzed by HPeak (http://www.sph.umich.edu/csg/qin/HPeak/ and Supplemental Text) to identify enriched peak regions.
Quantitative PCR
Q-PCR was performed using Power SYBR Green Mastermix (Applied Biosystems) on an Applied Biosystems 7300 Real Time PCR machine as previously described (Yu et al., 2007a). All primers (listed in Supplemental Text) were designed using Primer 3 and synthesized by Integrated DNA Technologies.
In Vitro Protein Interaction Assay
ERG and its sub-domains were cloned into pFN19A (HaloTag) vector (Promega, Wisconsin) and expressed in TNT® SP6 High-Yield Wheat Germ Reaction. A total of 2.0 μl of cell-free reaction containing the HaloTag® fusion protein was mixed with 8μl of HaloTag® Biotin Ligand (final concentration 1μM), and incubated at room temperature (RT) for 30 minutes. The biotin-labeled samples were separated on SDS gel and blotted using HRP-streptavidin to confirm Halo-Tag fusion protein expression. In in vitro pull down assay AR protein fragments containing the DNA-binding and ligand-binding domains were expressed in bacteria with GST-tag at N-terminal and purified by glutathione beads. A total of 12.5μl of cell-free reaction containing HaloTag fusion proteins were incubated with 100ng of GST-AR proteins in PBST (0.1% tween) at 4°C overnight. Ten microliter HaloLink beads (Promega) were blocked in BSA at 4°C overnight. After 3 washes with PBS, the beads were mixed with Halo-ERG and incubated at RT for 1 hr. Halolink beads were then washed with PBST for 4 times and eluted in SDS loading buffer. Proteins were separated on SDS gel and blotted with anti-GST mAb (Sigma) to detect AR. Bare HaloLink beads without HaloTag fusion proteins were used as negative controls.
Luciferase Reporter Assay
Luciferase reporter assays were performed as previously described (Cao et al.). Briefly, LNCaP cells were infected with ERG or LacZ adenovirus. At 24 hours post-infection promoter-reporter constructs were co-transfected along with pRL-TK (internal control). Cells were lysed with passive lysis buffer and luciferase activities were monitored using dual luciferase assay system (Promega, Madison, WI) after an additional 24 hours of incubation. Reporter constructs, pGL3-CDH1-Luc, pGL3 PSA6.0-Luc and pGL3-ALP-Luc were provided by Drs, Eric Fearon (Hajra et al., 1999), Evan T. Keller (Mizokami et al., 2000) and Mitsutoki Hatta (Hatta et al., 2002), respectively. TMPRSS2 (chr21:41,801,764-41,802,692) and AR (chrX:66,679,691-66,680,682) promoters were PCR amplified and sub-cloned into pGL4.14 vector (Promega) using Bgl2 and Hind3 enzymes.
Bioinformatics Analysis
Pre-defined motifs of transcription factors was searched using MatInspector (Cartharius et al., 2005) (Genomatix). Over-representation of a motif in ChIP-Seq binding sites were evaluated against control sequences randomly collected from regions other than binding sites and the significance tested by Fisher’s exact test. De novo motif was analyzed using MEME (Bailey and Elkan, 1994). GSEA analysis was performed as detailed in Supplementary Text. Molecular Concept Analysis (MCM) was performed using a query gene list to search for all concepts available in the Oncomine database as previously described (Yu et al., 2007b). Concepts with significant enrichment by the query concept were exported into a table. Representative concepts were selectively shown as a network in a figure.
HIGHLIGHTS
- Genome-wide location analysis of AR, ERG, and epigenetic marks in prostate cancer
- Interaction and co-localization of AR and ERG to target genes in prostate cancer
- ERG disrupts AR-mediated lineage-specific differentiation of the prostate
- ERG induces EZH2 facilitating de-differentiation in prostate cancer
SIGNIFICANCE
Despite its high prevalence in prostate cancer, the TMPRSS2-ERG gene fusion was thought to merely represent one of many downstream mutations emanating from AR signaling. Here we mapped the genomic landscape of AR, ERG and key histone modifications in prostate cancer. While AR was found to activate genetic programs involved in prostate differentiation, we found that ERG binds to AR and a majority of AR target genes disrupting androgen signaling. In addition, ERG activates the PcG protein EZH2 facilitating a stem cell-like de-differentiation program. Thus, TMPRSS2-ERG plays a fundamental role in cancer by abrogating lineage-specific differentiation of the prostate. Furthermore, we provide a compendium of genome-wide location analyses related to AR and TMPRSS2-ERG that will be useful to the research community.
Supplementary Material
01
Acknowledgments
We thank Rohit Mehra and Bo Han for histopathologic assessment of tissue, Jincheng Shen, Shankar Kalyana-Sundaram and Arun Sreekumar for technical assistance. A.M.C. is supported by a Burroughs Welcome Foundation Award in Clinical Translational Research, a Doris Duke Charitable Foundation Distinguished Clinical Investigator Award, and the Howard Hughes Medical Institute. A.M.C. is an American Cancer Society Research Professor. This work was supported in part by the NIH Prostate Specialized Program of Research Excellence grant P50CA69568, Early Detection Research Network grant UO1 111275 (to A.M.C.), 1R01CA132874-01A1 (to A.M.C.) and K99CA129565-01A1 (to J.Y.), the Prostate Cancer Foundation (to A.M.C.), and the U.S. Department of Defense PC051081 (to A.M.C. and S.V.), and PC080665 (to J.Y.).
Footnotes
ACCESSION NUMBERS
The microarray and short-read sequencing data have been deposited in the National Center for Biotechnology Information Gene Expression Omnibus and Short-Read Archive with the accession number GSE14097.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Anderson J. The role of antiandrogen monotherapy in the treatment of prostate cancer. BJU international. 2003;91:455–461. doi: 10.1046/j.1464-410x.2003.04026.x. [DOI] [PubMed] [Google Scholar]
- Bailey TL, Elkan C. Fitting a mixture model by expectation maximization to discover motifs in biopolymers. Proceedings/International Conference on Intelligent Systems for Molecular Biology ; ISMB. 1994;2:28–36. [PubMed] [Google Scholar]
- Barski A, Cuddapah S, Cui K, Roh TY, Schones DE, Wang Z, Wei G, Chepelev I, Zhao K. High-resolution profiling of histone methylations in the human genome. Cell. 2007;129:823–837. doi: 10.1016/j.cell.2007.05.009. [DOI] [PubMed] [Google Scholar]
- Ben-Porath I, Thomson MW, Carey VJ, Ge R, Bell GW, Regev A, Weinberg RA. An embryonic stem cell-like gene expression signature in poorly differentiated aggressive human tumors. Nat Genet. 2008;40:499–507. doi: 10.1038/ng.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyer LA, Plath K, Zeitlinger J, Brambrink T, Medeiros LA, Lee TI, Levine SS, Wernig M, Tajonar A, Ray MK, et al. Polycomb complexes repress developmental regulators in murine embryonic stem cells. Nature. 2006;441:349–353. doi: 10.1038/nature04733. [DOI] [PubMed] [Google Scholar]
- Cao Q, Yu J, Dhanasekaran SM, Kim JH, Mani RS, Tomlins SA, Mehra R, Laxman B, Cao X, Yu J, et al. Repression of E-cadherin by the polycomb group protein EZH2 in cancer. Oncogene. 2008;27:7274–7284. doi: 10.1038/onc.2008.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cartharius K, Frech K, Grote K, Klocke B, Haltmeier M, Klingenhoff A, Frisch M, Bayerlein M, Werner T. MatInspector and beyond: promoter analysis based on transcription factor binding sites. Bioinformatics (Oxford, England) 2005;21:2933–2942. doi: 10.1093/bioinformatics/bti473. [DOI] [PubMed] [Google Scholar]
- Carver BS, Tran J, Chen Z, Carracedo-Perez A, Alimonti A, Nardella C, Gopalan A, Scardino PT, Cordon-Cardo C, Gerald W, Pandolfi PP. ETS rearrangements and prostate cancer initiation. Nature. 2009;457:E1. doi: 10.1038/nature07738. discussion E2–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H, Tu SW, Hsieh JT. Down-regulation of human DAB2IP gene expression mediated by polycomb Ezh2 complex and histone deacetylase in prostate cancer. J Biol Chem. 2005;280:22437–22444. doi: 10.1074/jbc.M501379200. [DOI] [PubMed] [Google Scholar]
- Clark J, Merson S, Jhavar S, Flohr P, Edwards S, Foster CS, Eeles R, Martin FL, Phillips DH, Crundwell M, et al. Diversity of TMPRSS2-ERG fusion transcripts in the human prostate. Oncogene. 2007;26:2667–2673. doi: 10.1038/sj.onc.1210070. [DOI] [PubMed] [Google Scholar]
- Hajra KM, Ji X, Fearon ER. Extinction of E-cadherin expression in breast cancer via a dominant repression pathway acting on proximal promoter elements. Oncogene. 1999;18:7274–7279. doi: 10.1038/sj.onc.1203336. [DOI] [PubMed] [Google Scholar]
- Hatta M, Daitoku H, Matsuzaki H, Deyama Y, Yoshimura Y, Suzuki K, Matsumoto A, Fukamizu A. Regulation of alkaline phosphatase promoter activity by forkhead transcription factor FKHR. International journal of molecular medicine. 2002;9:147–152. [PubMed] [Google Scholar]
- Heemers HV, Tindall DJ. Androgen receptor (AR) coregulators: a diversity of functions converging on and regulating the AR transcriptional complex. Endocrine reviews. 2007;28:778–808. doi: 10.1210/er.2007-0019. [DOI] [PubMed] [Google Scholar]
- Hermans KG, van Marion R, van Dekken H, Jenster G, van Weerden WM, Trapman J. TMPRSS2:ERG fusion by translocation or interstitial deletion is highly relevant in androgen-dependent prostate cancer, but is bypassed in late-stage androgen receptor-negative prostate cancer. Cancer Res. 2006;66:10658–10663. doi: 10.1158/0008-5472.CAN-06-1871. [DOI] [PubMed] [Google Scholar]
- Hsu T, Trojanowska M, Watson DK. Ets proteins in biological control and cancer. Journal of cellular biochemistry. 2004;91:896–903. doi: 10.1002/jcb.20012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia L, Berman BP, Jariwala U, Yan X, Cogan JP, Walters A, Chen T, Buchanan G, Frenkel B, Coetzee GA. Genomic androgen receptor-occupied regions with different functions, defined by histone acetylation, coregulators and transcriptional capacity. PLoS ONE. 2008;3:e3645. doi: 10.1371/journal.pone.0003645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson DS, Mortazavi A, Myers RM, Wold B. Genome-wide mapping of in vivo protein-DNA interactions. Science (New York, NY) 2007;316:1497–1502. doi: 10.1126/science.1141319. [DOI] [PubMed] [Google Scholar]
- King JC, Xu J, Wongvipat J, Hieronymus H, Carver BS, Leung DH, Taylor BS, Sander C, Cardiff RD, Couto SS, et al. Cooperativity of TMPRSS2-ERG with PI3-kinase pathway activation in prostate oncogenesis. Nat Genet. 2009;41:524–526. doi: 10.1038/ng.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klezovitch O, Risk M, Coleman I, Lucas JM, Null M, True LD, Nelson PS, Vasioukhin V. A causal role for ERG in neoplastic transformation of prostate epithelium. Proc Natl Acad Sci U S A. 2008;105:2105–2110. doi: 10.1073/pnas.0711711105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koivisto P, Kononen J, Palmberg C, Tammela T, Hyytinen E, Isola J, Trapman J, Cleutjens K, Noordzij A, Visakorpi T, Kallioniemi OP. Androgen receptor gene amplification: a possible molecular mechanism for androgen deprivation therapy failure in prostate cancer. Cancer Res. 1997;57:314–319. [PubMed] [Google Scholar]
- Kokontis JM, Hay N, Liao S. Progression of LNCaP prostate tumor cells during androgen deprivation: hormone-independent growth, repression of proliferation by androgen, and role for p27Kip1 in androgen-induced cell cycle arrest. Molecular endocrinology (Baltimore, Md) 1998;12:941–953. doi: 10.1210/mend.12.7.0136. [DOI] [PubMed] [Google Scholar]
- Lee TI, Jenner RG, Boyer LA, Guenther MG, Levine SS, Kumar RM, Chevalier B, Johnstone SE, Cole MF, Isono K, et al. Control of developmental regulators by Polycomb in human embryonic stem cells. Cell. 2006;125:301–313. doi: 10.1016/j.cell.2006.02.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lupien M, Eeckhoute J, Meyer CA, Wang Q, Zhang Y, Li W, Carroll JS, Liu XS, Brown M. FoxA1 translates epigenetic signatures into enhancer-driven lineage-specific transcription. Cell. 2008;132:958–970. doi: 10.1016/j.cell.2008.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massie CE, Adryan B, Barbosa-Morais NL, Lynch AG, Tran MG, Neal DE, Mills IG. New androgen receptor genomic targets show an interaction with the ETS1 transcription factor. EMBO Rep. 2007;8:871–878. doi: 10.1038/sj.embor.7401046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikkelsen TS, Ku M, Jaffe DB, Issac B, Lieberman E, Giannoukos G, Alvarez P, Brockman W, Kim TK, Koche RP, et al. Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature. 2007;448:553–560. doi: 10.1038/nature06008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Min J, Zaslavsky A, Fedele G, McLaughlin SK, Reczek EE, De Raedt T, Guney I, Strochlic DE, Macconaill LE, Beroukhim R, et al. An oncogene-tumor suppressor cascade drives metastatic prostate cancer by coordinately activating Ras and nuclear factor-kappaB. Nat Med. doi: 10.1038/nm.2100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizokami A, Gotoh A, Yamada H, Keller ET, Matsumoto T. Tumor necrosis factor-alpha represses androgen sensitivity in the LNCaP prostate cancer cell line. The Journal of urology. 2000;164:800–805. doi: 10.1097/00005392-200009010-00053. [DOI] [PubMed] [Google Scholar]
- Morris MJ, Huang D, Kelly WK, Slovin SF, Stephenson RD, Eicher C, Delacruz A, Curley T, Schwartz LH, Scher HI. Phase 1 Trial of High-Dose Exogenous Testosterone in Patients with Castration-Resistant Metastatic Prostate Cancer. European urology. 2009 doi: 10.1016/j.eururo.2009.03.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nye JA, Petersen JM, Gunther CV, Jonsen MD, Graves BJ. Interaction of murine ets-1 with GGA-binding sites establishes the ETS domain as a new DNA-binding motif. Genes Dev. 1992;6:975–990. doi: 10.1101/gad.6.6.975. [DOI] [PubMed] [Google Scholar]
- Oikawa T, Yamada T. Molecular biology of the Ets family of transcription factors. Gene. 2003;303:11–34. doi: 10.1016/s0378-1119(02)01156-3. [DOI] [PubMed] [Google Scholar]
- Perner S, Demichelis F, Beroukhim R, Schmidt FH, Mosquera JM, Setlur S, Tchinda J, Tomlins SA, Hofer MD, Pienta KG, et al. TMPRSS2:ERG fusion-associated deletions provide insight into the heterogeneity of prostate cancer. Cancer Res. 2006;66:8337–8341. doi: 10.1158/0008-5472.CAN-06-1482. [DOI] [PubMed] [Google Scholar]
- Scher HI, Sawyers CL. Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J Clin Oncol. 2005;23:8253–8261. doi: 10.1200/JCO.2005.03.4777. [DOI] [PubMed] [Google Scholar]
- Shin S, Kim TD, Jin F, van Deursen JM, Dehm SM, Tindall DJ, Grande JP, Munz JM, Vasmatzis G, Janknecht R. Induction of prostatic intraepithelial neoplasia and modulation of androgen receptor by ETS variant 1/ETS-related protein 81. Cancer Res. 2009;69:8102–8110. doi: 10.1158/0008-5472.CAN-09-0941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soller MJ, Isaksson M, Elfving P, Soller W, Lundgren R, Panagopoulos I. Confirmation of the high frequency of the TMPRSS2/ERG fusion gene in prostate cancer. Genes Chromosomes Cancer. 2006;45:717–719. doi: 10.1002/gcc.20329. [DOI] [PubMed] [Google Scholar]
- Sun C, Dobi A, Mohamed A, Li H, Thangapazham RL, Furusato B, Shaheduzzaman S, Tan SH, Vaidyanathan G, Whitman E, et al. TMPRSS2-ERG fusion, a common genomic alteration in prostate cancer activates C-MYC and abrogates prostate epithelial differentiation. Oncogene. 2008;27:5348–5353. doi: 10.1038/onc.2008.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomlins SA, Laxman B, Dhanasekaran SM, Helgeson BE, Cao X, Morris DS, Menon A, Jing X, Cao Q, Han B, et al. Distinct classes of chromosomal rearrangements create oncogenic ETS gene fusions in prostate cancer. Nature. 2007;448:595–599. doi: 10.1038/nature06024. [DOI] [PubMed] [Google Scholar]
- Tomlins SA, Laxman B, Varambally S, Cao X, Yu J, Helgeson BE, Cao Q, Prensner JR, Rubin MA, Shah RB, et al. Role of the TMPRSS2-ERG gene fusion in prostate cancer. Neoplasia (New York, NY) 2008;10:177–188. doi: 10.1593/neo.07822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomlins SA, Rhodes DR, Perner S, Dhanasekaran SM, Mehra R, Sun XW, Varambally S, Cao X, Tchinda J, Kuefer R, et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science. 2005;310:644–648. doi: 10.1126/science.1117679. [DOI] [PubMed] [Google Scholar]
- Varambally S, Dhanasekaran SM, Zhou M, Barrette TR, Kumar-Sinha C, Sanda MG, Ghosh D, Pienta KJ, Sewalt RG, Otte AP, et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature. 2002;419:624–629. doi: 10.1038/nature01075. [DOI] [PubMed] [Google Scholar]
- Wang J, Cai Y, Yu W, Ren C, Spencer DM, Ittmann M. Pleiotropic biological activities of alternatively spliced TMPRSS2/ERG fusion gene transcripts. Cancer Res. 2008;68:8516–8524. doi: 10.1158/0008-5472.CAN-08-1147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q, Li W, Liu XS, Carroll JS, Janne OA, Keeton EK, Chinnaiyan AM, Pienta KJ, Brown M. A hierarchical network of transcription factors governs androgen receptor-dependent prostate cancer growth. Mol Cell. 2007;27:380–392. doi: 10.1016/j.molcel.2007.05.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q, Li W, Zhang Y, Yuan X, Xu K, Yu J, Chen Z, Beroukhim R, Wang H, Lupien M, et al. Androgen receptor regulates a distinct transcription program in androgen-independent prostate cancer. Cell. 2009;138:245–256. doi: 10.1016/j.cell.2009.04.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright ME, Tsai MJ, Aebersold R. Androgen receptor represses the neuroendocrine transdifferentiation process in prostate cancer cells. Mol Endocrinol. 2003;17:1726–1737. doi: 10.1210/me.2003-0031. [DOI] [PubMed] [Google Scholar]
- Xie D, Gore C, Liu J, Pong RC, Mason R, Hao G, Long M, Kabbani W, Yu L, Zhang H, et al. Role of DAB2IP in modulating epithelial-to-mesenchymal transition and prostate cancer metastasis. Proc Natl Acad Sci U S A. 107:2485–2490. doi: 10.1073/pnas.0908133107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshimoto M, Joshua AM, Chilton-Macneill S, Bayani J, Selvarajah S, Evans AJ, Zielenska M, Squire JA. Three-color FISH analysis of TMPRSS2/ERG fusions in prostate cancer indicates that genomic microdeletion of chromosome 21 is associated with rearrangement. Neoplasia. 2006;8:465–469. doi: 10.1593/neo.06283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J, Cao Q, Mehra R, Laxman B, Yu J, Tomlins SA, Creighton CJ, Dhanasekaran SM, Shen R, Chen G, et al. Integrative genomics analysis reveals silencing of beta-adrenergic signaling by polycomb in prostate cancer. Cancer cell. 2007a;12:419–431. doi: 10.1016/j.ccr.2007.10.016. [DOI] [PubMed] [Google Scholar]
- Yu J, Rhodes DR, Tomlins SA, Cao X, Chen G, Mehra R, Wang X, Ghosh D, Shah RB, Varambally S, et al. A polycomb repression signature in metastatic prostate cancer predicts cancer outcome. Cancer Res. 2007b;67:10657–10663. doi: 10.1158/0008-5472.CAN-07-2498. [DOI] [PubMed] [Google Scholar]
- Yu J, Yu J, Rhodes DR, Tomlins SA, Cao X, Chen G, Mehra R, Wang X, Ghosh D, Shah RB, et al. A polycomb repression signature in metastatic prostate cancer predicts cancer outcome. Cancer Res. 2007c;67:10657–10663. doi: 10.1158/0008-5472.CAN-07-2498. [DOI] [PubMed] [Google Scholar]
- Zong Y, Xin L, Goldstein AS, Lawson DA, Teitell MA, Witte ON. ETS family transcription factors collaborate with alternative signaling pathways to induce carcinoma from adult murine prostate cells. Proc Natl Acad Sci U S A. 2009;106:12465–12470. doi: 10.1073/pnas.0905931106. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
01