Talin is required for integrin-mediated platelet function in hemostasis and thrombosis - PubMed (original) (raw)
Talin is required for integrin-mediated platelet function in hemostasis and thrombosis
Brian G Petrich et al. J Exp Med. 2007.
Abstract
Integrins are critical for hemostasis and thrombosis because they mediate both platelet adhesion and aggregation. Talin is an integrin-binding cytoplasmic adaptor that is a central organizer of focal adhesions, and loss of talin phenocopies integrin deletion in Drosophila. Here, we have examined the role of talin in mammalian integrin function in vivo by selectively disrupting the talin1 gene in mouse platelet precursor megakaryocytes. Talin null megakaryocytes produced circulating platelets that exhibited normal morphology yet manifested profoundly impaired hemostatic function. Specifically, platelet-specific deletion of talin1 led to spontaneous hemorrhage and pathological bleeding. Ex vivo and in vitro studies revealed that loss of talin1 resulted in dramatically impaired integrin alphaIIbbeta3-mediated platelet aggregation and beta1 integrin-mediated platelet adhesion. Furthermore, loss of talin1 strongly inhibited the activation of platelet beta1 and beta3 integrins in response to platelet agonists. These data establish that platelet talin plays a crucial role in hemostasis and provide the first proof that talin is required for the activation and function of mammalian alpha2beta1 and alphaIIbbeta3 integrins in vivo.
Figures
Figure 1.
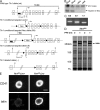
Deletion of talin1 in platelets and megakaryocytes. (A) Scheme of the targeting strategy. Homologous recombination of the Tln1 conditional targeting vector into the Tln1 gene of embryonic stem cells introduced a loxP site (triangle) downstream of exon 4 and a floxed Neo cassette upstream of exon 1 to generate the targeted Neo allele (flN). Partial Cre-mediated recombination in vivo was used to delete only the floxed Neo cassette leaving floxed exons 1–4 (conditional allele, fl). In cells expressing both the Tln1 conditional allele and PF4-Cre, Cre recombinase–mediated recombination will result in deletion of coding exons 1–4, generating the _Tln1_-deleted allele (2). E, EcoRI; probe, external probe for Southern analysis. Primers used for genotyping mice are indicated. (B) Demonstration of homologous recombination in embryonic stem cells by Southern blotting. Genomic DNA from embryonic stem clones was digested with EcoRI and probed with an external probe shown in A. The wild-type allele gives rise to a 13.8-kb band (wt/wt), whereas the targeted allele gives rise to a 7.6-kb band due to introduction of an internal EcoRI site (flN/wt). (C) PCR genotyping of mice possessing the conditional allele. Genomic DNA isolated from ear biopsies of Tln1 fl/fl, Tln1 fl/+, and Tln1 fl/+ mice was analyzed by PCR using the primer pair shown in A. (D) Coomassie blue–stained SDS-PAGE gel of platelet lysates from Tln1 fl/fl Cre+ and Tln1 fl/fl Cre2 mice shows reduction of talin expression in Tln1 fl/fl Cre+ samples, whereas other protein bands are expressed at similar levels. (E) Immunostaining of freshly isolated bone marrow samples showing reduced talin expression in CD41+ megakaryocytes from Tln1 fl/fl Cre+ mice. Bar, 20 mm.
Figure 2.
Reduced survival and perinatal hemorrhage in _Tln1_fl/fl Cre+ mice. (A) Tln1 fl/fl Cre+ mice have prolonged bleeding times in a tail bleeding assay. Time to cessation of bleeding after tail resection was recorded for up to 10 min, at which time bleeding was stopped by cauterization. (B) Example of 1-d-old Tln1 fl/fl Cre+ pup found dead with visible internal hemorrhage (arrows). Table showing reduced survival of Tln1 fl/fl Cre+ mice. The number of animals obtained from Tln1 fl/fl Cre2 × Tln1 fl/fl Cre+ breeding at 3 wk of age is shown. (C) Incidence of fecal blood in Tln1 fl/fl Cre+ and Tln1 fl/fl Cre2 mice at 8–10 wk of age was determined by a guaiac-based hemoccult assay. (D) Peripheral red blood cell counts from 10-wk-old Tln1 fl/fl Cre+ and Tln1 fl/fl Cre2 littermates.
Figure 3.
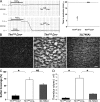
Impaired thrombus formation in _Tln1_fl/fl Cre+ mice in vivo and ex vivo. (A) Time to occlusion of the carotid artery was determined with a Doppler flow probe after a 3-min application of 10% ferric chloride. The experiment was stopped 20 min after injury in all animals. (B) Adhesion of Tln1 fl/fl Cre+, Tln1 fl/fl Cre2, and b3(L476A) platelets to collagen and subsequent thrombus formation in flowing blood were analyzed by epifluorescence and confocal microscopy at a shear rate of 1,500 s21. Heparinized whole blood containing 10 mM mepacrine to render platelets fluorescent was perfused over glass coated with fibrillar type I collagen for 2 min. Tln1 fl/fl Cre2 platelets adhere to the collagen-coated surface and form thrombi (larger aggregates of bright fluorescence). In contrast, Tln1 fl/fl Cre+ platelets form only transient contacts resulting in sparse coverage of platelets on the surface. b3(L746A) platelets form a monolayer that cover much of the surface but do not form thrombi, as seen by the lack of highly fluorescent aggregates that form with Tln1 fl/fl Cre2 platelets. Images shown are single frames from a real-time recording (Video S1). Bar, 20 mm. (C) Percent of the collagen-coated surface covered with platelets was calculated as the number of fluorescent pixels (due to adhesion of fluorescently labeled platelets) divided by the total number of pixels (representing the total surface). *, P < 0.0005; NS, not significant. (D) Quantification of the volume of the thrombi formed on the collagen-coated surface after perfusion for 2 min with blood from Tln1 fl/fl Cre+, Tln1 fl/fl Cre2, and b3(L746A) mice. Confocal serial Z-section reconstructions of the platelet thrombi were used to calculate the thrombi volume as described previously (reference 32). *, P < 0.0005.
Figure 4.
Impaired agonist-induced activation of b3 and b1 integrins in _Tln1_fl/fl Cre+ platelets. (A) The amount of FITC-labeled fibrinogen bound to platelets from Tln1 fl/fl Cre+ or Tln1 fl/fl Cre2 mice was measured by flow cytometry and expressed as the amount of fibrinogen bound to platelets in each group relative to the amount of fibrinogen bound to platelets in the presence of 0.5 mM MnCl2. *, P < 0.001. (B) Fibrinogen binding to Tln1 fl/fl Cre+ and Tln1 fl/fl Cre2 platelets was similar in the presence of 0.5 mM MnCl2. (C) Activation of b1 integrin in Tln1 fl/fl Cre+, Tln1 fl/fl Cre2, and b3(L746A) platelets after stimulation with 1 mM PAR4, 100 mm ADP, and 100 mM epinephrine was measured by binding of the conformation-sensitive antibody 9EG7 and expressed relative to total b1 integrin surface expression measured by the conformation-insensitive antibody HMb1-1. *, P < 0.05; **, P < 0.0005.
Figure 5.
Platelets from _Tln1_fl/fl Cre+ mice are structurally normal and express normal levels of surface receptors. (A) Electron micrographs showing normal structural features of Tln1 fl/fl Cre+ platelets. Platelets from Tln1 fl/fl Cre+ (top left) and Tln1 fl/fl Cre2 (bottom left) mice both display a discoid shape characteristic of resting platelets and similar granular contents. Insets show microtubule coils in platelets from both Tln1 fl/fl Cre+ and Tln1 fl/fl Cre2 mice. Equatorial section of a Tln1 fl/fl Cre+ platelet (top right) shows circumferential microtubule coil (arrow heads). Bars, 1 mm. (B) Platelet fibrinogen content was determined by Coomassie blue staining of platelet lysates separated by SDS-PAGE. 5 mg of purified human fibrinogen served as a marker for the prominent protein band corresponding to fibrinogen in the platelet lysate samples. Consistent with previous reports (reference 17), fibrinogen content is reduced in platelets from b3 integrin null mice. (C) Surface expression of integrin aIIb, a2, b1, b3, and P-selectin was measured by flow cytometry. For P-selectin expression, platelets were incubated with or without PAR4/ADP/epinephrine (1 mm/100 mM/100 mM) for 5 min before the addition of an FITC-conjugated P-selectin antibody.
References
- Ruggeri, Z.M. 2002. Platelets in atherothrombosis. Nat. Med. 8:1227–1234. -PubMed
- Shattil, S.J., and P.J. Newman. 2004. Integrins: dynamic scaffolds for adhesion and signaling in platelets. Blood. 104:1606–1615. -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- HL31950/HL/NHLBI NIH HHS/United States
- U54 GM064346/GM/NIGMS NIH HHS/United States
- P01 HL031950/HL/NHLBI NIH HHS/United States
- WT_/Wellcome Trust/United Kingdom
- P01 HL078784/HL/NHLBI NIH HHS/United States
- 077532/WT_/Wellcome Trust/United Kingdom
- HL078784/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases