Responses of gut microbiota to diet composition and weight loss in lean and obese mice - PubMed (original) (raw)
Responses of gut microbiota to diet composition and weight loss in lean and obese mice
Yann Ravussin et al. Obesity (Silver Spring). 2012 Apr.
Abstract
Maintenance of a reduced body weight is accompanied by a decrease in energy expenditure beyond that accounted for by reduced body mass and composition, as well as by an increased drive to eat. These effects appear to be due--in part--to reductions in circulating leptin concentrations due to loss of body fat. Gut microbiota have been implicated in the regulation of body weight. The effects of weight loss on qualitative aspects of gut microbiota have been studied in humans and mice, but these studies have been confounded by concurrent changes in diet composition, which influence microbial community composition. We studied the impact of 20% weight loss on the microbiota of diet-induced obese (DIO: 60% calories fat) mice on a high-fat diet (HFD). Weight-reduced DIO (DIO-WR) mice had the same body weight and composition as control (CON) ad-libitum (AL) fed mice being fed a control diet (10% calories fat), allowing a direct comparison of diet and weight-perturbation effects. Microbial community composition was assessed by pyrosequencing 16S rRNA genes derived from the ceca of sacrificed animals. There was a strong effect of diet composition on the diversity and composition of the microbiota. The relative abundance of specific members of the microbiota was correlated with circulating leptin concentrations and gene expression levels of inflammation markers in subcutaneous white adipose tissue in all mice. Together, these results suggest that both host adiposity and diet composition impact microbiota composition, possibly through leptin-mediated regulation of mucus production and/or inflammatory processes that alter the gut habitat.
Conflict of interest statement
DISCLOSURE
The authors declared no conflict of interest.
Figures
Figure 1
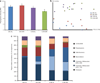
Effects of diet and weight reduction on the gut microbiota. (a) Phylogenetic Diversity (PD) of the cecal samples from the four groups of mice (mean ± s.e.m. compared by two-way ANOVA) and (b) Principal Coordinates Analysis (PCoA) plot of the unweighted UniFrac distances. PC1 and PC3 values for each mouse sample are plotted; percent variation explained by each PC is shown in parentheses: DIO-AL, diet-induced obese mice: blue; DIO-WR, weight-reduced DIO: red; CON-AL, control diet-fed mice: purple; CON-WR, weight-reduced CON: green. (c) Relative abundances of the different phyla in each of the groups. The phylum Firmicutes was broken down into OTU 303, OTU 716 (which are both classified as Allobaculum), and all other Firmicutes that did not fall into these two OTUs.
Figure 2
Members of the microbiota that differ in abundance by diet composition and treatment (WR vs. AL). (a) Nearest shrunken centroid analysis of the 15 OTUs accounting for the differences among the four groups of mice. For each OTU listed in center, direction of the horizontal bars indicates relatively over-represented (right of vertical line) and under-represented (left of vertical line); the length of the bar indicates the strength of the effect. (b) Heat map of the “classifying” OTUs. Columns show, for each mouse, the abundance data of OTUs listed in center. The abundances of the OTUs were clustered using unsupervised hierarchical clustering (blue, low abundance; red, high abundance). The Phylum, Genus of each of the classifying OTUs is noted. AL, ad-libitum diets; OTU, relative operational taxonomic unit; WR, weight-reduced.
Figure 3
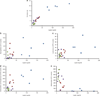
Associations between host serum leptin concentrations and gut microbiota. (a) Correlations of fat mass content (by nuclear magnetic resonance) with circulating leptin concentrations. (b, c, d, e) Correlations between leptin concentrations and the abundance of relative operational taxonomic units of interest. CON-WR, weight-reduced CON: green, CON-AL, control diet-fed mice: purple, DIO-AL, diet-induced obese mice: blue, DIO-WR, weight-reduced DIO: red.
Figure 4
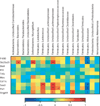
Heat map describing the correlation of the abundances of different operational taxonomic units and transcription levels of inflammation-related genes in inguinal adipose tissue. The colors range from blue (negative correlation; −1) to red (positive correlation; 1). Significant correlations are noted by *P < 0.05 and **P < 0.01 (The computed false discovery rate is about 0.25 using the Benjamini Hochberg procedure (38)).
Figure 5
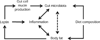
Schematic depicting possible inter-relationships among diet composition, gut microbiota, circulating leptin, body fat, markers of inflammation, and gut mucin. Body fat directly determines leptin production and elevated body fat increases macrophage infiltration (with associated production/release of inflammatory molecules such as tumor necrosis factor α, serum amyloid A3, and chemokine (C-C motif) ligand 2 (MCP-1) in adipose tissue). The results presented here suggest that diet composition (fractional fat content) directly affects gut microbiota independent of effects mediated by body weight and body composition. Leptin promotes proliferation, differentiation, and survival of immune cells. Leptin also stimulates mucin production in mouse and human intestinal cells (27,28). Mucin affects local bacterial “micro-niches” in the gut by favoring the growth of some bacteria (25,26). Leptin can affect intestinal barrier function by inhibiting apoptosis and promoting regeneration of intestinal epithelium (39,40). These changes in epithelial composition may in turn affect microbiota populations in the gut. The dashed line between body fat and gut microbiota suggests biologically possible connection(s) that might be mediated by adipocytokines or other molecules secreted from adipose tissue.
Similar articles
- Panax notoginseng saponins modulate the gut microbiota to promote thermogenesis and beige adipocyte reconstruction via leptin-mediated AMPKα/STAT3 signaling in diet-induced obesity.
Xu Y, Wang N, Tan HY, Li S, Zhang C, Zhang Z, Feng Y. Xu Y, et al. Theranostics. 2020 Sep 14;10(24):11302-11323. doi: 10.7150/thno.47746. eCollection 2020. Theranostics. 2020. PMID: 33042284 Free PMC article. - Diet-induced obese mice are leptin insufficient after weight reduction.
Shi H, Akunuru S, Bierman JC, Hodge KM, Mitchell MC, Foster MT, Seeley RJ, Reizes O. Shi H, et al. Obesity (Silver Spring). 2009 Sep;17(9):1702-9. doi: 10.1038/oby.2009.106. Epub 2009 Apr 16. Obesity (Silver Spring). 2009. PMID: 19373220 Free PMC article. - Dietary α-lactalbumin alters energy balance, gut microbiota composition and intestinal nutrient transporter expression in high-fat diet-fed mice.
Boscaini S, Cabrera-Rubio R, Speakman JR, Cotter PD, Cryan JF, Nilaweera KN. Boscaini S, et al. Br J Nutr. 2019 May;121(10):1097-1107. doi: 10.1017/S0007114519000461. Epub 2019 Mar 5. Br J Nutr. 2019. PMID: 30834845 - Obesity and the gut microbiota.
Flint HJ. Flint HJ. J Clin Gastroenterol. 2011 Nov;45 Suppl:S128-32. doi: 10.1097/MCG.0b013e31821f44c4. J Clin Gastroenterol. 2011. PMID: 21992951 Review. - Resistant starch and energy balance: impact on weight loss and maintenance.
Higgins JA. Higgins JA. Crit Rev Food Sci Nutr. 2014;54(9):1158-66. doi: 10.1080/10408398.2011.629352. Crit Rev Food Sci Nutr. 2014. PMID: 24499148 Free PMC article. Review.
Cited by
- Enhanced Anti-Obesity Effects of Euphorbia Kansui Extract through Macrophage and Gut Microbiota Modulation: A Real-World Clinical and In Vivo Study.
Noh JW, Yoo JH, Lee BC. Noh JW, et al. Pharmaceuticals (Basel). 2024 Aug 27;17(9):1131. doi: 10.3390/ph17091131. Pharmaceuticals (Basel). 2024. PMID: 39338296 Free PMC article. - Chronic binge drinking-induced susceptibility to colonic inflammation is microbiome-dependent.
Fonseca-Pereira D, Bae S, Michaud M, Glickman JN, Garrett WS. Fonseca-Pereira D, et al. Gut Microbes. 2024 Jan-Dec;16(1):2392874. doi: 10.1080/19490976.2024.2392874. Epub 2024 Aug 20. Gut Microbes. 2024. PMID: 39163515 Free PMC article. - Integrated analysis of gut microbiome and its metabolites in ACE2-knockout and ACE2-overexpressed mice.
Song L, Ji W, Cao X. Song L, et al. Front Cell Infect Microbiol. 2024 Jul 17;14:1404678. doi: 10.3389/fcimb.2024.1404678. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 39086603 Free PMC article. - Examining immune-inflammatory mechanisms of probiotic supplementation in depression: secondary findings from a randomized clinical trial.
Sempach L, Doll JPK, Limbach V, Marzetta F, Schaub AC, Schneider E, Kettelhack C, Mählmann L, Schweinfurth-Keck N, Ibberson M, Lang UE, Schmidt A. Sempach L, et al. Transl Psychiatry. 2024 Jul 24;14(1):305. doi: 10.1038/s41398-024-03030-7. Transl Psychiatry. 2024. PMID: 39048549 Free PMC article. Clinical Trial. - Termite Fungus Comb Polysaccharides Alleviate Hyperglycemia and Hyperlipidemia in Type 2 Diabetic Mice by Regulating Hepatic Glucose/Lipid Metabolism and the Gut Microbiota.
Xiao H, Song X, Wang P, Li W, Qin S, Huang C, Wu B, Jia B, Gao Q, Song Z. Xiao H, et al. Int J Mol Sci. 2024 Jul 6;25(13):7430. doi: 10.3390/ijms25137430. Int J Mol Sci. 2024. PMID: 39000541 Free PMC article.
References
- Flegal KM, Carroll MD, Ogden CL, Curtin LR. Prevalence and trends in obesity among US adults, 1999–2008. JAMA. 2010;303:235–241. - PubMed
- MacLean PS, Higgins JA, Johnson GC, et al. Enhanced metabolic efficiency contributes to weight regain after weight loss in obesityprone rats. Am J Physiol Regul Integr Comp Physiol. 2004;287:R1306–R1315. - PubMed
- Hambly C, Speakman JR. Contribution of different mechanisms to compensation for energy restriction in the mouse. Obes Res. 2005;13:1548–1557. - PubMed
- Rosenbaum M, Vandenborne K, Goldsmith R, et al. Effects of experimental weight perturbation on skeletal muscle work efficiency in human subjects. Am J Physiol Regul Integr Comp Physiol. 2003;285:R183–R192. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 5P30DK026687/DK/NIDDK NIH HHS/United States
- P01 DK078669/DK/NIDDK NIH HHS/United States
- HG4872/HG/NHGRI NIH HHS/United States
- 5R01DK064773/DK/NIDDK NIH HHS/United States
- U01 HG004866/HG/NHGRI NIH HHS/United States
- HG4866/HG/NHGRI NIH HHS/United States
- P30 DK026687/DK/NIDDK NIH HHS/United States
- R01 DK052431/DK/NIDDK NIH HHS/United States
- DK78669/DK/NIDDK NIH HHS/United States
- HHMI/Howard Hughes Medical Institute/United States
- DK063608-08/DK/NIDDK NIH HHS/United States
- R01 DK064773/DK/NIDDK NIH HHS/United States
- R01 HG004872/HG/NHGRI NIH HHS/United States
- P30 DK063608/DK/NIDDK NIH HHS/United States
- 5R01DK052431/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical