Total synthesis of the originally assigned structure of vannusal B - PubMed (original) (raw)
Figure 1
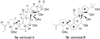
Originally assigned structures of vannusals A (1a) and B (1b).
Figure 2
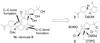
Retrosynthetic analysis vannusal B (1b).
Figure 3
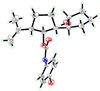
X-Ray derived ORTEP drawing of 14.
Figure 4
X-Ray derived ORTEP drawing of 19.
Figure 5
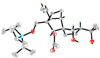
X-Ray derived ORTEP drawing of 34.
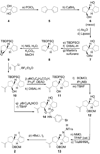
Scheme 1
Enantioselective construction of vinyl iodide 2. Reagents and conditions: (a) POCl3 (2.2 equiv), py, 90 °C, 2 h, 97 %; (b) BH3•THF (2.5 equiv), cylcohexene (2.5 equiv), −40 → 0 °C, 2 h; then 4, −40 → 25 °C, 12 h; then 50 °C, 30 min; then 30 % H2O2 / 3 N NaOH (2:1 v/v), 25 → 50 °C, 12 h, 51 %; (c) Ac2O (3.0 equiv), 4-DMAP (0.02 equiv), py, 25 °C, 3 h, 100 %; (d) Lipase Amano PS (100 wt %), acetone:phosphate buffer (pH = 7) (2:1), 25 °C, 48 h, 100 %, 99 % ee; (e) TBDPSCl (1.2 equiv), imid (3.0 equiv), CH2Cl2, 25 °C, 12 h, 99 %; (f) DIBAL-H (1.0 M in hexanes, 2.5 equiv), CH2Cl2, −78 °C, 30 min, 98 %; (g) Martin’s sulfurane (1.3 equiv), Et3N (3.0 equiv), CH2Cl2, 25 °C, 12 h, 99 %; (h) NIS (1.5 equiv), THF:H2O (4:1), 0 → 25 °C, 1.5 h; then K2CO3 (2.5 equiv), MeOH, 25 °C, 18 h, 91 %; (i) 2-bromopropene (8.5 equiv), _t_BuLi (1.7 M in pentane, 8.0 equiv), THF, −78 °C, 5 min; then BF3•Et2O, 2 min; 9, −78 → −20 °C, 30 min, 83 %; (j) _p_NO2C6H4CO2H (1.5 equiv), DEAD (1.5 equiv), Ph3P (1.8 equiv), benzene, 0 → 25 °C, 18 h, 94 %; (k) DIBAL-H (1.0 M in hexanes, 2.5 equiv), CH2Cl2, −78 °C, 30 min, 96 %; (l) BOMCl (3.0 equiv), _i_Pr2NEt (10 equiv), PhMe, 90 °C 12 h; (m) TBAF (1.0 M in THF, 15 equiv), 70 °C, 97 % over two steps; (n) NMO (1.5 equiv), TPAP (0.03 equiv), CH2Cl2:CH3CN (9:1), 12 h, 96 %; (o) TrisNHNH2 (1.8 equiv), THF, 5 h, 80 %; (p) _n_BuLi (2.5 M in hexanes, 2.1 equiv) THF, −78 → −25 °C, 20 min; then I2 (2.0 equiv), −78 → −25 °C, 20 min, 90 %; (q) _p_BrC6H4NCO (4.0 equiv), Et3N (6.0 equiv), 25 °C; (r) TBAF, THF, 25 °C, 91 % for two steps. 4-DMAP = 4-dimethylaminopyridine, py = pyridine, TBDPS = _tert_-butyldiphenylsilyl, imid = imidazole, DIBAL-H = diisobutylaluminum hydride NIS = _N_-iodosuccinimide, DEAD = diethylazodicarboxylate, BOM = benzyloxymethyl, TBAF = tetra-_n_-butylammonium fluoride, NMO = _N_-methylmorpholine-_N_-oxide, TPAP = tetra-_n_-propylammoniumperuthenate, Tris = triisopropylsulfonyl.
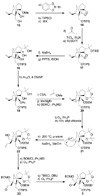
Scheme 2
Construction of aldehyde 3. Reagents and conditions: (a) B-Br-catecholborane (3.5 equiv), CH2Cl2, 25 → 50 °C, 3 h; (b) TIPSCl (1.5 equiv), imid (6.0 equiv), DMF, 24 h, 70 % for two steps; (c) IBX (4.0 equiv), DMSO, 50 °C, 4 h, 90 %; (d) TiCl4 (1.0 M in CH2Cl2, 1.2 equiv), Et3N (3.0 equiv), CH2Cl2, −78 → −30 °C, 30 min; then acetone, −92 °C, 12 h (9:1 dr); (e) TESOTf (3.0 equiv), 2,6-lut. (5.0 equiv), −78 → −40 °C, 1 h, 81 % for the two steps; (f) NaBH4 (20 equiv), THF:MeOH (1:1), −10 → 25 °C, 5 h; (g) PPTS (0.2 equiv), EtOH, 25 °C, 2 h, 85 % for two steps; (h) Ac2O (30 equiv), 4-DMAP (0.1 equiv), Et3N (40 equiv), CH2Cl2, 25 °C, 18 h, 79 %; (i) 2-methoxypropene (20 equiv), CSA (1.0 equiv), CH2Cl2, −78 → −30 °C, 3 h, 82 %; (j) CH3MgBr (50 equiv), PhMe, 50 °C, 8 h, 94 %; (k) SEMCl (10 equiv), _i_Pr2NEt (30 equiv), TBAI (1.0 equiv), CH2Cl2, 50 °C, 48 h, 96 %; (l) O3, py (1.0 equiv), CH2Cl2:MeOH (1:1), −78 °C; then Ph3P (5.0 equiv), −78 → 25 °C, 1 h, 96 %; (m) KH (10 equiv), allyl chloride (20 equiv), HMPA (5.0 equiv), DME, −10 → 25 °C, 3 h, 92 %; (n) _i_Pr2NEt (1.0 equiv), 1,2-dichlorobenzene, 200 °C (µ-wave), 20 min; then NaBH4 (20 equiv), MeOH, 1 h, 25 °C, 88 % for two steps; (o) BOMCl (6.0 equiv), _i_Pr2NEt (15 equiv), CH2Cl2, 50 °C 12 h; (p) O3, py (1.0 equiv), CH2Cl2:MeOH (1:1), −78 °C; then Ph3P (5.0 equiv), −78 → 25 °C, 1 h, 85 % for two steps; (q) TBSCl (10 equiv), DBU (20 equiv), CH2Cl2, 25 °C, 36 h; (r) O3, py (1.0 equiv), CH2Cl2:MeOH (1:1), −78 °C; then Ph3P (5.0 equiv), −78 → 25 °C, 1 h, 97 % for two steps. TIPS = triisopropylsilyl, TES = triethylsilyl, 2,6-lut. = 2,6-dimethylpyridine, PPTS = pyrdinium _p_-toluene-sulfonate, CSA = camphorsufonic acid, HMPA = hexamethylphosphoramide, DBU = 1,8-diazoicyclo[5.4.0]undec-7-ene.
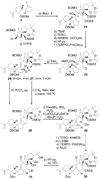
Scheme 3
Completion of the synthesis of structure 1b. Reagents and conditions: (a) 2 (1.3 equiv), _t_BuLi (2.6 equiv), THF, −78 → −40 °C, 30 min; then 3 (1.0 equiv), −40 → 0 °C, 20 min, 80 %; (b) TBAF (1.0 M in THF, 5.0 equiv), THF, 25 °C, 1 h, 98 %; (c) TESCl (1.5 equiv), imid (5.0 equiv), CH2Cl2, 25 °C, 1 h, 99 %; (d) KHMDS (0.5 M in PhMe, 3.0 equiv), ClCO2Me (5.0 equiv), Et3N (5.0 equiv), THF, −78 → 25 °C, 2 h; (e) HF•py/py (1:4), 0 → 25 °C, 12 h, 92 % for two steps; (f) TEMPO (1.0 equiv), PhI(OAc)2 (3.0 equiv), CH2Cl2, 25 °C, 24 h, 98 %; (g) SmI2 (0.1 M in THF, 5.0 equiv), HMPA (15 equiv), THF, −10 → 25 °C, 30 min, 80 % (26: 28 %, 27: 52 %); (h) POCl3 (60.0 equiv), py, 60 °C, 3 h, 85 %; (i) CS2 (8.0 equiv), NaH (6.0 equiv), THF, 0 → 25 °C, 30 min; then CH3I (12 equiv), 0 → 25 °C, 3 h; then 185 °C (µ-wave), 1,2-dichlorobenzene, 15 min, 92 %; (j) ThexBH2 (5.0 equiv), THF, −10 → 25 °C, 1 h; then BH3•THF (15 equiv), 0 → 25 °C, 30 min; then 30 % H2O2/3 N NaOH (1:1), 25 → 40 °C, 1 h; 65 % (1:1.3 mix); (k) _o_NO2C6H4SeCN (2.0 equiv), _n_Bu3P (6.0 equiv), py (12 equiv), THF, 25 °C; then 30 % H2O2, 0 → 25 °C, 67 %; (l) KHMDS (0.5 M in PhMe, 5.0 equiv), TESCl (5.0 equiv), Et3N (8.0 equiv), THF, −78 → 25 °C, 30 min, 94 %; (m) LiDBB (excess), THF, −78 → −50°C, 30 min, 84 %; (n) TEMPO (1.0 equiv), PhI(OAc)2 (3.0 equiv), CH2Cl2, 25 °C, 24 h, 88 %; (o) Ac2O (30 equiv), Et3N (30 equiv), 4-DMAP (1.0 equiv), CH2Cl2, 25 °C, 12 h, 100 %; (p) HF•py/THF (1:4), 25 °C, 3 h; then 3 N aq. HCl/THF (1:3), 25 °C, 6 h, 80 %. KHMDS = potassium hexamethyldisilyazide, TEMPO = 2,2,6,6-teramethyl-1-piperidinyloxy free radical, LiDBB = Lithium di_tert_- butylbiphenyl.
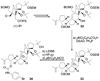
Scheme 4
Synthesis of crystalline derivative 34. Reagents and conditions: (a) DEAD (10 equiv), Ph3P (10 equiv), _p_NO2C6H4CO2H (10 equiv), benzene, 60 °C, 2 h, 88 % based on 58 % conversion; (b) LiDBB, THF, −78 → −50 °C; (c) HF•py/THF, 25 °C, 4 h; (d) _p_BrC6H4NCO (10 equiv), py, 40 °C, 70 % for the three steps.