Arrested B Lymphopoiesis and Persistence of Activated B Cells in Adult Interleukin 7−/− Mice (original) (raw)
Abstract
Interleukin 7 is a crucial factor for the development of murine T and B lymphocytes. We now report that, in the absence of interleukin 7, B lymphocyte production takes place exclusively during fetal and perinatal life, ceasing after 7 wk of age. In peripheral organs, however, the pool of B lymphocytes is stable throughout adult life and consists only of cells that belong to the B1 and marginal zone (MZ) compartments. This is accompanied by a 50-fold increase in the frequency of immunoglobulin (Ig)M- and IgG-secreting cells, and the concentration of serum immunoglobulins is increased three- to fivefold. Both the MZ phenotype and the increase in serum IgM are T cell independent. These findings reveal a previously undescribed pathway of B lymphopoiesis that is active in early life and is interleukin 7 independent. This pathway generates B1 cells and a normal sized MZ B lymphocyte compartment.
Keywords: B lymphocyte, interleukin 7, marginal zone B cells, B1 cells, B cell development
Introduction
Murine B lymphocyte development initially takes place in the fetal liver, shifting after birth to the bone marrow, where it occurs throughout adult life. Precursor populations are phenotypically well characterized and expression of various surface markers allowed the subdivision of precursor B cells into sequentially developing populations 1.
During B lymphocyte development in the bone marrow, IL-7, produced by stromal cells, plays a crucial role in the expansion and survival of those precursors 2. The identification of the receptor complex for IL-7 revealed that it is composed of a private α chain, IL-7Rα 3, and the common γ chain (γc), shared with other cytokine receptor complexes 4. Severe lymphopenia occurs in mice made deficient for either IL-7 5 or IL-7Rα 6, demonstrating the fundamental role played by this receptor/ligand system in lymphopoiesis. Both mutant strains of mice display a 10-fold reduction in the number of peripheral T and B cells, but their phenotype does not seem to be identical. Thus, IL-7Rα–/– mice show an arrest in B cell development at the transition from Hardy's fraction 1 A to fraction B 6, while the blockage in B cell development in IL-7−/− mice was described as occurring from fractions B/C to D 5.
The final outcome of this process of lymphopoiesis is a peripheral B lymphocyte compartment consisting of a majority of small resting cells, but containing also around 10% of activated cells 7. A defined population of activated B cells are the marginal zone (MZ) cells which exhibit a phenotype characterized by the expression of high levels of IgM and CD21 on their surface, together with low levels of IgD and CD23. This is in contrast to the majority 90–95% of follicular (FO) B lymphocytes which display an opposite phenotype (IgDhiCD23hiCD21lo), consistent with a resting state. Multireactive cells, with “sticky” antigen receptors, appear to be recruited efficiently into the MZ and respond promptly to stimulation by LPS or CD40 ligation whereas FO B lymphocytes respond to B cell receptor (BCR)-dependent stimulation, and are thought to be responsible for the production of high affinity antibodies and memory cells 8 9. A distinct subpopulation of B lymphocytes displaying activation markers, B1 cells, is enriched in the peritoneal cavity and appears to derive from precursors generated early in development 10 11. These B1 cells were shown to express Ig receptors with fewer N sequence additions than the conventional B2 cells 12 13 and preferentially secrete auto- and multireactive antibodies 14.
In the present work we examine the development, persistence, and activation of B lymphocytes in mice deficient for IL-7. We show that the bulk of B lymphocytes in IL-7−/− mice has a fetal and perinatal origin, because, contrary to previously held beliefs, B cell development in adult bone marrow is arrested at a very early stage, before the transition to fraction B. In agreement with this finding, the majority of B lymphocytes found in the spleen of adult IL-7−/− mice carry little or no N sequence additions at their VDJ borders. Nevertheless, the pool of peripheral, mature B lymphocytes in these mice is stable in numbers, at some 10% of control levels, and consists predominantly of large activated cells with a phenotype closely resembling that of MZ cells. This phenotype is observed even when thymus-derived lymphocytes are absent, as shown by the analysis of athymic nu/nu, IL-7−/− mice. Furthermore, the lymphopenia caused by the lack of IL-7 affects neither the B1 population nor the levels of serum Ig, as IL-7−/− mice have in fact more circulating antibodies than wild-type controls.
Materials and Methods
Mice.
The IL-7–deficient mice were derived from chimeras obtained by microinjecting the original IL-7 targeted ES cell line 5 in C57BL/6 blastocysts. The mice used in this study were backcrossed with C57Bl/6 for at least six generations. Nu/nu, IL-7−/− mice were obtained by crossing fully inbred (N10) IL-7−/− mice to C57BL/6J nu/nu animals obtained from The Jackson Laboratory. C57BL/6 mice were used as wild-type controls and were originally purchased from The Jackson Laboratory. All mice were bred and kept in our animal facilities in Oeiras under specific pathogen-free (SPF) conditions.
Cell Purification and Counting.
Single cell splenocyte suspensions were obtained by disrupting spleens with a nylon mesh in FACS® medium (PBS, 2% FCS, 0.02% NaN3). Bone marrow cells were obtained by flushing femurs with 2 ml of FACS® medium using a 27 1/2 gauge needle. Peritoneal cavity exudate cells were collected by injecting peritoneal cavities with 10 ml of FACS® medium using a 27 1/2 gauge needle. After a gentle massage the liquid was recovered with a 20 gauge needle. Cells were counted in a Burker chamber under a light microscope excluding dead cells with Trypan blue (Sigma-Aldrich).
Antibodies, Staining, and FACS® Analysis.
FITC-, PE-, allophycocyanin (APC)-, or biotin-conjugated antibodies against B220 (RA3–6B2), CD11b/Mac-1 (M1/70), CD19 (1D3), CD21 (7G6), CD23 (B3B4) heat stable antigen (HSA) (M1/69), CD43 (S7), BP-1 (6C3), and IgD (11–26c.2a) were purchased from BD PharMingen. Polyclonal, μ chain specific, FITC-labeled anti-IgM was obtained from Pierce Chemical Co. and PE-conjugated monoclonal anti-IgM antibody 1B4B1 was obtained from Southern Biotechnology Associates, Inc. 106 cells per well were incubated with 25 μl of pretitrated antibody in 96-well plates (Costar) on ice for 20 min in the dark. The cells were then washed three times with FACS® medium before analysis. Biotinylated antibodies were revealed with APC-, PE-, or FITC-conjugated streptavidin (BD PharMingen), after washing twice. Samples were analyzed on FACScan™ or FACSCalibur™ flow cytometers (Becton Dickinson), and dead cells were excluded with propidium iodide.
Analysis of B Cell Precursor Frequencies.
Bone marrow precursor cells (B220+IgM− or CD19+IgM−) were purified by cell sorting, using a FACStarPLUS™ (Becton Dickinson), to a purity >98%. The cells were then plated under limiting dilution conditions in 96-well plates (48 wells/dilution) at 100, 33, and 11 cells/well (in the case of B220+IgM−) or 10, 3.3, and 1.1 cells/well (in the case of (CD19+IgM−) onto irradiated (2,000 Gy) S17 stromal cells in culture medium (Opti-MEM [GIBCO BRL]), with 10% FCS, Pen/Strep, and βM-E), supplemented with excess IL-7 and Stem cell factor (c-kit ligand), as described previously 15. Growth of lymphocyte colonies was scored at day 7 of culture.
CDR3 Cloning and Sequencing.
Cell separation was performed by midiMACS magnetic sorting, according to the manufacturer's protocol (Miltenyi Biotec). CD19+ cells from the spleen of IL-7−/− and C57/Bl6 mice were purified using FITC-conjugated anti-CD19 antibody and anti-FITC MicroBeads (Miltenyi Biotec). Purity was >95%, as verified by FACS® analysis. DNA from 5 × 106 CD19+ cells was obtained by incubating at 37°C overnight in DNA quick lysis solution (50 mM Tris-HCl, pH 7.6, 1 mM EDTA, 100 mM NaCl, 0.2% SDS) containing 100 μg/ml proteinase K, precipitated with 3 volumes of ice cold absolute ethanol and 0.1 volumes of 5 M NaCl, washed in 70% ethanol, and resuspended in milliQ water. J558 rearrangements were amplified with the following primers: VHJ558 AAGGCCACACTGACTGTAGAC (sense), and Cμ CTGGATCCGGCACATGCAGATCTC. PCR was performed with 1.5 μM MgCl2, 0.2 μM dNTP's, 0.2 μM of each primer, and 0.25 μl of 5 U/μl Taq polymerase using manufacturer's buffer (Life Technologies). Reactions were incubated at 94°C for 2 min, and then run through 35 cycles of 94°C for 1 min, 60°C for 1 min, and 72°C for 1 min, with a final extension of 10 min at 72°C. A 750-bp band, corresponding to J558-JH4 rearrangements from this reaction was purified from a 0.8% agarose gel by gel extraction (QIAGEN). Purified fragments were then cloned using the TA cloning kit from Invitrogen according to the manufacturer's instructions. Ampicilin resistant clones were screened by PCR for recombinants, and positive clones were sequenced using BigDye Terminator Cycle-sequencing kit (Applied Byosystems), with 3.2 pM of either Sp6 primer, ATTTAGGTGACACTATA, or M13(-20) forward primer, GTAAAACGACGGCCAG, depending on insert orientation. Sequencing was performed on an ABI Prism 377 (PerkinElmer) automatic sequencer, and the results were analyzed with Sequencher software (Gene Codes).
ELISA and ELISA Spot Assay.
Sera were obtained from tail bleeding and kept at –20°C until used. Total IgM, IgG, and IgA concentrations were determined by ELISA as described elsewhere 16 using monoclonal antibodies of each class as standards. Results are presented as μg/ml equivalents of the standard protein. Total IgM and IgG plasma cell numbers in ex vivo experiments were determined by a modification of the method described elsewhere 16. Briefly, flat-bottom 96-well plates (Luxlon M29 LSE; CEB) were coated with anti-IgM or anti-IgG antibodies (Southern Biotechnology Associates, Inc.). The plates were blocked with 1% gelatin for 1 h, and the cells were seeded according to a serial dilution and incubated at 37°C overnight. After washing, the spots generated by the binding of secreted Ig were revealed with alkaline phosphatase–conjugated anti–mouse IgM or IgG (Southern Biotechnology Associates, Inc.) for 90 min at 37°C. The development of an alkaline phosphatase reaction was obtained by the use of the substrate 5-bromo-4-chloro-3-indolyl phosphate (Sigma-Aldrich). The fraction of plasma cells per cells seeded was determined by counting the number of spots in at least three different dilutions. When evaluating the number of plasma cells in various organs, the number of spots was further adjusted according to the number of B cells as determined by FACS® analysis.
Results
B Lymphocytes Do Not Accumulate with Age in IL-7−/− Mice.
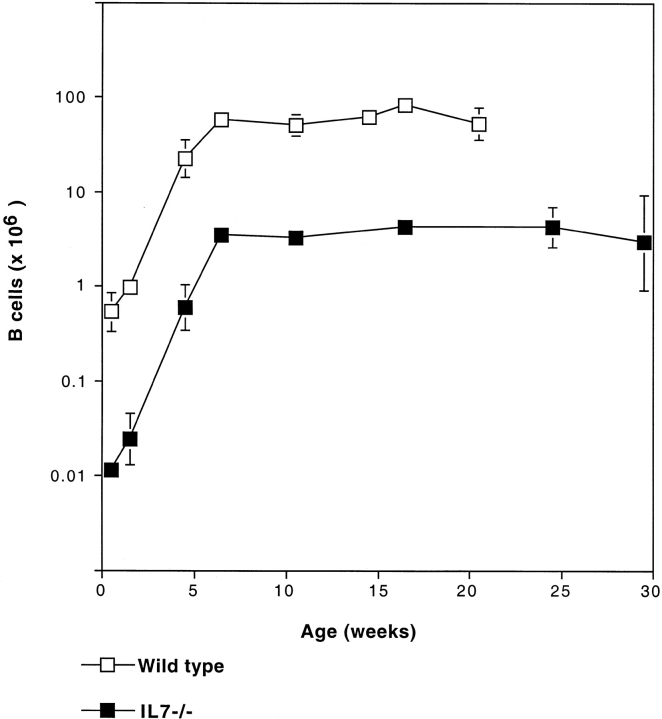
Deficiency in IL-7 leads to a severe reduction in the number of both T and B lymphocytes in peripheral organs 5. To examine whether the lymphopenia in the B cell compartment persists throughout the life of IL-7−/− mice we determined the number of B220+IgM+ cells present in the spleen, from birth until 7 mo of age. Fig. 1 shows that the number of splenic B lymphocytes found in IL-7−/− and control mice increases exponentially only until the age of 6 wk, and from then on remains constant. At all ages tested the number of B lymphocytes in the spleen of IL-7−/− mice is reduced by ∼10-fold when compared with age-matched wild-type controls.
Figure 1.
Adult IL-7−/− mice do not accumulate B cells in the periphery. Spleen cell suspension from C57BL/6 and IL-7−/− mice were prepared at the indicated ages and the number of live B220+IgM+ B cells was determined. The figure shows the logarithmic mean and standard deviations of three to five animals per time point.
Bone Marrow Lymphopoiesis Is Blocked at an Early Stage in IL-7−/− Mice.
The observation that IL-7−/− mice cease to accumulate B lymphocytes at around 6 wk of age, maintaining a severe lymphopenia throughout life, led us to investigate if production of B cells occurs in adult mice, albeit at reduced levels. Development of B lymphocytes from hematopoietic precursors has been subdivided into fractions A (the more immature) to F (the more mature, IgM+IgD+ population) according to the expression of various surface markers 1. IL-7 deficiency is thought to cause a blockade at the transition of precursor B cells from fractions B/C to D 5.
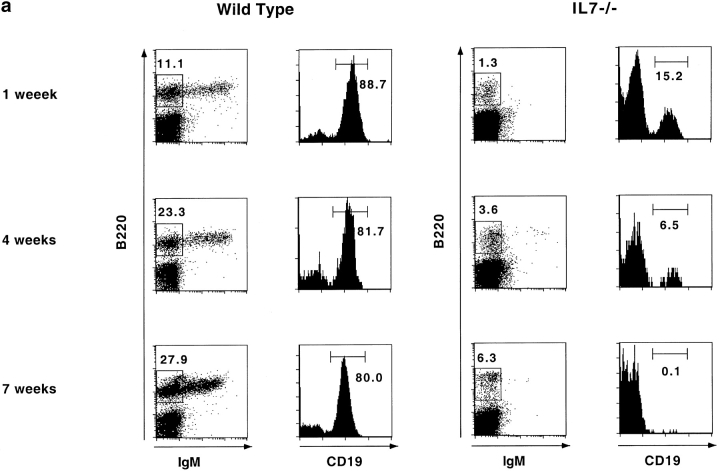
We analyzed the populations of precursor B lymphocytes in IL-7−/− mice by flow cytometry using the marker CD19, which is expressed by B lineage cells after fraction A 17. This surface marker is known to be more specific for precursor B lymphocyte populations than B220 (CD45R), which is also expressed by other cells in the bone marrow, for example NK cells 18.
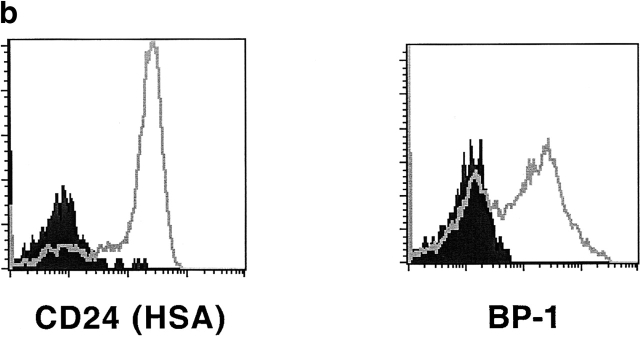
Fig. 2 a shows that the population of B220+ IgM−CD19+ cells, which contains pro- and pre-B cells, easily detectable in the bone marrow of wild-type animals, is much reduced in IL-7−/− mice already at 1 wk of age and by the time the animals reach 7 wk this population is almost completely absent. Because cells in fraction B already express CD19, this result shows that, in contrast to previous convictions, the block in B lymphocyte development observed in adult IL-7−/− mice occurs before the transition to this population. Moreover, as shown in Fig. 2 b, the B220+IgM− population in the bone marrow of adult IL-7−/− mice express neither BP-1 19 nor HSA (HSA/CD24), two other markers found in precursor B lymphocytes 1.
Figure 2.
Bone marrow B lymphopoiesis is arrested in adult IL-7−/− mice. (a) Expression of B220, IgM, and CD19 by wild-type (left) and IL-7−/− (right) bone marrow cells. Dot plots show B220/IgM stainings and the B220+IgM− gate used for histogram analysis. Histograms show the percentage of CD19+ cells in the B220+IgM− population. Numbers represent percentage of live lymphoid cells in the bone marrow. The average number of cells recovered from the femurs of wild-type and IL-7−/− were the following: wild-type 6.106 (± 2.3), 25 × 106 (± 7.0), and 30.8 × 106 (± 4.5); IL-7−/− 4.8 × 106 (± 0.5), 12.6 × 106 (± 4.5), and 19.4 × 106 (± 6.3), at 1, 4, and 7 wk of age, respectively. Results are representative of at least three mice for each age. (b) Expression of CD24 (HSA) and BP-1 among B220+IgM− (left histogram) and B220+ (right histogram) cells in the bone marrow of wild-type (open) and IL-7−/− (filled) mice.
Finally, as shown in Table , the frequency of B cell precursors in either CD19+IgM− or B220+IgM− cells purified from the bone marrow of young mice is comparable in control and in IL-7–/– mice (1/6 vs. 1/4 and 1/50 vs. 1/80, respectively). In contrast, in the case of adult mice (where there are no CD19+IgM− in IL-7–/– mice) this frequency among the B220+IgM− population is 1/240 in control mice, but below 1/6,500 for IL-7–/– animals. These results show that B lymphopoiesis in the bone marrow of IL-7–/– mice is already reduced some 40-fold in the total number of B cell precursors at 1 wk of age. At 7 wk of age both the absence of CD19+ cells in the bone marrow and the undetectable frequency of B cell precursors in vitro indicate a virtual absence of B lymphopoiesis.
Table 1.
Frequency of B Cell Precursors Among Bone Marrow Cells
| Age | Population | C57BL/6 | IL-7−/− |
|---|---|---|---|
| 1 wk old | CD19+IgM− | 1/6 | 1/4 |
| B220+IgM− | 1/50 | 1/80 | |
| 8–10 wk old | B220+IgM− | 1/240 | <1/6,500 |
B Lymphocytes in IL-7−/− Mice Do Not Express an Adult Repertoire.
Variable region genes in B lymphocytes of young mice characteristically lack N sequence additions 12 20 21 due to the developmental regulation of the enzyme TdT 22. Because of the block in adult B lymphopoiesis shown above, we determined N nucleotide additions by sequencing junctional segments where VHJ558 rearranged to JH4 in 6-wk-old mice. The full sequence of the J558 locus is not known, so we considered the end of the germline VH J558 segments to be the predominant Cys-Ala-Arg motif. D elements were identified as the longest stretch of nucleotides between the VH and the JH4 genes that showed sequence identity to published D segments.
Table shows 19 such sequences from IL-7−/− and 5 from wild-type mice. All of the wild type sequences showed N additions at both the V-D and the D-J borders, as described previously 12 13. In contrast, of the 19 sequences from IL-7−/− mice analyzed, only 6 showed a similar pattern with more than one N nucleotide at both the V-D and the D–J junctions. In accordance with these results, CDR3 length analysis of VHQ52 family rearrangements revealed a shorter average length in IL-7−/− mice compared with control mice (not shown). We also sequenced VDJ rearrangements in the fetal liver of IL-7−/− and wild-type mice and found no N sequence additions in both cases (not shown).
Table 2.
N-Additions at Ig CDR3
| VH | V/N/P | D | N/P | JH4 |
|---|---|---|---|---|
| C57BL/6 | ||||
| TAC TGT GCA AGA | CAG A | TC TAC TAT GGT AAC TAC | GTG | TAC TAT GCT ATG GAC TAC TGG GGN |
| TAT TGT GCA AGA | GCG GAG GC | C TAT GAT TAC GAC | TAT | TAC TAT GCT ATG GAC TAC TGG GGT |
| TAC TGT GCA AGA | GAG G | AT TAC T | CC T | CT ATG GAC TAC TGG GGT |
| TAC T | CT TCC | ATT ACG AC | G AGA | GGA CTA CTG GGG |
| TAT TGT GCA AGA | GAT AGC TCA | GGC TAC | GTA GAG | GCT ATG GAC TAC TGG GGT |
| IL-7 − **/**− | ||||
| TAT TGT GCA AG | T GG | C TAT GCT ATG GAC TAC TGG GGT | ||
| TAT TGT GCA AG | T AGC TAC | TAC TAT GCT ATG GAC TAC TGG GGT | ||
| TAT TGT GCA AGA | CTA CGG | TAC TAT GCT ATG GAC TAC TGG GGT | ||
| TAC TGT GCA AGA | TAT GGT TAC G | AC TAT GCT ATG GAC TAC TGG GGT | ||
| TAT TGT GCA | N | AT GGT TAC T | AC TAC TGG GGT | |
| TTC TGT GCA AGA | G | AC GGT A | AT GCT ATG GAC TAC TGG GGT | |
| TAC TGT GCA A | CC C | AG | TAT GCT ATG GAC TAC TGG GGT | |
| TAC TGT GCA AGG | G | GG TA | A CTA TGC TAT GGA CTA CTG GGG | |
| TTC TGT GCA A | A | T CTA CTA TGA TTA CGA | T | TA CTA TGC TAT GGA CTA CTG GGG |
| TTC TGT GCA AGG | GGG | TAT TAC TAC GGT AGT AGC T | T | C TAT GCT ATG GAC TAC TGG GGT |
| TTC TGT GCA AGA | AGG | TAC TAC GGT AGT AGC T | C | C TAT GCT ATG GAC TAC TGG GGT |
| TAC TGT GCA AGA | T | CG G | AG | TAT GCT ATG GAC TAC TGG GGT |
| TAC TGT GCA AGA | T | CT ATG ATT ACG AC | G A | AC TAT GCT ATG GAC TAC TGG GGT |
| TAC TGT GCA A | TA | TTA TTA CGG | TCC G | AT GCT ATG GAC TAC TGG GGT |
| TAC TGT GCA AGA | TGG | GGT ACT AC | A GGG GG | T ATG GAC TAC TGG GGT |
| TTC TGT GCA AGA | TCG A | GA TT | C CTG CT | T ATG GAC TAC TGG GGT |
| TAC TGT GCA AGA | GGG AGG AAT TCT A | TC TAT GAT GG | C CAC CCT CT | C TAT GCT ATG GAC TAC TGG GGT |
| TAC TGT GCA AGA | TAC GGT A | AA AAG ACT AT | C TAT GCT ATG GAC TAC TGG GGT | |
| TAC TGT GCA AGA | GGG AC | A GCT CAG GGC CTT | C TAT GCT ATG GAC TAC TGG GGT |
B1 Cells Are Normally Represented in IL-7−/− Mice.
A population of B lymphocytes which express an activated phenotype, named B1 cells, is long-lived, self renewing, and persists independently of adult bone marrow lymphopoiesis 11 23. These cells have little or no N sequence additions at their VDJ borders 13 24 and are enriched in the peritoneal and pleuropericardial cavities, where they express Mac 1 (CD11b) 25 and CD5 (in the case of B1a cells) 26. Splenic B1 cells, on the other hand, are negative for Mac 1, but were described as CD23lowCD43+ 27.
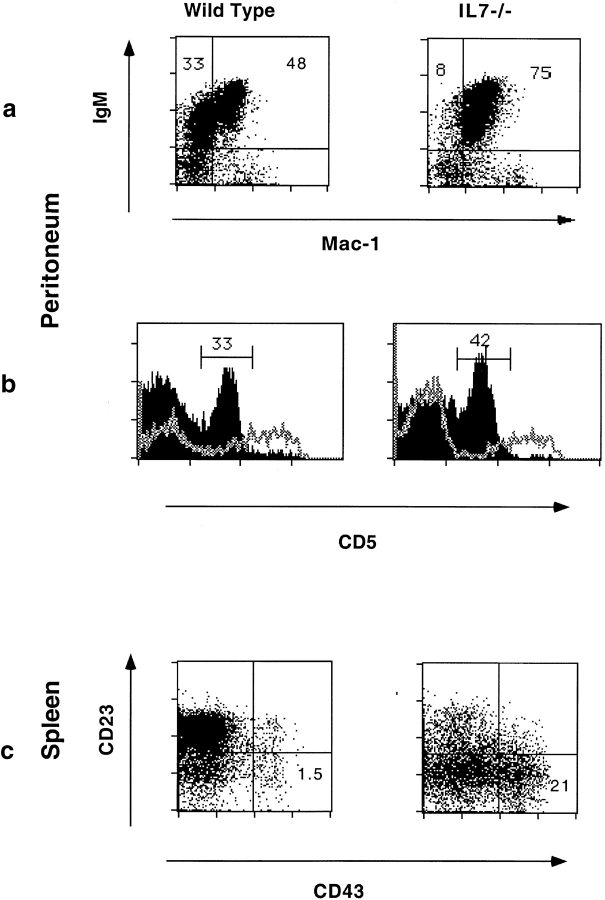
We examined peritoneal B cells by flow cytometric analysis for the expression of Mac 1. Fig. 3 a shows that essentially all the B cells recovered from the peritoneal cavity of IL-7−/− mice are Mac 1+ B1 cells, whereas wild-type mice show a prominent Mac 1− B2 population that represents 40% of the B lymphocytes in the peritoneal cavity. The overall distribution of peritoneal B1a and B1b cells is not significantly altered in IL-7−/− mice, as seen by the proportions of CD5+ and CD5− cells among the Mac 1+ B cells (Fig. 3 b). The lack of B2 cells in the peritoneal cavity of IL-7−/− mice is reflected in the absence of IgMlo–intIgDhi B cells (not shown).
Figure 3.
B1 lymphocytes are present in normal numbers. (a) Percentage of Mac-1+ and Mac-1− B cells (IgM+) in the peritoneal cavity at 8 wk of age. Plots were gated to exclude dead cells, peritoneal macrophages, and granulocytes. (b) CD5 expression on gated peritoneal B1 cells (filled line) and on IgM− cells (open line) from C57BL/6 (left panel) or IL-7−/− (right panel) mice. (c) CD23 and CD43 expression by gated IgM+ cells in the spleen of C57BL/6 (left panel) or IL-7−/− (right panel) mice. The numbers in the panels represent the percentage of CD23lowCD43+ cells.
In the spleen of adult IL-7−/− mice around 15–20% of the B lymphocytes express CD43 and low levels of CD23 (i.e. they are B1 cells). This compares to the 1–2% B1 cells detected in control mice (Fig. 3 c). Because in IL-7−/− mice the reduction in the total number of splenic B cells is 12-fold (see Fig. 1), this result shows that also in the spleen the actual number of B1 cells is similar in IL-7−/− and in wild-type mice.
A High Proportion of Mature Splenic B Cells in IL-7−/− Mice Display an Activated Phenotype.
B cell lymphopenia is frequently associated with altered proportions of peripheral resting and activated cells 28 29. Most of the large activated cells in the spleen are found associated to the macrophage population in the MZ, while most of the small resting B cells localize to the T cell rich areas of the follicles 8. These two populations differentially express various surface markers 30.
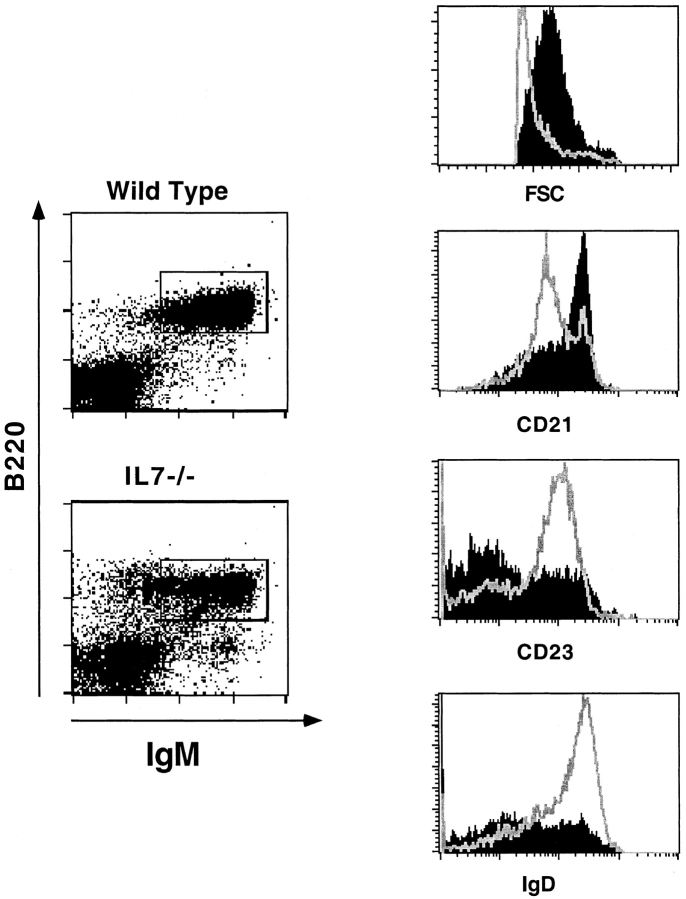
Fig. 4 shows that most B cells (B220+IgM+) from IL-7−/− mice have a higher forward scatter (FSC), express high levels of IgM and CD21 and lower levels of IgD and CD23, when compared with splenocytes from wild-type mice. Splenic B cells from IL-7−/− mice, in addition, respond to LPS at doses as low as 0.25 μg/ml (not shown), which is a characteristic of MZ cells 8. As shown above, only 20% of these cells are CD43+, therefore we conclude that most B lymphocytes present in the spleen of adult IL-7−/− mice are activated cells with a phenotype very similar to that of MZ B cells in normal mice.
Figure 4.
IL-7−/− mice B lymphocytes are predominantly activated cells. Expression of activation markers by splenic B cells of IL-7−/− and wild-type mice at 10 wk of age. Dot plots on the left show the B220+IgM+ gate used for histogram analysis in wild-type (top) and IL-7−/− (bottom) cells. Histograms on the right show from top to bottom the size of B220+IgM+ cells of wild-type (open) and IL-7−/− (filled) mice, measured by FSC, and the expression of CD21, CD23, and IgD. Splenocytes were gated for live lymphoid cells, and the results are representative of more than 20 adult animals analyzed.
In IL-7−/− mice, the reduced number of T lymphocytes present in the spleen also display markers compatible with high levels of activation (unpublished data). We therefore assessed whether the increased B cell activation is a property of the B lymphocyte compartment or rather a consequence of T cell hyperactivity. Fully inbred (N10) IL-7−/− mice were crossed to C57BL/6J nu/nu animals in order to obtain athymic animals deficient for IL-7. In the spleen of such IL-7−/−, nu/nu mice CD3+ T lymphocytes are virtually undetectable, in contrast to the small (1–3%) population of CD3+ cells present in IL-7+/+, nu/nu animals (not shown). As shown in Fig. 5, the majority of the B lymphocytes present in the spleen of IL-7−/−, nu/nu mice consist of large, CD21hiCD23lo cells that resemble MZ B lymphocytes, similarly to what is observed in IL-7−/− mice.
Figure 5.
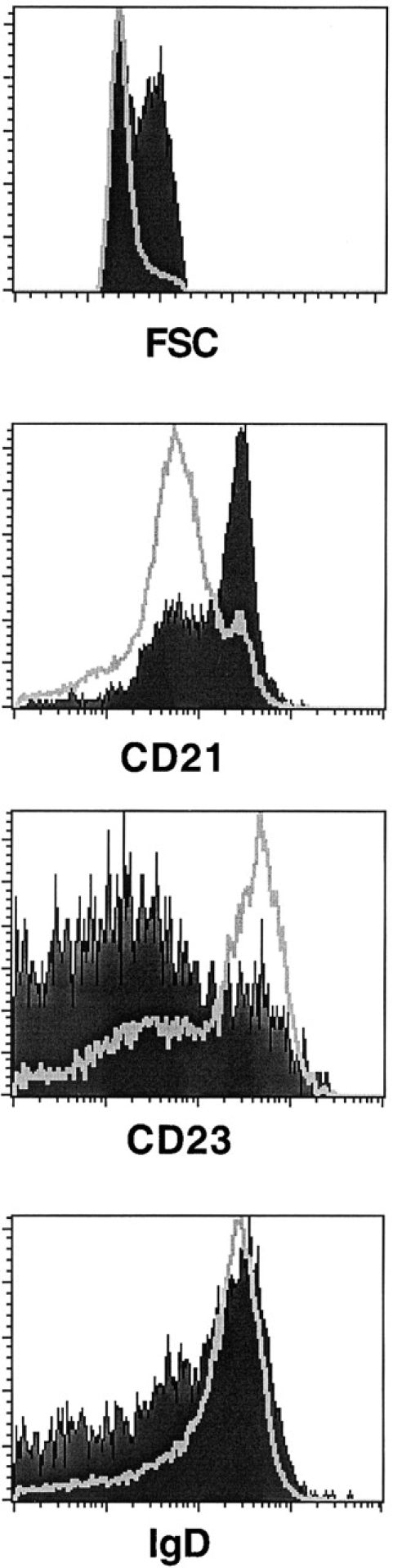
B lymphocytes in IL-7−/− mice are predominantly activated in the absence of thymus-derived cells. Expression of B cell activation markers in splenic B lymphocytes from IL-7−/−, nu/nu and C57BL/6J nu/nu mice at 7 wk of age. The histograms show, from top to bottom, the size of B220+IgM+ cells of C57BL/6J nu/nu (open), and IL-7−/−, nu/nu (filled) mice, measured by FSC, and the expression of CD21, CD23, and IgD. Splenocytes were gated for live B cells as in Fig. 4. The result is representative of two adult animals analyzed.
Plasma Cell Numbers and Serum Antibody Levels in IL-7−/− Mice Are Higher than Normal.
Normal, unimmunized mice maintain a constant level of serum Ig resulting from endogenous activation of B lymphocytes and their differentiation into plasma cells. These natural antibodies are found even in mice kept under germ-free conditions and fed chemically defined diets 7.
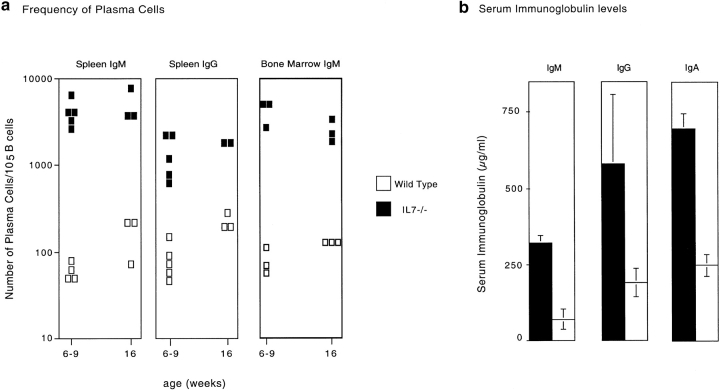
We determined the number of plasma cells in the spleen and bone marrow of IL-7−/− and wild-type mice and measured their levels of serum IgM, IgG, and IgA. ELISA-spot analysis shows a 30- to 50-fold increase in the frequency of IgM- and IgG-secreting plasma cells in the spleen of IL-7−/− compared with control mice. The frequency of bone marrow IgM secreting plasma cells is also increased 50-fold in IL-7–deficient mice (Fig. 6 a). Fig. 6 b shows that IL-7−/− mice have elevated titers of serum antibodies relative to control mice. The concentrations of Ig were fivefold higher for IgM, and threefold higher for IgG and IgA in the serum of IL-7−/− than in wild-type mice. Serum antibody levels remained higher in IL-7−/− mice at least until 4 mo of age. Serum IgM titers were also approximately twofold elevated in IL-7−/−, nu/nu mice, compared with control nu/nu animals (not shown).
Figure 6.
Antibody production and serum Ig levels. (a) Frequency of IgM and IgG plasma cells in the spleen, and of IgM plasma cells in the bone marrow, as determined by ELISA-spot assay in young (6–9 wk) and old (16 wk) mice. Results are displayed as number of antibody-secreting cells per 105 B220+IgM+-positive cells for wild-type (open symbols) and IL-7−/− (closed symbols) mice. (b) IgM, IgG, and IgA serum antibody titers from 6-wk-old IL-7−/− (black bars) and wild-type (white bars) mice. Results show the mean and standard deviation (n = 6).
Discussion
This work shows that in mice deficient for IL-7 the bone marrow ceases to produce significant numbers of B lymphocytes after 7 wk of age. In IL-7−/− mice the percentage of precursor B lymphocytes (B220+IgM−CD19+) decreases from 0.2% of total bone marrow cells at 1 and 4 wk of age to below 0.01% at 7 wk. In wild-type mice, in contrast, the frequency of precursors increases from 10 to 20%. Because the cellularity of the bone marrow is similar in both strains of mice (5; and our observations) this leads to a >1,000× reduction in the number of pro- and pre-B cells in adult IL-7–/– mice. Curiously, B lymphocytes accumulate in the periphery at the same rate and over the same time period as in control mice, suggesting either a more efficient recruitment of B cells into the peripheral pool, or a higher rate of peripheral expansion. In spite of the absence of newly generated B cells, the peripheral compartment maintains constant numbers throughout adult life, at a level ∼10% of normal. Most splenic B lymphocytes in adult IL-7−/− animals carry little or no N-sequence additions at their VDJ borders, which is consistent with their production during fetal and perinatal life, before expression of TdT. In spite of the severe lymphopenia, the mice have higher than normal titers of Ig in their serum and an increased frequency of plasma cells secreting IgM and IgG.
The defect described here resembles that reported for mice deficient in the expression of λ5, where a severe lymphopenia and stunted B cell development are also associated with elevated serum IgM 29. A crucial difference however is that λ5-deficient mice accumulate cells with time and approach normal B cell numbers after 4–6 mo of age. This is not the case in IL-7−/− mice, where B cell numbers always parallel those of wild-type animals, with a reduction of little over one order of magnitude.
The conclusion that the block in B cell development seen in IL-7−/− mice after 7 wk of age occurs at the transition between Hardy's fractions 1 A to B, and not B/C to D as originally described 5, is supported by several pieces of evidence. The population of B220+IgM− cells in the bone marrow of adult IL-7−/− mice does not express the coreceptor molecule CD19, a marker specific for the B cell lineage 17 18. The same population does not express other markers found on precursor B cells, like BP-1 19 and, contrary to the original publication describing the IL-7−/− mice 5, we could not detect expression of HSA/CD24. Although some of the B220+IgM− cells, which are CD19− in IL-7−/− mice, may represent very early B cell precursors as described 17 31, we show that the frequency of cells in this population capable of giving rise to B cells in culture is below 1/6,500 in IL-7−/− mice, whereas it is 1/240 for B220+IgM− isolated from C57BL/6 mice. It is known that B lymphocytes generated during perinatal life have little diversification at the VDJ junctions 12 20 21, therefore the finding that most B cells present in the spleen of adult IL-7−/− mice have little N-sequence additions suggests that they were produced during the fetal and neonatal period. Both IL-3 32 and thymic stromal lymphopoietin (TSLP) 33 are candidate factors for supporting B lymphopoiesis in young mice, but it is evident from the data that they play a minor role, if any, during adult bone marrow lymphopoiesis.
A significant proportion of the B220+IgM−CD19− population expresses the NK 1.1 marker both in IL-7−/− mice (not shown) and in wild-type mice, as reported (18; and not shown). Our results therefore offer another interpretation to the observation that the population in IL-7−/− mice does not express TdT and Cμ 34. Other studies that identify these B220+IgM− cells as B lymphocyte precursors 35 need to be reevaluated in light of the present results.
Splenic B cells in adult IL-7−/− mice display an activated phenotype, in some ways resembling that of B1 cells 11. These cells express CD43 27 and are enriched in the peritoneal cavity, where they also express the Mac 1 marker 25. Most B lymphocytes in the spleen of IL-7−/− mice have very few or no N-sequence additions, similarly to B1 cells 13, and 20% of them are CD43+, indicating that the splenic B1 compartment is of normal size. We find that the severe lymphopenia observed in IL-7−/− mice does not affect the number of peritoneal Mac 1+ B cells and also that the proportions of peritoneal B1a (CD5+) and B1b (CD5−) lymphocytes are normal.
In a normal animal the bulk of B cells are resting, have a small size, and express high levels of IgD, whereas most B lymphocytes in IL-7−/− mice are IgDlow. A predominance of IgDlow cells was reported for IL-7Rα–deficient mice, which was interpreted as representing immature B cells 36. This is not the case in IL-7−/− mice, because most cells are mature activated B lymphocytes (CD21hi CD23loIgDlo and large size as measured by their FSC profile). Approximately 80% of these cells are CD43−, therefore they do not belong to the B1 lineage and their phenotype is very similar to what was reported for MZ B cells 8 although direct histological evidence for the existence of a MZ in IL-7−/− mice is lacking. The B cells in the spleen of IL-7−/− mice also promptly respond to doses of LPS as low as 0.25 μg/ml (not shown), another characteristic of MZ cells 8.
The B cell activation and higher than normal levels of serum Ig and plasma cells in IL-7−/− mice may be the result of disregulation in whatever mechanisms are responsible for controlling the compartment of activated and Ig-secreting peripheral B cells. Evidence for T cell control of the size of the B cell compartment comes from studies conducted in mice made deficient for the common cytokine receptor γ chain 37. It would be possible, therefore, that the alteration observed in the B cell compartment of IL-7−/− mice is secondary to the T cell lymphopenia. However, it is unlikely to be simply the result of it since T cell–deficient mice (nude or T cell receptor knockout mice) do not show this prominent phenotype of B cell activation and high levels of serum Ig. The analysis of IL-7−/− nude mice allowed us to determine that the prominent B lymphocyte activation is T cell independent because, in the absence of thymus derived T cells, the majority of the B cells are still CD21hi and CD23lo. The only difference we observed when comparing IL-7−/−, +/+, and IL-7−/−, nu/nu mice is the appearance of a somewhat larger population, yet not exceeding 40% of the splenic B cells, of small IgDhi lymphocytes (compare Fig. 4 and Fig. 5). It may be that a fraction of the B cells in IL-7−/− mice are activated by T cell–dependent mechanisms, but the B lymphocytes that do develop in the absence of IL-7 still replenish a normal sized MZ compartment and the FO B cell pool is severely depleted, even when thymus-derived cells are absent. Consistent with these results is the observation that also in IL-7−/−, nu/nu mice the level of serum IgM is elevated relative to the level found in C57BL/6J nu/nu (not shown).
The high activation level of the B cells could also be the result of external pathogen pressure on a quantitatively limited immune system. This hypothesis is unlikely because our mice were raised under SPF condition, and because the high frequency of plasma cells in IL-7−/− is already evident in 10-d-old animals (not shown).
Soluble factors encountered during development can modulate activation thresholds and influence cell fate decisions, namely activation and Ig secretion, as shown for type I interferon 38. It is therefore possible that the lack of IL-7 in the periphery leads, directly or indirectly, to B cell activation. However, in a different system, where inducible deletion of recombination activating gene (Rag)-2 stops B cell production in the bone marrow soon after birth, the reduced number of B lymphocytes present in the adult also show a prominent MZ phenotype, and the compartment of peritoneal B1 cells is normally represented 38a. This finding argues that cells with a MZ/B1 phenotype arise in our mice because they were produced early in life, and not as a consequence of development in the absence of IL-7.
Available evidence suggests that strong selection operates to maintain a diverse pool of activated and Ig secreting cells. Analysis of immunodeficient or irradiated hosts reconstituted with small numbers of peripheral B cells shows that even when the total number of B220+IgM+ cells is below normal, the number of activated and Ig secreting B cells, as well as the titer of serum Ig, can reach normal values. Furthermore, a small resting B cell pool is only apparent after the compartment of activated cells is significantly reconstituted 28. Peritoneal B cells of the B1 phenotype are also easily and quickly reconstituted in such models, and give rise at high frequency to IgM and IgA plasma cells 39. Studies conducted in BCR transgenic mice reveal a stepwise selection for nontransgene-expressing cells in the progression from pre-B to mature and activated B to Ig-secreting cells 40, which is taken as evidence that selection forces operate to regulate the compartment of activated and plasma cells. One explanation for the results reported in this paper is therefore that only the MZ and B1 compartments attain full reconstitution as a consequence of the persistent lymphopenia that results from the strict IL-7–dependency of adult bone marrow lymphopoesis.
The finding that the B1 compartment is normally represented even when B cell production is restricted to fetal and perinatal life, lends support to the idea that this subpopulation originates from a distinct developmental pathway 10 23 41. Our data would suggest as well that MZ B cells are, to a significant extent, also derived from precursors that are seeded early in life.
In conclusion, we report an example of an adult mouse where the B lymphocytes originate from an embryonic and perinatal “layer” 42, shown in this work to be independent of IL-7.
Acknowledgments
We wish to acknowledge Rute Marques for expert help, Julia Lobato for the sequencing facility, Dan Holmberg for helpful discussions, and Antonio Coutinho for critical reading of this manuscript. We also wish to thank Drs. Zhenyue Hao and Klaus Rajewsky for sharing unpublished data.
This work was supported by the CNRS-Laboratoire Européen Associé (LEA): Génétique et Développement de la Tolérance Naturelle, and grants from Association Nationale de Recherche du Sida (ANRS) and Association de Recherche Pour Le Cancer (ARC). T.L. Carvalho and J. Demengeot were funded by PRAXIS XXI. T. Mota-Santos was supported by a fellowship from CAPES, Brasil.
Footnotes
Abbreviations used in this paper: FO, follicular; FSC, forward scatter; HSA, heat stable antigen; MZ, marginal zone.
T.L. Carvalho and T. Mota-Santos contributed equally to this work.
T. Mota-Santos' present address is Departamento de Bioquimica e Imunologia, Instituto de Ciências, Biológicas, Universidade Federal de Minas Gerais, Belo Horizonte 30161-970, Brazil.
References
- Hardy R.R., Carmack C.E., Shinton S.A., Kemp J.D., Hayakawa K. Resolution and characterization of pro-B and pre-pro-B cell stages in normal mouse bone marrow. J. Exp. Med. 1991;173:1213–1225. doi: 10.1084/jem.173.5.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sudo T., Ito M., Ogawa Y., Iizuka M., Kodama H., Kunisada T., Hayashi S., Ogawa M., Sakai K., Nishikawa S. Interleukin 7 production and function in stromal cell-dependent B cell development. J. Exp. Med. 1989;170:333–338. doi: 10.1084/jem.170.1.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodwin R.G., Friend D., Ziegler S.F., Jerzy R., Falk B.A., Gimpel S., Cosman D., Dower S.K., March C.J., Namen A.E. Cloning of the human and murine interleukin-7 receptorsdemonstration of a soluble form and homology to a new receptor superfamily. Cell. 1990;60:941–951. doi: 10.1016/0092-8674(90)90342-c. [DOI] [PubMed] [Google Scholar]
- Noguchi M., Nakamura Y., Russell S.M., Ziegler S.F., Tsang M., Cao X., Leonard W.J. Interleukin-2 receptor gamma chaina functional component of the interleukin-7 receptor. Science. 1993;262:1877–1880. doi: 10.1126/science.8266077. [DOI] [PubMed] [Google Scholar]
- von Freeden-Jeffry U., Vieira P., Lucian L.A., McNeil T., Burdach S.E., Murray R. Lymphopenia in interleukin (IL)-7 gene-deleted mice identifies IL-7 as a nonredundant cytokine. J. Exp. Med. 1995;181:1519–1526. doi: 10.1084/jem.181.4.1519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peschon J.J., Morrissey P.J., Grabstein K.H., Ramsdell F.J., Maraskovsky E., Gliniak B.C., Park L.S., Ziegler S.F., Williams D.E., Ware C.B. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J. Exp. Med. 1994;180:1955–1960. doi: 10.1084/jem.180.5.1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pereira P., Forni L., Larsson E.L., Cooper M., Heusser C., Coutinho A. Autonomous activation of B and T cells in antigen-free mice. Eur. J. Immunol. 1986;16:685–688. doi: 10.1002/eji.1830160616. [DOI] [PubMed] [Google Scholar]
- Oliver A.M., Martin F., Kearney J.F. IgMhighCD21high lymphocytes enriched in the splenic marginal zone generate effector cells more rapidly than the bulk of follicular B cells. J. Immunol. 1999;162:7198–7207. [PubMed] [Google Scholar]
- Martin F., Kearney J.F. Positive selection from newly formed to marginal zone B cells depends on the rate of clonal production, CD19, and btk. Immunity. 2000;12:39–49. doi: 10.1016/s1074-7613(00)80157-0. [DOI] [PubMed] [Google Scholar]
- Hayakawa K., Hardy R.R., Herzenberg L.A. Progenitors for Ly-1 B cells are distinct from progenitors for other B cells. J. Exp. Med. 1985;161:1554–1568. doi: 10.1084/jem.161.6.1554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayakawa K., Hardy R.R. Development and function of B-1 cells. Curr. Opin. Immunol. 2000;12:346–353. doi: 10.1016/s0952-7915(00)00098-4. [DOI] [PubMed] [Google Scholar]
- Gu H., Forster I., Rajewsky K. Sequence homologies, N sequence insertion and JH gene utilization in VHDJH joiningimplications for the joining mechanism and the ontogenetic timing of Ly1 B cell and B-CLL progenitor generation. EMBO J. 1990;9:2133–2140. doi: 10.1002/j.1460-2075.1990.tb07382.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tornberg U.C., Holmberg D. B-1a, B-1b and B-2 B cells display unique VHDJH repertoires formed at different stages of ontogeny and under different selection pressures. EMBO J. 1995;14:1680–1689. doi: 10.1002/j.1460-2075.1995.tb07157.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lalor P.A., Morahan G. The peritoneal Ly-1 (CD5) B cell repertoire is unique among murine B cell repertoires. Eur. J. Immunol. 1990;20:485–492. doi: 10.1002/eji.1830200305. [DOI] [PubMed] [Google Scholar]
- Godin I., Dieterlen-Lievre F., Cumano A. Emergence of multipotent hemopoietic cells in the yolk sac and paraaortic splanchnopleura in mouse embryos, beginning at 8.5 days postcoitus (published erratum at 92:10815) Proc. Natl. Acad. Sci. USA. 1995;92:773–777. doi: 10.1073/pnas.92.3.773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmberg D., Forsgren S., Ivars F., Coutinho A. Reactions among IgM antibodies derived from normal, neonatal mice. Eur. J. Immunol. 1984;14:435–441. doi: 10.1002/eji.1830140510. [DOI] [PubMed] [Google Scholar]
- Li Y.S., Wasserman R., Hayakawa K., Hardy R.R. Identification of the earliest B lineage stage in mouse bone marrow. Immunity. 1996;5:527–535. doi: 10.1016/s1074-7613(00)80268-x. [DOI] [PubMed] [Google Scholar]
- Rolink A., ten Boekel E., Melchers F., Fearon D.T., Krop I., Andersson J. A subpopulation of B220+ cells in murine bone marrow does not express CD19 and contains natural killer cell progenitors. J. Exp. Med. 1996;183:187–194. doi: 10.1084/jem.183.1.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper M.D., Mulvaney D., Coutinho A., Cazenave P.A. A novel cell surface molecule on early B-lineage cells. Nature. 1986;321:616–618. doi: 10.1038/321616a0. [DOI] [PubMed] [Google Scholar]
- Carlsson L., Holmberg D. Genetic basis of the neonatal antibody repertoiregermline V-gene expression and limited N-region diversity. Int. Immunol. 1990;2:639–643. doi: 10.1093/intimm/2.7.639. [DOI] [PubMed] [Google Scholar]
- Feeney A.J. Lack of N regions in fetal and neonatal mouse immunoglobulin V-D-J junctional sequences. J. Exp. Med. 1990;172:1377–1390. doi: 10.1084/jem.172.5.1377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregoire K.E., Goldschneider I., Barton R.W., Bollum F.J. Ontogeny of terminal deoxynucleotidyl transferase-positive cells in lymphohemopoietic tissues of rat and mouse. J. Immunol. 1979;123:1347–1352. [PubMed] [Google Scholar]
- Herzenberg L.A. B-1 cellsthe lineage question revisited. Immunol. Rev. 2000;175:9–22. [PubMed] [Google Scholar]
- Hardy R.R., Carmack C.E., Li Y.S., Hayakawa K. Distinctive developmental origins and specificities of murine CD5+ B cells. Immunol. Rev. 1994;137:91–118. doi: 10.1111/j.1600-065x.1994.tb00660.x. [DOI] [PubMed] [Google Scholar]
- Stall A.M., Adams S., Herzenberg L.A., Kantor A.B. Characteristics and development of the murine B-1b (Ly-1 B sister) cell population. Ann. NY Acad. Sci. 1992;651:33–43. doi: 10.1111/j.1749-6632.1992.tb24591.x. [DOI] [PubMed] [Google Scholar]
- Marcos M., Huetz F., Pereira P., Andreu J., Martinez-A C., Coutinho A. Further evidence for coelomic-associated B lymphocytes. Eur. J. Immunol. 1989;19:2031–2035. doi: 10.1002/eji.1830191110. [DOI] [PubMed] [Google Scholar]
- Wells S.M., Kantor A.B., Stall A.M. CD43 (S7) expression identifies peripheral B cell subsets. J. Immunol. 1994;153:5503–5515. [PubMed] [Google Scholar]
- Agenes F., Freitas A.A. Transfer of small resting B cells into immunodeficient hosts results in the selection of a self-renewing activated B cell population. J. Exp. Med. 1999;189:319–330. doi: 10.1084/jem.189.2.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitamura D., Kudo A., Schaal S., Muller W., Melchers F., Rajewsky K. A critical role of lambda 5 protein in B cell development. Cell. 1992;69:823–831. doi: 10.1016/0092-8674(92)90293-l. [DOI] [PubMed] [Google Scholar]
- Oliver A.M., Martin F., Gartland G.L., Carter R.H., Kearney J.F. Marginal zone B cells exhibit unique activation, proliferative and immunoglobulin secretory responses. Eur. J. Immunol. 1997;27:2366–2374. doi: 10.1002/eji.1830270935. [DOI] [PubMed] [Google Scholar]
- Allman D., Li J., Hardy R.R. Commitment to the B lymphoid lineage occurs before DH-JH recombination. J. Exp. Med. 1999;189:735–740. doi: 10.1084/jem.189.4.735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler T.H., Melchers F., Rolink A.G. Interleukin-3 and interleukin-7 are alternative growth factors for the same B-cell precursors in the mouse. Blood. 1995;85:2045–2051. [PubMed] [Google Scholar]
- Ray R.J., Furlonger C., Williams D.E., Paige C.J. Characterization of thymic stromal-derived lymphopoietin (TSLP) in murine B cell development in vitro. Eur. J. Immunol. 1996;26:10–16. doi: 10.1002/eji.1830260103. [DOI] [PubMed] [Google Scholar]
- Wei C., Zeff R., Goldschneider I. Murine pro-B cells require IL-7 and its receptor complex to up-regulate IL-7R alpha, terminal deoxynucleotidyltransferase, and c mu expression. J. Immunol. 2000;164:1961–1970. doi: 10.4049/jimmunol.164.4.1961. [DOI] [PubMed] [Google Scholar]
- Lu L., Chaudhury P., Osmond D.G. Regulation of cell survival during B lymphopoiesisapoptosis and Bcl-2/Bax content of precursor B cells in bone marrow of mice with altered expression of IL-7 and recombinase-activating gene-2. J. Immunol. 1999;162:1931–1940. [PubMed] [Google Scholar]
- Maraskovsky E., Peschon J.J., McKenna H., Teepe M., Strasser A. Overexpression of Bcl-2 does not rescue impaired B lymphopoiesis in IL-7 receptor-deficient mice but can enhance survival of mature B cells. Int. Immunol. 1998;10:1367–1375. doi: 10.1093/intimm/10.9.1367. [DOI] [PubMed] [Google Scholar]
- Vosshenrich C.A., Sharara L.I., Guy-Grand D., Rajewsky K., Muller W., Di Santo J.P. Common cytokine receptor gamma chain (gammac)-deficient B cells persist in T cell-deficient gammac-mice and respond to a T-independent antigen. Eur. J. Immunol. 2000;30:1614–1622. doi: 10.1002/1521-4141(200006)30:6<1614::AID-IMMU1614>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- Vasconcellos R., Braun D., Coutinho A., Demengeot J. Type I IFN sets the stringency of B cell repertoire selection in the bone marrow. Int. Immunol. 1999;11:279–288. doi: 10.1093/intimm/11.2.279. [DOI] [PubMed] [Google Scholar]
- Hao Z., Rajewsky K. Homeostasis of peripheral B cells in the absence of B cell influx from the bone marrow. J. Exp. Med. 2001;194:1151–1163. doi: 10.1084/jem.194.8.1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kroese F.G., Butcher E.C., Stall A.M., Lalor P.A., Adams S., Herzenberg L.A. Many of the IgA producing plasma cells in murine gut are derived from self-replenishing precursors in the peritoneal cavity. Int. Immunol. 1989;1:75–84. doi: 10.1093/intimm/1.1.75. [DOI] [PubMed] [Google Scholar]
- Grandien A., Modigliani Y., Freitas A., Andersson J., Coutinho A. Positive and negative selection of antibody repertoires during B-cell differentiation. Immunol. Rev. 1994;137:53–89. doi: 10.1111/j.1600-065x.1994.tb00659.x. [DOI] [PubMed] [Google Scholar]
- Godin I.E., Garcia-Porrero J.A., Coutinho A., Dieterlen-Lievre F., Marcos M.A. Para-aortic splanchnopleura from early mouse embryos contains B1a cell progenitors. Nature. 1993;364:67–70. doi: 10.1038/364067a0. [DOI] [PubMed] [Google Scholar]
- Herzenberg L.A. Toward a layered immune system. Cell. 1989;59:953–954. doi: 10.1016/0092-8674(89)90748-4. [DOI] [PubMed] [Google Scholar]