A phase I/II trial of intensity modulated radiation (IMRT) dose escalation with concurrent fixed-dose rate gemcitabine (FDR-G) in patients with unresectable pancreatic cancer - PubMed (original) (raw)
Clinical Trial
. 2012 Dec 1;84(5):1166-71.
doi: 10.1016/j.ijrobp.2012.02.051. Epub 2012 Apr 27.
Mathew Schipper, Isaac R Francis, Scott Hadley, Randall Ten-Haken, Theodore Lawrence, Daniel Normolle, Diane M Simeone, Christopher Sonnenday, Ross Abrams, William Leslie, Gazala Khan, Mark M Zalupski
Affiliations
- PMID: 22543215
- PMCID: PMC3421048
- DOI: 10.1016/j.ijrobp.2012.02.051
Clinical Trial
A phase I/II trial of intensity modulated radiation (IMRT) dose escalation with concurrent fixed-dose rate gemcitabine (FDR-G) in patients with unresectable pancreatic cancer
Edgar Ben-Josef et al. Int J Radiat Oncol Biol Phys. 2012.
Abstract
Purpose: Local failure in unresectable pancreatic cancer may contribute to death. We hypothesized that intensification of local therapy would improve local control and survival. The objectives were to determine the maximum tolerated radiation dose delivered by intensity modulated radiation with fixed-dose rate gemcitabine (FDR-G), freedom from local progression (FFLP), and overall survival (OS).
Methods and materials: Eligibility included pathologic confirmation of adenocarcinoma, radiographically unresectable, performance status of 0-2, absolute neutrophil count of ≥ 1,500/mm(3), platelets ≥ 100,000/mm(3), creatinine <2 mg/dL, bilirubin <3 mg/dL, and alanine aminotransferase/aspartate aminotransferase ≤ 2.5 × upper limit of normal. FDR-G (1000 mg/m(2)/100 min intravenously) was given on days -22 and -15, 1, 8, 22, and 29. Intensity modulated radiation started on day 1. Dose levels were escalated from 50-60 Gy in 25 fractions. Dose-limiting toxicity was defined as gastrointestinal toxicity grade (G) ≥ 3, neutropenic fever, or deterioration in performance status to ≥ 3 between day 1 and 126. Dose level was assigned using TITE-CRM (Time-to-Event Continual Reassessment Method) with the target dose-limiting toxicity (DLT) rate set to 0.25.
Results: Fifty patients were accrued. DLTs were observed in 11 patients: G3/4 anorexia, nausea, vomiting, and/or dehydration (7); duodenal bleed (3); duodenal perforation (1). The recommended dose is 55 Gy, producing a probability of DLT of 0.24. The 2-year FFLP is 59% (95% confidence interval [CI]: 32-79). Median and 2-year overall survival are 14.8 months (95% CI: 12.6-22.2) and 30% (95% CI 17-45). Twelve patients underwent resection (10 R0, 2 R1) and survived a median of 32 months.
Conclusions: High-dose radiation therapy with concurrent FDR-G can be delivered safely. The encouraging efficacy data suggest that outcome may be improved in unresectable patients through intensification of local therapy.
Copyright © 2012 Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of Interest: None
Figures
Figure 1. Trial schema
A general schema of the trial. The trial started with one 3-week cycle of run-in FDR-G. This was followed by 5 weeks of concurrent FDR-G and dose-escalated IMRT. After completion of IMRT patients were observed for additional 13 weeks for the development of DLT. During this observation period, additional chemotherapy was allowed (single agent gemcitabine was recommended).
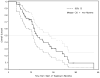
Figure 2. Overall survival
Kaplan Meier plot of overall survival. The median and 2-year overall survival are 14.8 months (95% CI: 12.6–22.2) and 30% (95% CI 17–45).
Figure 3. Freedom from local progression
Kaplan Meier plot of Freedom from Local Progression. The 2-year FFLP is 59% (95% CI: 32– 79).
References
- Murphy JD, Adusumilli S, Griffith KA, et al. Full-dose gemcitabine and concurrent radiotherapy for unresectable pancreatic cancer. Int J Radiat Oncol Biol Phys. 2007;68:801–808. -PubMed
- Loehrer PJMEP, Cardenes HR, Wagner L, Brell JM, Ramanathan RK, Crane CH, Alberts SR, Benson AB Eastern Cooperative Oncology Group. A randomized phase III study of gemcitabine in combination with radiation therapy versus gemcitabine alone in patients with localized, unresectable pancreatic cancer: E4201. J Clin Oncol. 2008;26
- Ceha HM, van Tienhoven G, Gouma DJ, et al. Feasibility and efficacy of high dose conformal radiotherapy for patients with locally advanced pancreatic carcinoma. Cancer. 2000;89:2222–2229. -PubMed
- Lawrence TS, Chang EY, Hahn TM, et al. Radiosensitization of pancreatic cancer cells by 2',2'-difluoro-2'-deoxycytidine. Int J Radiat Oncol Biol Phys. 1996;34:867–872. -PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical