The evolving role of lipid rafts and caveolae in G protein-coupled receptor signaling: implications for molecular pharmacology - PubMed (original) (raw)
Review
The evolving role of lipid rafts and caveolae in G protein-coupled receptor signaling: implications for molecular pharmacology
Rennolds S Ostrom et al. Br J Pharmacol. 2004 Sep.
Abstract
The many components of G-protein-coupled receptor (GPCR) signal transduction provide cells with numerous combinations with which to customize their responses to hormones, neurotransmitters, and pharmacologic agonists. GPCRs function as guanine nucleotide exchange factors for heterotrimeric (alpha, beta, gamma) G proteins, thereby promoting exchange of GTP for GDP and, in turn, the activation of 'downstream' signaling components. Recent data indicate that individual cells express mRNA for perhaps over 100 different GPCRs (out of a total of nearly a thousand GPCR genes), several different combinations of G-protein subunits, multiple regulators of G-protein signaling proteins (which function as GTPase activating proteins), and various isoforms of downstream effector molecules. The differential expression of such protein combinations allows for modulation of signals that are customized for a specific cell type, perhaps at different states of maturation or differentiation. In addition, in the linear arrangement of molecular interactions involved in a given GPCR-G-protein-effector pathway, one needs to consider the localization of receptors and post-receptor components in subcellular compartments, microdomains, and molecular complexes, and to understand the movement of proteins between these compartments. Co-localization of signaling components, many of which are expressed at low overall concentrations, allows cells to tailor their responses by arranging, or spatially organizing in unique and kinetically favorable ways, the molecules involved in GPCR signal transduction. This review focuses on the role of lipid rafts and a subpopulation of such rafts, caveolae, as a key spatial compartment enriched in components of GPCR signal transduction. Recent data suggest cell-specific patterns for expression of those components in lipid rafts and caveolae. Such domains likely define functionally important, cell-specific regions of signaling by GPCRs and drugs active at those GPCRs.
Figures
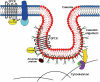
Figure 1
Schematic representation of the lipid and protein organization of a caveola. Sphingolipid- and cholesterol-rich domain is shown in red and nonraft lipid domains are shown in blue. Caveolae contain a coat of oligomeric caveolin molecules inserted into the cytoplasmic leaflet of the membrane. Some proteins, including certain GPCR (shown as heptahelical structures with associated G protein), partition to caveolar domains due to either acylation, binding to caveolin or formation of a sphingolipid ‘shell' around the protein (or by a combination of these, and/or yet unknown, mechanisms). Also shown are undefined cytoskeletal interacting proteins (orange, green, purple) and noncaveolar membrane proteins (blue) and partners (light blue).
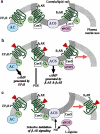
Figure 2
Schematic representation of GPCR-Gs-AC signaling in lipid rafts and caveolae of cardiac myocytes and the effects of eNOS. (a) Caveolar/lipid raft domains contain _β_1AR, _β_2AR, AC6, Gs, eNOS, and caveolin-3 (Cav3), but exclude prostanoid EP2R (Ostrom et al., 2000b). (b) Catecholamines stimulate _β_AR and the generation of cAMP in the caveolar/lipid raft domain. Prostanoids activate receptors located outside lipid rafts and, as a result of compartmentation by phosphodiesterases (PDE), a separate pool of cAMP is generated. _β_2AR translocate out of caveolae upon agonist activation, presumably to internalize via clathrin-coated pits, thus generating a more transient activation of the caveolar/lipid raft pool of cAMP (Ostrom et al., 2001). (c) Activation of eNOS activity leads to the generation of high NO concentrations in the caveolar/lipid raft domain. Due to the localization of eNOS in caveolae and the sensitivity of AC6 to NO-mediated nitrosylation, eNOS activity selectively inhibits the caveolar/lipid raft pool of cAMP (i.e., that stimulated by _β_AR) (Ostrom et al., 2004).
Figure 3
Immunoblot analyses of AC isoform expression in caveolae/lipid raft fractions isolated from human BSMC, HEK-293 cells or human airway epithelial cells (Calu-3). Buoyant lipid raft fractions were isolated from the indicated cells using a nondetergent method followed by centrifugation on a sucrose density gradient, as described previously (Ostrom et al., 2004). The buoyant fractions (cav) and the nonbuoyant fractions (non-cav) were collected from the gradient and, along with whole-cell lysates (WCL), were subjected to SDS–PAGE and immunoblot analyses. The data and others discussed in the text are consistent with the idea that AC5, AC6, and AC3 (except in Calu-3 cells) localize to caveolae/lipid raft fractions, while other AC isoforms localize to nonraft fractions. AC8 has also been shown to localize to lipid raft fractions (Fagan et al., 2000; Smith et al., 2002).
References
- ALOUSI A.A., JASPER J.R., INSEL P.A., MOTULSKY H.J. Stoichiometry of receptor-Gs-adenylate cyclase interactions. FASEB J. 1991;5:2300–2303. -PubMed
- ANDERSON R.G. The caveolae membrane system. Annu. Rev. Biochem. 1998;67:199–225. -PubMed
- ANDERSON R.G., JACOBSON K. A role for lipid shells in targeting proteins to caveolae, rafts, and other lipid domains. Science. 2002;296:1821–1825. -PubMed
- AWASTHI-KALIA M., SCHNETKAMP P.P., DEANS J.P. Differential effects of filipin and methyl-beta-cyclodextrin on B cell receptor signaling. Biochem. Biophys. Res. Commun. 2001;287:77–82. -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- HL63885/HL/NHLBI NIH HHS/United States
- R01 HL071781/HL/NHLBI NIH HHS/United States
- HL66941/HL/NHLBI NIH HHS/United States
- P01 HL066941/HL/NHLBI NIH HHS/United States
- HL71781/HL/NHLBI NIH HHS/United States
- HL53773/HL/NHLBI NIH HHS/United States
- P50 HL053773/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources