Curcumin activates the p38MPAK-HSP25 pathway in vitro but fails to attenuate diabetic nephropathy in DBA2J mice despite urinary clearance documented by HPLC - PubMed (original) (raw)
Curcumin activates the p38MPAK-HSP25 pathway in vitro but fails to attenuate diabetic nephropathy in DBA2J mice despite urinary clearance documented by HPLC
Jun Ma et al. BMC Complement Altern Med. 2010.
Abstract
Background: Curcumin has anti-inflammatory, anti-oxidant, and anti-proliferative properties, and depending upon the experimental circumstances, may be pro- or anti-apoptotic. Many of these biological actions could ameliorate diabetic nephropathy.
Methods/design: Mouse podocytes, cultured in basal or high glucose conditions, underwent acute exposure to curcumin. Western blots for p38-MAPK, COX-2 and cleaved caspase-3; isoelectric focusing for HSP25 phosphorylation; and DNase I assays for F- to G- actin cleavage were performed for in vitro analyses. In vivo studies examined the effects of dietary curcumin on the development of diabetic nephropathy in streptozotocin (Stz)-induced diabetes in DBA2J mice. Urinary albumin to creatinine ratios were obtained, high performance liquid chromatography was performed for urinary curcuminoid measurements, and Western blots for p38-MAPK and total HSP25 were performed.
Results: Curcumin enhanced the phosphorylation of both p38MAPK and downstream HSP25; inhibited COX-2; induced a trend towards attenuation of F- to G-actin cleavage; and dramatically inhibited the activation of caspase-3 in vitro. In curcumin-treated DBA2J mice with Stz-diabetes, HPLC measurements confirmed the presence of urinary curcuminoid. Nevertheless, dietary provision of curcumin either before or after the induction of diabetes failed to attenuate albuminuria.
Conclusions: Apart from species, strain, early differences in glycemic control, and/or dosing effects, the failure to modulate albuminuria may have been due to a decrement in renal HSP25 or stimulation of the 12/15 lipoxygenase pathway in DBA2J mice fed curcumin. In addition, these studies suggest that timed urine collections may be useful for monitoring curcumin dosing and renal pharmacodynamic effects.
Figures
Figure 1
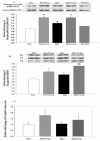
Curcumin activates p38MAPK and phosphorylates HSP25 in cultured Pods. 5.5 mM glucose (NG), 5.5 mM glucose+30 μM curcumin (NG+Cur), 30 mM glucose (HG), 30 mM glucose+30 μM curcumin (HG+Cur), 5.5 mM glucose+24.5 mM mannitol (NG+M). (a) Representative Western blots of phospho-specific p38MAPK (pp38MAPK) and quantitative evaluation of pp38MAPK relative to total p38MAPK (p38MAPK) by densitometric analysis. (b) Representative IEF separating total HSP25 into its nonphosphorylated isoform (P0), mono- (P1), and bi-phoshorylated (P2) isoforms, Western blots of HSP25, and quantitative evaluation of relative phosphorylated HSP25 isoforms to total HSP25 ((P1 + P2)/(P0 + P1 + P2)) by densitometry analysis. Mannitol values (n = 2) are not displayed but were similar to NG. (c) DNAse I assay of F-actin/G-actin ratios. All data expressed as mean ± SEM (n = 3). *P < 0.05 compared with NG; #P < 0.01 compared with NG; **P < 0.05 compared with HG; ##P < 0.01 compared with HG.
Figure 2
Curcumin prevents activation of Caspase 3 and inhibits COX-2 in cultured Pods. Representative Western blot analysis of activated Caspase 3 (a) and COX-2 (b) in Pods. 5.5 mM glucose (NG), 5.5 mM glucose+30 μM curcumin (NG+Cur), 30 mM glucose (HG), 30 mM glucose+30 μM curcumin (HG+Cur). Results were normalized with GAPDH. All data expressed as mean ± SEM (n = 3). *P < 0.05 compared with NG; #P < 0.01 compared with NG; ##P < 0.01 compared with HG.
Figure 3
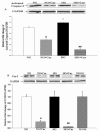
Fasting blood glucose values and effects of curcumin on DN measured by urinary albumin/creatinine in noDM and DM mice. (a) Fasting blood glucose in mice in Experiments 1 and 2. 24 h urine was collected to measure Ualb/cr in noDM and DM mice. (b) Ualb/cr from urine collected on days 9-15 for noDM and DM mice given control chow (0 ppm curcumin) or chow with 5,000 ppm curcumin. Mice were fed curcumin post-Stz injections (Experiment 1). (c) Ualb/cr at 2, 4 and 7 weeks for noDM and DM mice given control chow (0 ppm) or curcumin chow (data pooled for mice fed 5,000 and 7,500 ppm curcumin). Mice were fed curcumin pre-Stz injections (Experiment 2). All data are log-transformed (log10). *P < 0.05
Figure 4
Urinary curcuminoid excretion in noDM and DM mice fed with curcumin versus control chow. Curcumin metabolites were measured by High Performance Liquid Chromatography (HPLC) as a measure of bioavailability in a timed urine collection. (a) Total urinary curcuminoids in nmol and (b) Urinary curcumin/creatinine in nmol/mg. All data are log-transformed (log10). *P < 0.05; #P < 0.01.
Figure 5
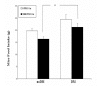
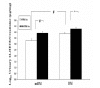
Mouse Food Intake. After Stz-injection and curcumin feeding began (Experiment 1), the total amount of control chow (0 ppm curcumin) or curcumin chow (5,000 ppm curcumin) consumed by each mouse over the preceding 4 days was measured on day 4. *P < 0.05.
Figure 6
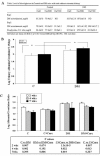
Effects of Curcumin on p38MAPK and total HSP25 on Stz-DM mice. Representative Western blot and quantitative evaluation of phospho-specific p38MAPK (a) and total HSP25 relative to GAPDH (b) in renal cortex of DMCur0 and DMCur5,000 mice. *P < 0.05; #P < 0.01.
Figure 7
Curcumin did not lower U 12-HETE/cr excretion in noDM and DM mice. Urinary 12-HETE/cr ratios for noDM and DM mice fed chow with 0 and 5,000 ppm curcumin. Mice were fed curcumin post-Stz injections (Experiment 1). Graph represents pooled data (day 9 and 15). *P < 0.05; #P < 0.01.
References
- Schieppati A, Remuzzi G. Proteinuria and its consequences in renal disease. Acta Paediatr Suppl. 2003;92(443):9–13. -PubMed
- Raman N, Atkinson SJ. Rho controls actin cytoskeletal assembly in renal epithelial cells during ATP depletion and recovery. Am J Physiol. 1999;276(6 Pt 1):C1312–24. -PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials