Microbead Arrays for the Analysis of ErbB Receptor Tyrosine Kinase Activation and Dimerization in Breast Cancer Cells (original) (raw)
Abstract
Receptor tyrosine kinases (RTKs) in the ErbB family (EGFR, ErbB2, ErbB3, and ErbB4) are implicated in a variety of human malignancies. Accordingly, determination of both expression and activation (dimerization/heterodimerization and phosphorylation) of ErbB proteins is critical in defining their functional role in cancer. Efficient and comprehensive methods to study molecular functions of ErbB family of RTKs are needed not only for improvements in diagnostics but also for early screening of targeted drugs (eg, small molecule inhibitors and therapeutic antibodies). We report development of 3 multiplex microbead immunoassays for simultaneous detection of expression, protein–protein interactions, and phosphorylation of these RTKs. These novel multiplex immunoassays were used to study ErbB RTKs under different cell activation conditions in 2 breast cancer cell lines (MDA-MB-453 and MDA-MB-468) and an epidermoid cancer cell line (A431). The results were confirmed by immunoprecipitation/western blot. Importantly, the multiplex immunoassay facilitated time-course studies in these cell lines after cell activation with EGF and neuregulin, revealing the kinetics of phosphorylation of the ErbB family RTKs. This study demonstrates the utility of the Luminex® multiplex system as an efficient and comprehensive approach to study different aspects of molecular roles of these RTKs. Importantly, the study provides proof-of-concept for the utility of the multiplex microbead immunoassay approach for potential use in efficient, robust, and rapid screening of drugs, particularly those targeting functional aspects of these potent signaling molecules. In addition, the assays described here may be useful for cancer diagnostics and monitoring efficacy of therapy targeting the ErbB family of RTKs.
Introduction
Overexpression of receptor tyrosine kinases (RTKs) belonging to the ErbB family of receptors (EGFR/ErbB1/HER1, ErbB2/HER2, ErbB3/HER3, and ErbB4/HER4) and/or their aberrant signaling is a major characteristic of many human malignancies.1–3 Most tumors express more than one of these ErbB family members. Overexpression of EGFR and ErbB2 has been documented in a variety of tumor types including breast tumor (63% of ErbB2-positive tumors were also strongly positive for ErbB3) with poor clinical outcome.4 EGFR is overexpressed in breast cancers with a positivity rate of 14%–91%, which is associated with a more aggressive phenotype and poor patient prognosis.5 ErbB2 is overexpressed in 20%–30% of invasive breast tumors4,6–8 and is a marker of poor prognosis.9 Although ErbB4 is the least well-understood family member in terms of its role in cancer, its expression in breast tumors has been associated with low cell proliferative index, increased survival, and reduced recurrence of tumors.4
These ErbB family RTKs are activated by various EGF-like growth factors. Binding of EGF to EGFR induces receptor dimerization and tyrosine autophosphorylation of specific residues within the cytoplasmic tail, initiating a complex cascade of cell signaling events leading to cell proliferation.1 Similarly, neuregulins transmit intracellular signals within target cells by interacting with ErbB3 and ErbB4.10 Ligand binding to the ErbB RTKs, followed by heterodimerization11 and phosphorylation, leads to the activation of downstream targets,12 resulting in intracellular signals stimulating cell proliferation and survival. Discovery of the role of RTKs in oncogenesis has lead to the development of novel anticancer therapeutics targeting these molecules. The advent of such therapies has in turn helped identify the need for a better understanding of molecular events involving RTK function for a variety of cancers. It is therefore critical to analyze intracellular signals transduced by RTKs under different conditions that may produce different cellular responses. From a mechanistic perspective, ErbB2 is considered to be a major factor in oncogenesis. However, ErbB2, when overexpressed alone, may exhibit different protein–protein interactions and phosphorylation dynamics that may affect its activity in comparison to its overexpression in conjunction with that of EGFR and/or ErbB3. Such differences in association of ErbB2 with other members of the ErbB RTK family may lead to differences in activation of intracellular signaling pathways, thus resulting in different cell behavior. To elucidate molecular events underlying the role of ErbB RTKs in oncogenesis, it is important to study expression of these proteins as well as their activation by dimerization/phosphorylation. Accordingly, capabilities of the multiplex approach to detect and quantitate not only protein expression, but also phosphorylation and protein–protein interactions are useful as tools for screening drugs in the early stage of development, that are designed to target functional properties of these clinically important ErbB signaling proteins.13,14 In addition, such screening tools may enhance the efficiency of studies on pharmacodynamic effects of small molecule inhibitors on activities of ErbB RTKs. Furthermore, the use of this efficient approach in studies on activation of these RTKs (phosphorylation and dimerization) may evolve into development of procedures for cancer diagnostics and monitoring efficacy of targeted therapy. In this report, we present proof-of-concept studies using a robust and efficient multiplex microbead immunoassay approach for the detection of expression and activation (phosphorylation/heterodimerization) of RTKs in the ErbB family. For each of the 4 ErbB family RTKs, 3 multiplex assays are described that enable the simultaneous detection of expression, heterodimerization, or phosphorylation of these key cell signaling proteins in cell line models.
Materials and Methods
Cells and Cell Activation
Breast carcinoma cells (MDA-MB-453 and MDA-MB-468) and human epidermoid carcinoma cells (A431) were cultured in Dulbecco's modified Eagle's medium (DMEM) supplied with 10% heat-inactivated newborn calf serum at 37°C and 50% CO2. Cells were grown to 65%–70% confluence in 25 or 75 cm2 tissue culture flasks, then starved in X-vivo medium (A431) or DMEM containing 0.1% FBS (MDA-MB-453 and MDA-MB-468) for 24 h before activation.15 Cells were treated with optimal concentration of EGF (16.5 nM concentration), neuregulin (1:500), or sodium pervanadate (6.6 mM).15
Lysate Preparation
Cells in tissue culture flasks were lysed with ice-cold buffer (PBS containing 1% Nonidet P-40, protease inhibitor cocktail, 0.5 mM sodium orthovanadate, and 1× serine/threonine phosphatase inhibitor). The cell lysate was immediately vortexed and incubated on ice for 15 min. Cell debris was removed by centrifugation at 12,000 rpm for 20 min.15 Total protein concentration of lysates was determined by BCA reagent kit (Bio-Rad, Laboratories, Hercules, CA). Lysates were aliquoted and stored at −80°C until used.
Antibodies and Reagents
Monoclonal antibodies against EGFR (05-104) for microbead coating and immunoprecipitation and biotinylated anti-phosphotyrosine (4G10) were purchased from Upstate, USA (Lake Placid, NY). Monoclonal antibodies against ErbB2, ErbB3, and ErbB4 for microbead coating and immunoprecipitation were purchased from R&D Systems (MAB1129, MAB3481, and MAB1131, respectively; Minneapolis, MN). Biotinylated antibody for total protein detection for EGFR (E101) was obtained from Leinco Technologies (St. Louis, MO) and those for ErbB2, ErbB3, and ErbB4 were purchased from R&D Systems (BAF1129, BAM3481, and BAF1131, respectively). Protease inhibitor cocktail tablets and purified 10% Nonidet P-40 were purchased from Roche Applied Science (Indianapolis, IN). Halt phosphatase inhibitor cocktail was purchased from Pierce (Rockford, IL). Protein G-conjugated Sepharose was from Sigma (St. Louis, MI). EGF was purchased from Upstate. Neuregulin (recombinant) was expressed and purified as previously described.16
Microbead Coating
Monoclonal antibodies against EGFR, ErbB2, ErbB3, and ErbB4 were coated as capture antibodies by conjugation to individual microbead sets. One microbead set was coated with BSA to control for nonspecific interactions, and another set was coated with biotin-conjugated goat IgG (Jackson Immunoresearch Laboratories, West Grove, PA) to serve as a positive control for the detection reagent (streptavidin-conjugated phycoerythrin). Proteins were conjugated to microbeads as previously described.15 In brief, microbeads were activated with sulfo-NHS (_N_-hydroxysulfosuccinamide; Pierce, Rockford, IL) and EDC (1-ethyl-3-[3-dimethylaminopropyl] carbodiimide; Pierce). The activated beads were washed with 50 mM MES (pH 6.0) buffer. To coat with antibody (protein), activated beads were resuspended in the relevant protein solution (25–100 μg/mL in 50 mM MES (pH 6.0) buffer. Mixture of activated beads and antibodies was incubated by shaking on a rocker for 2 h at RT for coupling. After coating, beads were washed twice with wash buffer (0.1% Tween-20 in phosphate-buffered saline [PBS], pH 7.40 and resuspended in 1 mL of blocking buffer (1% BSA; 0.1% Tween-20 in PBS, pH 7.4; 0.05% sodium azide). Blocking was performed by shaking on a rocker at room temperature for 30 min. After blocking, beads were washed twice in 1 mL blocking buffer. Finally, antibody-coated beads were resuspended in 1 mL blocking buffer and stored at 4°C for up to a week. For long-term storage, beads were kept frozen at −80°C for several months.
Microbead Suspension Array Immunoassay of Signaling Proteins
Immunoreactions were set up in 96-well, filter-bottomed plates designed for high-throughput separations (1.2 μm MultiScreen; Millipore Corporation, Billerica, MA) as previously detailed.15 Microbeads (2,000 beads of each set), coated with a specific antibody, were mixed. This multiplex, microbead mixture was added to each well. To this, 25 μL of cell lysate (0.4 mg/mL total protein) was added. The contents were mixed at 1,400 rpm on a plate shaker (Labnet International Inc., Woodbridge, NJ) for 2 h at room temperature (RT). After incubation with the lysate, liquid was drained from the bottom of the plate under vacuum. The microbeads were washed twice by adding 150 μL of wash buffer per well and draining out under vacuum successively. For detection of tyrosine-phosphorylated signaling proteins bound to antibodies coated on microbeads, 25 μL of biotinylated antiphosphotyrosine antibody 4G10 (0.5 μg/mL in wash buffer) was added as the detection reagent. To detect biotinylated 4G10, streptavidin conjugated to R-phycoerythrin was added at a dilution of 1:1,000 in wash buffer as the reporter molecule and incubated for 15 min at RT. Microbeads were washed once with wash buffer, resuspended in 100 μL of wash buffer per well, and analyzed in the Luminex-100TM instrument (Luminex Corporation, Austin, TX).
Luminex-100 Operation and Multiplex Data Analysis
The Luminex-100 instrument (Austin, TX) was set at the default settings, set by the manufacturer for routine applications, as directed in the user's manual. Data were acquired by Luminex Data Collection Software (Version 1.0). This software package was used for routine operation of the instrument, data acquisition, and data analysis. The instrument was calibrated with Calibration Beads supplied by the manufacturer to adjust the settings for bead set identification or “Classification” and for the detection of “Reporter” (phycoerythrin). Events were gated to exclude doublets and other aggregates. One hundred independent, gated events were acquired for each bead set. The median fluorescence intensity (MFI) or “signal” of a hundred events (beads) was used as a measure of the detection of protein phosphorylation. After acquisition by Luminex software, the data were further processed by Microsoft Excel software.15
Immunoprecipitation and Western Blotting
Immunoprecipitation and Western blotting were performed essentially as described previously.15 For immunoprecipitation, individual capture antibody (3 μg/mL) was mixed with 250 μL of cell lysates (900–1,500 μg/mL total protein) on a rotator overnight at 4°C. Protein G-conjugated Sepharose was added (90 μL of 20% slurry) and mixed on the rotator for 1 h at 4°C. Sepharose beads were washed 3 times in wash buffer (PBS containing 1% Tween-20, 100 mM NaCl) and resuspended in 24 μL of PBS plus 8 μL 4× sample buffer. The samples were boiled for 5 min. Immunocomplexes were resolved on SDS-polyacrylamide gels (8%–16% precast gradient Tris–glycine gels, Novex Immunodetection; Invitrogen, Carlsbad, CA). Membranes were blocked in 5% BSA (Roche Applied Science, Indianapolis, IN). Tyrosine phosphorylation was detected with biotin-conjugated anti-phosphotyrosine antibody (4G10, 0.5 μg/mL). Blots were developed with Vectastain ABC detection reagent (Vector Laboratories, Burlingame, CA) and ECL Plus Western blotting detection system (Amersham Biosciences, Piscataway, NJ) and visualized on a Typhoon 9410 variable mode imager (Amersham Biosciences).
For Western blotting, 30 μg of cell lysate was electrophoresed on 8%–16% SDS-polyacrylamide gels under reducing conditions for EGFR (biotinylated antibody, Upstate), ErbB3 (biotinylated antibody, Santa Cruz Biotechnology, Santa Cruz, CA), ErbB4 (biotinylated antibody R&D Systems), and actin antibody (R&D Systems), or under nonreducing conditions for ErbB2 (biotinylated antibody R&D Systems). After electrophoresis, proteins were transferred onto polyvinylidene difluoride (PVDF) membranes (Bio-Rad) and nonspecific binding sites were blocked with 5% nonfat dry milk (Oxiod Ltd, Basingstoke, Hampshire, England) in PBS with 1% Tween-20. Blots were probed with Anti-Mouse HRP-linked IgG (Cell Signaling Technologies, Cambridge, MA) for EGFR and ErbB2, anti-Rabbit antibody HRP-linked IgG (Cell Signaling) for ErbB3. The antibodies were used at a concentration of 0.1 mg/mL. Bands were visualized by horseradish peroxidase/hydrogen peroxide-catalyzed oxidation of luminol in the enhanced chemiluminescence (ECL) reaction.
Results
Expression of ErbB Family Receptors
To demonstrate specific detection of members of the ErbB family of RTKs by multiplex microbead suspension array, their expression was examined in 2 breast cancer cell lines MDA-MB-453 and MDA-MB-468, and a human epidermoid carcinoma cell line A431. The MDA-MB-453 cell line expresses ErbB2 and ErbB3 but does not show detectable amounts of EGFR and ErbB4. The MDA-MB-468 cell line expresses EGFR and ErbB3 receptor but does not produce detectable amounts of ErbB2; A431 cells express EGFR, ErbB2, and ErbB3 receptors.16,17
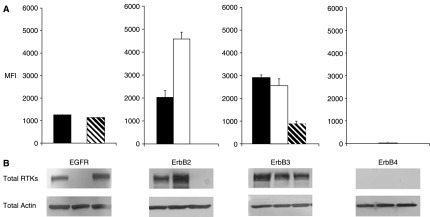
Four sets of microbeads, each set coated with monoclonal antibodies to EGFR, ErbB2, ErbB3, or ErbB4, were mixed and incubated with lysates prepared from nonactivated cells. In addition, a microbead set coated with BSA was included as a baseline control, and a microbead set coated with biotin was added as a positive control in the bead mixture. Total protein expression of EGFR, ErbB2, ErbB3, and ErbB4 was detected with biotin-conjugated detection antibody (distinct from capture antibody coupled to microbeads) against each individual RTK (Fig. 1A).
Fig. 1.
(A) Expression levels of ErbB family of receptors analyzed by multiplex microbead suspension immunoassay in MDA-MB-453 (hashed bars), MDA-MB-468 (open bars), and A431 (closed bars) cells. A mixture of microbeads coated with antibodies to EGFR, ErbB2, ErbB3, and ErbB4 were incubated with lysates from nonactivated cells. Detection of protein expression was achieved by a second antibody against each individual receptor protein. Error bars represent standard error of n = 4 values. (B) Western blot analysis of EGFR, ErbB2, ErbB3, and ErbB4 expression. Actin expression shows equal loading. Abbreviations: MFI, median fluorescence intensity; RTK, receptor tyrosine kinase.
The multiplex immunoassay results were confirmed by IP/WB for the detection of the 4 ErbB family RTKs (Fig. 1B). Both methods consistently showed that EGFR was expressed in MDA-MB-468 and A431 cells, but not in MDA-MB-453 cells. This finding is consistent with previous reports.16,17 MDA-MB-453 and A431 cells expressed ErbB2, whereas MDA-MB-468 cells showed no detectable expression of ErbB2. All 3 cell lines expressed ErbB3, and ErbB4 was not detected in any of the cell lines (Fig. 1). Taken together, these results confirmed that the multiplex immunoassay simultaneously detected expression of 3 of the 4 ErbB family RTKs in a single sample of cell lysate.
Phosphorylation of ErbB RTKs
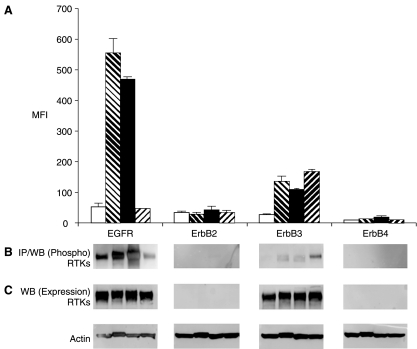
To assess the phosphorylation status of the ErbB receptors for cell activation, cells were treated with physiological ligands (EGF and neuregulin) or the phosphatase inhibitor sodium pervanadate. Pervanadate was used as a general cell activator to produce positive control cell lysates for tyrosine phosphorylated proteins. Treatment with pervanadate inhibits intracellular tyrosine phosphatases, resulting in sustained phosphorylation of various tyrosine kinases and substrates that reflects the activation state of the cells.15 Tyrosine kinases activated in this manner can also activate downstream serine/threonine kinases. In MDA-MB-468 cells, which express EGFR and ErbB3 but not ErbB2, the phosphorylation level of EGFR increased by 10.3-fold and 8.7-fold upon stimulation following treatment with sodium pervanadate or EGF, respectively (Fig. 2A). Activation of ErbB3 was also observed. Pervanadate treatment of these cells resulted in 4.6-fold increase in phosphorylation of ErbB3, and a 5.6-fold increase was observed upon treatment of cells with neuregulin. This cell line does not express the ErbB2 receptor17,18; accordingly, phosphorylation of ErbB2 or ErbB4 was not detected (Fig. 2).
Fig. 2.
(A) Phosphoproteomic analysis of RTKs by multiplex microbead suspension array immunoassay in MDA-MB-468 cells. Cells were used as untreated (empty bars), treated with 6.6 mM sodium pervanadate for 5 min (hashed bars), 16.5 nM EGF (closed bars), or 1:500 neuregulin for 7 min (reverse hashed bars). A mixture of microbeads coated with antibodies to EGFR, ErbB2, ErbB3, and ErbB4 were incubated with the cell lysates. Error bars represent standard error of n = 4 values. (B) Immunoprecipitation and western blot analysis of phosphorylation and total ErbB receptors in MDA-MB-468 cells. Antibodies used for immunoprecipitation were the same as those coated on the microbeads for multiplex analysis. (C) Western blot analyses were performed for the detection of total RTK proteins (actin shows equal loading). Abbreviations: MFI, median fluorescence intensity; RTK, receptor tyrosine kinase.
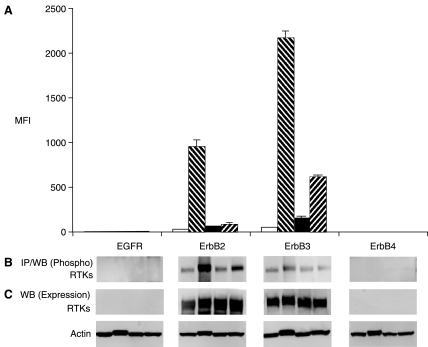
In MDA-MB-453 cells, treated with sodium pervanadate, levels of phosphorylated forms of ErbB2 and ErbB3 increased by 30.8-fold and 39.4-fold, respectively (Fig. 3A). Upon treatment with neuregulin (ErbB3 agonist), ErbB3 phosphorylation increased by 11.3-fold compared to untreated cells. In this experiment, neither expression nor phosphorylation of ErbB4 was detected (Fig. 3).
Fig. 3.
Phosphoproteomic analysis of RTKs by multiplex microbead immunoassay in MDA-MB-453 cells. Details are the same as described for Figure 2. Abbreviations: MFI, median fluorescence intensity; RTK, receptor tyrosine kinase.
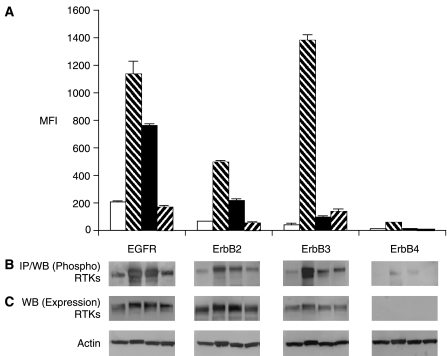
Analysis of A431 cells by multiplex microbead suspension array showed that EGFR, ErbB2, and ErbB3 were hyperphosphorylated upon treatment with pervanadate. Phosphorylation of EGFR increased by 5.5-fold, phosphorylation of ErbB2 receptor increased by 8.2-fold, and phosphorylation of ErbB3 receptor increased by 34.5-fold (Fig. 4A). These results confirmed that the multiplex assay was able to detect phosphorylation of 3 of 4 of the ErbB family RTKs. EGF induced phosphorylation of EGFR by 3.8-fold, and ErbB2 receptor by 3.3-fold. However, in the A431 cell line, neuregulin treatment did not increase the level of phosphorylation of ErbB3 as dramatically as in the MDA-MB-468 cell line (Fig. 4). This difference is likely due to EGFR sequestering ErbB2 away from ErbB3, and thereby compromising the ability of neuregulin to lead to dimerization of ErbB2 and ErbB3.
Fig. 4.
Phosphoproteomic analysis of RTKs by multiplex microbead immunoassay in A431 cells. Details are the same as described for Figure 2. Abbreviations: MFI, median fluorescence intensity; RTK, receptor tyrosine kinase.
To confirm the specificity of antibodies used in coating microbeads for capturing ErbB receptors, lysates of stimulated cells were also tested by IP and WB. Results obtained in IP/WB analysis were generally similar to those in the multiplex microbead assay (Figs. 2–4, panels B). However, in the case of MDA-MB-453 cells, increase in phosphorylation of ErbB3 above the baseline was not readily detected by IP/WB even after treatment with pervanadate (Fig. 3). The total amount of ErbB receptors in lysates from nontreated and treated cells was similar (Figs. 2–4, panel C, lower part). Thus, the results of the multiplex microbead immunoassays are consistent with those obtained by the conventional IP/WB method. However, the advantage of the multiplex microbead immunoassay is that it enables analysis of multiple proteins in a single sample with internal controls and affords a higher throughput.
Phosphorylation Kinetics of RTKs in Cells Treated With EGF or Neuregulin
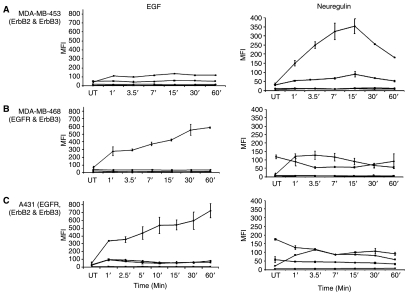
To study the kinetics of phosphorylation of ErbB family of RTKs upon stimulation with growth factors, a time-course experiment was performed by treating MDA-MB-453, MDA-MB-468, and A431 cells with EGF or neuregulin for various intervals of time ranging from 1 to 60 min. EGF activation induced phosphorylation of EGFR in MDA-MB-468 and A431 cells (Fig. 5B) but not in MDA-MB-453 cells that lack EGFR (Fig. 5A). Interestingly, patterns of phosphorylation of EGFR in MDA-MB-468 and A431 cells are very similar. After treatment with EGF, phosphorylation of EGFR increased in both MDA-MB-468 and A431 cells within 1 min; tyrosine phosphorylation continued to increase throughout the 60-min period of observation (Fig. 5).
Fig. 5.
Kinetics of phosphorylation of RTKs in cells treated with EGF or neuregulin. (A) MDA-MB-453, (B) MDA-MB-468, and (C) A431 cells were activated with 16.5 nM EGF or 1:500 neuregulin for various times ranging from 1 to 60 min. Microbeads used in the multiplex assay were coated with antibodies against EGFR (diamond), ErbB2 (square), ErbB3 (triangle), and ErbB4 (cross). Multiplex microbead immunoassay was performed as described for Figure 2. Detection of phosphorylation was performed by 4G10. Error bars represent standard error of n = 4 values. Abbreviations: MFI, median fluorescence intensity; RTK, receptor tyrosine kinase.
Analysis of the time-course treatment with neuregulin revealed the kinetics of phosphorylation of the ErbB3 receptor. A clear increase in phosphorylation was observed in MDA-MB-453 cells that peaked at 15 min and then declined over the next 45 min (Fig. 5A). A lower level of increase in phosphorylation of ErbB3 was observed in neuregulin-treated MDA-MB-468 and A431 cells in comparison to untreated cells (Fig. 5B and 5C).
Investigation of Protein-Protein Interactions of RTKs
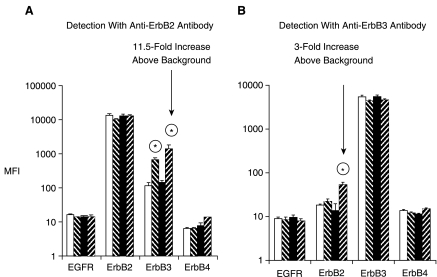
In breast cancer cell lines, protein-protein interaction between ErbB RTKs, under cell activation conditions were investigated by the multiplex microbead immunoassay. Cell lysates were analyzed from either nonactivated cells, or those treated with pervanadate, EGF, or neuregulin. As expected, when multiplex immunoassay was performed using ErbB2-specific detection antibody, this RTK displayed a strong signal for the microbeads coated with ErbB2 capture antibody (Fig. 6). This finding, as expected, shows the presence of ErbB2 in MDA-MB-453 cell lysates (Fig. 6A). As expected, the ErbB2 signal intensity on ErbB2-specific microbeads, for each of the 4 lysates, is very high regardless of cell activation condition. However, if heterodimer formation occurred under certain cell activation conditions, ErbB2 detection antibody would also produce a signal above the basal level for a microbead set other than the microbead set specific for capturing ErbB2. Indeed, ErbB2 detection antibody produced a significant signal on ErbB3-specific capture beads (Fig. 6A). This signal with ErbB2 detection antibody was only present in lysates obtained from cells that were activated by either neuregulin (11.5-fold signal increase over nonstimulated cells) or pervanadate (5-fold signal increase); that is, these treatment conditions lead to ErbB3 activation. This result indicates that the multiplex microbead immunoassay enabled the detection of ErbB2:ErbB3 heterodimers under the activation conditions that favored these protein–protein interactions.1 In addition, this result is consistent with the previous demonstration with other methods that neuregulin treatment of cells leads to ErbB2:ErbB3 heterodimer formation.1 In a reciprocal experiment, ErbB3 was detected on ErbB2-specific capture beads (Fig. 6B). Although the signal above background (untreated cell lysate) was lower, it was significant (3-fold signal increase). Note that there are differences in assay reciprocity as well as variability in the assay background for the different microbead sets for detection of ErbB2 and Erb3 in Figure 6. These differences could be attributed to the differences in reactivity of the antibodies coated on microbeads as well as differences in the 2 detection antibodies. Nevertheless, it is important to note that the signal for ErbB3 detection on ErbB2-specific beads (and vice versa) was observed only in lysates obtained from activated cells. Taken together, the above results indicate that ErbB2 and ErbB3 RTKs actively engaged in heterodimer formation in the MDA-MB-453 cell line when ErbB3 was stimulated. The MDA-MB-468 cell line does not express ErbB2 receptor.16,17 ErbB2:ErbB3 heterodimers were not detected in this cell line (these authors, data not shown).
Fig. 6.
Detection of heterodimerization of ErbB family of receptors. Cells were used as untreated (open bars), treated with 6.6 mM sodium pervanadate for 5 min (hashed bars), 16.5 nM EGF (closed bars), or 1:500 neuregulin for 7 min (reverse hashed bars). A second antibody (biotinylated), either specific to ErbB2 (A) or ErbB3 (B), was used for detection of the respective proteins. Treatment of cells by pervanadate, EGF, or neuregulin is indicated. Error bars represent standard deviation error of n = 4 values. Significant difference (P value < 0.01) in detection signal for ErbB2 and ErbB3 (between untreated control and activated cell lysates) is indicated by an encircled asterisk.
Discussion
To understand molecular mechanisms underlying the role of the ErbB family of RTKs in cancer, it is important not only to detect expression but also phosphorylation and heterodimerization of these RTKs.18,19 Importantly, there is increasing evidence that interplay between the 4 RTKs contributes to more aggressive phenotype and affects response to therapy in breast cancer.18 A multi-pronged approach, enabling determination of expression as well as function of these potent signaling molecules for drug screening, use as biomarkers in cancer diagnosis/prognosis and monitoring efficacy of therapy, would be more valuable. In this article, we present proof-of-concept studies for the use of the multiplex microbead suspension array method, in 3 multiplex panels that enable detection of expression, phosphorylation, and heterodimerization of ErbB RTKs.
For the detection of expression of RTKs in breast cancer cell lines (MDA-MB-453 and MDA-MB-468) and an epidermoid cell line (A431), multiplex microbead suspension array yielded results similar to those obtained by western blot (Fig. 1). As expected, the MDA-MB-453 cell line expressed ErbB2 and ErbB3, and the MDA-MB-468 cell line expressed EGFR and ErbB3. A431 cells expressed 3 RTKs (EGFR, ErbB2, and ErbB3). These RTKs are activated by EGF-like growth factors that promote receptor-mediated homo- and heterodimers.11 Among ErbB family members, ErbB2 does not have a known growth-factor ligand, whereas ErbB3 has a defective kinase activity but retains the capacity to bind neuregulin.20 Therefore, these 2 ErbB receptors must heterodimerize with each other, or with other members of this RTK family to transmit signals. These protein–protein interactions may vary from cell type to cell type.4,20 Neuregulin leads to ErbB2-ErbB3 heterodimerization, which subsequently produces biological effects.12,20,21 Indeed in MDA-MB-453 cells activated with neuregulin, ErbB2-ErbB3 heterodimers were readily detected by the multiplex assay (Fig. 6). Similarly, ErbB2-ErbB3 heterodimers were found in A431 and prostate cancer cell lines (data not shown). In the cell lines used in this study, EGFR-ErbB3 or EGFR-ErbB2 heterodimers were not detected. The simplest explanation for this observation is that the predominant form of activated EGFR may be a homodimer.
Dimerization of ErbB RTKs induces tyrosine kinase catalytic activity, which leads to the autophosphorylation of tyrosine residues at the C-terminus of the kinase.22 These phosphorylated residues serve as docking sites for recruitment of proteins, which activate downstream signaling cascades,6,12 including Ras-Raf–mitogen-activated protein kinase (Ras–MAPK), phosphatidylinositol-3 kinase–protein kinase B (PI3K–PKB/Akt, and phospholipase C–protein kinase C (PLC–PKC) pathways.22,23 We have developed a multiplex assay for the simultaneous detection of phosphorylation for efficient phosphoproteomic profiling of ErbB RTKs under different cell activation conditions. In MDA-MB-453 cells, neuregulin treatment resulted in the phosphorylation of ErbB3. Because EGFR is not present in this cell line, EGF treatment did not result in detectable levels of phosphorylation of RTKs (Fig. 3). In contrast, MDA-MB-468 cells displayed a strong increase in phosphorylation of EGFR upon treatment with EGF (Fig. 2). In addition, some increase in the phosphorylation of ErbB3 was also observed. EGF treatment of A431 cells also resulted in a robust increase in phosphorylation of EGFR (Fig. 4). However, despite the high level of ErbB3 expression in these cells (Fig. 1), treatment with neuregulin did not result in phosphorylation of ErbB3. This is surprising because, in pervanadate-treated A431 cells, ErbB3 was found to be heavily phosphorylated (Fig. 4). A simple explanation is that in A431 cell line EGFR sequesters ErbB2, and under the conditions of activation with neuregulin, ErbB2 is not available to dimerize with ErbB3. Thus, ErbB3 phosphorylation is inhibited. However, pervanadate is a nonspecific activator that acts by inhibiting tyrosine phosphatases leading to general hyperphosphorylation of tyrosine-phosphorylated signaling proteins. Therefore, in A431 cells treated with pervanadate, phosphorylation of ErbB3 is presumably not ErbB2-dependent.
To illustrate the efficiency of the multiplex microbead suspension array system in studying intracellular signaling, one may consider the example of phosphoproteomic profiling of RTKs by other methods. Phosphoproteomic analysis of the 4 RTKs in breast cancer cell lines needed approximately 8 × 104 cells for the multiplex microbead assay (Fig. 1). Importantly, the same number of cells would suffice for the phosphoproteomic analysis of several other downstream signaling proteins simultaneously with ErbB RTKs to obtain more detailed information on intracellular signaling (downstream signaling studies in breast cancer to be published elsewhere).13 In contrast, to obtain similar information by IP/WB and ELISA analyses of ErbB RTKs, much larger number of cells (4 × 107 and 8 × 106, respectively) were required (Figs. 2–4; ELISA data (these authors) not shown). Similarly, expression of a wide variety of cell signaling proteins could be performed in a relatively small number of cells (8 × 104 cells). Thus, the need for only a small amount of sample for profiling of several signaling proteins (theoretically up to 100) is a clear advantage of the multiplex microbead assay system.
Utility and small-sample requirement of this novel multiplex immunoassay for detection of phosphorylation were further demonstrated by the analysis of phosphorylation of ErbB RTKs in a time-course activation of MDA-MB-453, MDA-MB-468, and A431 cell lines. Cells were activated by both agonists (EGF and neuregulin) and samples were collected at 7 time points over a period of 1 h for the phosphoproteomic analysis (56 data points) (Fig. 5). The kinetic analysis highlighted the following features of RTK activation: (a) the assay readily enabled investigation of temporal changes in phosphorylation of several proteins simultaneously, involving multiple time points, (b) activation kinetics of EGFR in the 2 cell lines that expressed it (MDA-MB-468 and A431) were similar, displaying a continuous increase in overall tyrosine phosphorylation over 60 min, and (c) the assay was convenient for monitoring signal increase as well as its decay (ErbB3). In comparison to fixed formats such as peptide array systems,24 the multiplex microbead format offers a flexibility and ease of adaptability where microbead sets coated with capture antibodies can be included or excluded from the mixture at will. Methods based on 2-D gel electrophoresis and mass spectroscopy have recently been applied for simultaneous analysis of multiple signaling proteins in cells.25–29 However, these methods require complex protocols for sample analysis as well as complicated and very costly instrumentation for biological and clinical applications.
This study outlines an efficient approach for the determination of expression, phosphorylation, and identification of heterodimeric partner(s), performed on the Luminex platform. As shown earlier, results obtained by the multiplex immunoassays can be quantitatively compared across different cell types for each protein individually. For example, the relative expression levels of individual ErbB RTKs in 3 different cell lines are clearly reflected by their respective MFI values (Fig. 1). Similarly, phosphorylation levels of each of the RTKs in 3 different cell lines are shown by their respective MFI values (Fig. 2–4).
In conclusion, the studies reported here provide proof-of-concept for the use of the multiplex microbead suspension array as a highly efficient method for simultaneous detection of expression, phosphorylation, and heterodimerization of the ErbB RTKs in cancer cells. This approach is relatively high throughput and quantitative and enables the study of critically important attributes of ErbB RTKs in cancer. The results presented here demonstrate the feasibility of the multiplex suspension array approach for analysis of molecular mechanisms by which RTKs exert their oncogenic effects. This approach can also be used for the simultaneous detection and quantitation of expression, phosphorylation, and heterodimerization of many other signaling proteins, and hence the multiplex microbead suspension array system holds promise as a novel method for drug screening, cancer diagnosis, and the prediction of outcomes in cancer patients.
Abbreviations
RTK
receptor tyrosine kinase
PBS
phosphate buffered saline
Sulfo-NHS
N-hydroxysulfosuccinamide
EDC
1-ethyl-3-[3-dimethylaminopropyl] carbodiimide
RT
room temperature
ECL
enhanced chemiluminescence
Acknowledgments
We thank the UC Davis Cancer Center and the Department of Pathology and Laboratory Medicine at UC Davis for support.
Author Disclosure Statement
I.H.K., J.Z., P.G., M.Z., C.S., H.-J.K., and P.A.L. are employees of the University of California Davis.
References
- 1.Stern DF. ERBB3/HER3 and ERBB2/HER2 duet in mammary development and breast cancer. J Mammary Gland Biol Neoplasia. 2008;13:215–223. doi: 10.1007/s10911-008-9083-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lo HW. Hsu SC. Hung MC. EGFR signaling pathway in breast cancers: from traditional signal transduction to direct nuclear translocalization. Breast Cancer Res Treat. 2006;95:211–218. doi: 10.1007/s10549-005-9011-0. [DOI] [PubMed] [Google Scholar]
- 3.Ono M. Kuwano M. Molecular mechanisms of epidermal growth factor receptor (EGFR) activation and response to gefitinib and other EGFR-targeting drugs. Clin Cancer Res. 2006;12:7242–7251. doi: 10.1158/1078-0432.CCR-06-0646. [DOI] [PubMed] [Google Scholar]
- 4.Sweeney C. Miller JK. Shattuck DL. Carraway KL. ErbB receptor negative regulatory mechanisms: implications in cancer. J Mammary Gland Biol Neoplasia. 2006;11:89–99. doi: 10.1007/s10911-006-9015-3. [DOI] [PubMed] [Google Scholar]
- 5.Agrawal A. Gutteridge E. Gee JM. Nicholson RI. Robertson JF. Overview of tyrosine kinase inhibitors in clinical breast cancer. Endocr Relat Cancer. 2005;12(Suppl 1):S135–S144. doi: 10.1677/erc.1.01059. [DOI] [PubMed] [Google Scholar]
- 6.Niu G. Carter WB. Human epidermal growth factor receptor 2 regulates angiopoietin-2 expression in breast cancer via AKT and mitogen-activated protein kinase pathway. Cancer Res. 2007;67:1487–1493. doi: 10.1158/0008-5472.CAN-06-3155. [DOI] [PubMed] [Google Scholar]
- 7.Engel RH. Kaklamani VG. HER2-Positive breast cancer: current and future treatment strategies. Drugs. 2007;67:1329–1341. doi: 10.2165/00003495-200767090-00006. [DOI] [PubMed] [Google Scholar]
- 8.Johnston JB. Navaratnam S. Pitz MW. Maniate JM. Wiechec E. Baust H. Gingerich J, et al. Targeting the EGFR pathway for cancer therapy. Curr Med Chem. 2006;13:3483–3492. doi: 10.2174/092986706779026174. [DOI] [PubMed] [Google Scholar]
- 9.Leong TY. Leong AS. Controversies in the assessment of HER-2: more questions than answers. Adv Anat Pathol. 2006;13:263–269. doi: 10.1097/01.pap.0000213043.16200.92. [DOI] [PubMed] [Google Scholar]
- 10.Britsch S. The neuregulin-I/ErbB signaling system in development and disease. Adv Anat Embryol Cell Biol. 2007;190:1–65. [PubMed] [Google Scholar]
- 11.Stortelers C. van der Woning SP. Jacobs-Oomen S. Wingens M. van Zoelen EJ. Selective formation of ErbB-2/ErbB-3 heterodimers depends on the ErbB-3 affinity of epidermal growth factor-like ligands. J Biol Chem. 2003;278:12055–12063. doi: 10.1074/jbc.M211948200. [DOI] [PubMed] [Google Scholar]
- 12.Graus-Porta D. Beerli RR. Daly JM. Hynes NE. ErbB-2, the preferred heterodimerization partner of all ErbB receptors, is a mediator of lateral signaling. EMBO J. 1997;6:1647–1655. doi: 10.1093/emboj/16.7.1647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Abd El-Rehim DM. Pinder SE. Paish CE. Rampaul RS. Balmey RW. Robertson JFR, et al. Expression and co-expression of the members of the epidermal growth factor receptor (EGFR) family in invasive carcinoma. Br J Cancer. 2004;91:1532–1542. doi: 10.1038/sj.bjc.6602184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Normanno N. Maiello MR. De Luca A. Epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs): Simple drugs with a complex mechanism of action? J Cell Phys. 2002;194:13–19. doi: 10.1002/jcp.10194. [DOI] [PubMed] [Google Scholar]
- 15.Khan IH. Mendoza S. Rhyne P. Ziman M. Tuscano J. Eisinger D, et al. Multiplex Analysis of intracellular signaling pathways in lymphoid cells by microbead suspension arrays. Mol Cell Proteomics. 2006;5:758–768. doi: 10.1074/mcp.T500032-MCP200. [DOI] [PubMed] [Google Scholar]
- 16.Crovello CS. Lai C. Cantley LC. Carraway KL., III Differential signaling by the epidermal growth factor-like growth factors neuregulin-1 and neuregulin-2. J Biol Chem. 1998;273:26954–26961. doi: 10.1074/jbc.273.41.26954. [DOI] [PubMed] [Google Scholar]
- 17.Lupu R. Colomer R. Kannan B. Lippman ME. Characterization of a growth factor that binds exclusively to the erbB-2 receptor and induce cellular responses. Proc Natl Acad Sci USA. 1992;89:2287–2291. doi: 10.1073/pnas.89.6.2287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zaczek A. Brandt B. Bielawski KP. The diverse signaling network of EGFR, Her2, HER3 and HER4 tyrosine kinase receptors and the consequences of therapeutic approaches. Histol Histopathol. 2005;20:1005–1015. doi: 10.14670/HH-20.1005. [DOI] [PubMed] [Google Scholar]
- 19.Anido J. Matar P. Albanell J. Guzman M. Rojo F. Arribas J, et al. ZD1839, a specific epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, induces the formation of inactive EGFR/HER2 and EGFR/HER3 heterodimers and prevents heregulin signaling in HER2-overexpressing breast cancer cells. Clin Cancer Res. 2003;9:1274–1283. [PubMed] [Google Scholar]
- 20.Citri A. Yarden Y. EGF-ERBB signalling: towards the systems level. Nat Rev Mol Cell Biol. 2006;7:505–516. doi: 10.1038/nrm1962. [DOI] [PubMed] [Google Scholar]
- 21.Karamouzis MV. Badra FA. Papavassiliou AG. Breast cancer: the upgraded role of HER-3 and HER-4. Int J Biochem Cell Biol. 2007;39:851–856. doi: 10.1016/j.biocel.2006.11.017. [DOI] [PubMed] [Google Scholar]
- 22.Jimeno A. Hidalgo M. Pharmacogenomics of epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors. Biochim Biophys Acta. 2006;1766:217–229. doi: 10.1016/j.bbcan.2006.08.008. [DOI] [PubMed] [Google Scholar]
- 23.Chan SK. Hill ME. Gullick WJ. The role of the epidermal growth factor receptor in breast cancer. J Mammary Gland Biol Neoplasia. 2006;11:3–11. doi: 10.1007/s10911-006-9008-2. [DOI] [PubMed] [Google Scholar]
- 24.Reimer U. Reineke U. Schneider-Mergener J. Peptide arrays: from macro to micro. Curr Opin Biotechnol. 2002;13:315–320. doi: 10.1016/s0958-1669(02)00339-7. [DOI] [PubMed] [Google Scholar]
- 25.Imam-Sghiouar N. Laude-Lemaire I. Labas V. Pflieger D. Le Caer JP. Caron M, et al. Subproteomics analysis of phosphorylated proteins: application to the study of B-lymphoblasts from a patient with Scott syndrome. Proteomics. 2002;2:828–838. doi: 10.1002/1615-9861(200207)2:7<828::AID-PROT828>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- 26.Steinberg TH. Agnew BJ. Gee KR. Leung WY. Goodman T. Schulenberg B, et al. Global quantitative phosphoprotein analysis using Multiplexed Proteomics technology. Proteomics. 2003;3:1128–1144. doi: 10.1002/pmic.200300434. [DOI] [PubMed] [Google Scholar]
- 27.Brill LM. Salomon AR. Ficarro SB. Mukherji M. Stettler-Gill M. Peters EC. Robust phosphoproteomic profiling of tyrosine phosphorylation sites from human T cells using immobilized metal affinity chromatography and tandem mass spectrometry. Anal Chem. 2004;76:2763–2772. doi: 10.1021/ac035352d. [DOI] [PubMed] [Google Scholar]
- 28.Veenstra TD. Prieto DA. Conrads TP. Proteomic patterns for early cancer detection. Drug Discov Today. 2004;9:889–897. doi: 10.1016/S1359-6446(04)03246-5. [DOI] [PubMed] [Google Scholar]
- 29.Ficarro SB. Salomon AR. Brill LM. Mason DE. Stettler-Gill M. Brock A, et al. Automated immobilized metal affinity chromatography/nano-liquid chromatography/electrospray ionization mass spectrometry platform for profiling protein phosphorylation sites. Rapid Commun Mass Spectrometry. 2005;19:57–71. doi: 10.1002/rcm.1746. [DOI] [PubMed] [Google Scholar]