The art of total synthesis through cascade reactions - PubMed (original) (raw)
Review
. 2009 Nov;38(11):2993-3009.
doi: 10.1039/b903290h. Epub 2009 Apr 21.
Affiliations
- PMID: 19847336
- PMCID: PMC2766600
- DOI: 10.1039/b903290h
Review
The art of total synthesis through cascade reactions
K C Nicolaou et al. Chem Soc Rev. 2009 Nov.
Abstract
The growing importance of cascade reactions reflects and imparts advances in the state of the art of organic synthesis and underscores the desire of synthetic chemists to achieve higher levels of elegance and efficiency. Besides their esthetic appeal, cascade processes offer economical and environmentally friendly means for generating molecular complexity. Because of their many advantages, these reactions have found numerous applications in the synthesis of complex molecules, both natural and designed. In this tutorial review, we highlight the design and execution of cascade reactions within the context of total synthesis as demonstrated with selected examples from these laboratories.
Figures
Figure 1
Molecular structures of endiandric acid A (1), hybocarpone (2), thiostrepton (3), 2,2′-_epi_-cytoskyrin A (4), biyouyanagin A (5), and BE-43472B (6).
Figure 2
Molecular structures of endiandric acids A–D (1, 7–9).
Figure 3
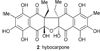
Molecular structure of hybocarpone (2).
Figure 4
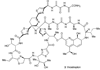
Molecular structure of thiostrepton (3).
Figure 5
Molecular structures of 2,2′-_epi_-cytoskyrin A (4), rugulosin (58), and the proposed structure of rugulin (59).
Figure 6
Retrosynthetic analysis of 2,2′-_epi_-cytoskyrin A (4), rugulosin (58), and the proposed structure of rugulin (59).
Figure 7
Originally proposed (a) and revised (b) structures of biyouyanagin A.
Figure 8
Retrosynthetic analysis of the proposed structures for biyouyanagin A.
Figure 9
Molecular structure of BE-43472B (6).
Figure 10
Retrosynthetic analysis of BE-43472B (6).
Scheme 1
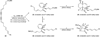
Black’s hypothesis for the biosynthesis of the endiandric acids.
Scheme 2
Biomimetic cascade total synthesis of endiandric acids A (21), D (19), and E (20) methyl esters.
Scheme 3
Biomimetic cascade total synthesis of endiandric acids B (25), C (26), F (23), and G (24) methyl esters.
Scheme 4
Proposed biosynthesis of hybocarpone (2) from 6-methylchristazarin (27).
Scheme 5
Highlights of the photoenolization/Diels–Alder synthesis of 6-methylchristazarin (27)
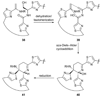
Scheme 6
Oxidative radical dimerization of 33 and total synthesis of hybocarpone (2).
Scheme 7
Proposed biosynthesis of the dehydropiperidine core of thiostrepton.
Scheme 8
Precedent for the proposed aza-Diels–Alder dimerization reaction.
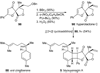
Scheme 9
Biomimetic aza-Diels–Alder dimerization cascade and construction of the dehydropiperidine core of thiostrepton (49).
Scheme 10
Competing amide formation pathways and completion of the total synthesis of thiostrepton (3).
Scheme 11
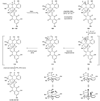
Cascade synthesis of cage-like compounds 82, 83, and 60.
Scheme 12
Completion of the total syntheses of 2,2′-_epi_-cytoskyrin A (4), rugulosin (58), and the proposed structure of rugulin (59).
Scheme 13
Palladium-catalyzed cascade synthesis of spirocycle 98.
Scheme 14
Highlights of the completion of the total synthesis of biyouyanagin A (5).
Scheme 15
Diels–Alder/SN(Ar) cascade synthesis of the BE-43472B skeleton (99 and 107).
Scheme 16
Photoisomerization and completion of the total synthesis of BE-43472B (6).
References
- Nicolaou KC, Edmonds DJ, Bulger PG. Angew. Chem., Int. Ed. 2006;45:7134. -PubMed
- Nicolaou KC, Bulger PG, Sarlah D. Angew. Chem., Int. Ed. 2005;44:4442. -PubMed
- Nicolaou KC, Bulger PG, Sarlah D. Angew. Chem., Int. Ed. 2005;44:4490. -PubMed
- Bandaranayake WM, Banfield JE, St D, Black C, Fallon GD, Gatehouse BM. J. Chem. Soc., Chem. Commun. 1980:162.
- Bandaranayake WM, Banfield JE, St D, Black C. J. Chem. Soc., Chem. Commun. 1980:902.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources