Glycoprotein biomarker panel for pancreatic cancer discovered by quantitative proteomics analysis - PubMed (original) (raw)
. 2014 Apr 4;13(4):1873-84.
doi: 10.1021/pr400967x. Epub 2014 Mar 10.
Affiliations
- PMID: 24571389
- PMCID: PMC3993962
- DOI: 10.1021/pr400967x
Glycoprotein biomarker panel for pancreatic cancer discovered by quantitative proteomics analysis
Song Nie et al. J Proteome Res. 2014.
Abstract
Pancreatic cancer is a lethal disease where specific early detection biomarkers would be very valuable to improve outcomes in patients. Many previous studies have compared biosamples from pancreatic cancer patients with healthy controls to find potential biomarkers. However, a range of related disease conditions can influence the performance of these putative biomarkers, including pancreatitis and diabetes. In this study, quantitative proteomics methods were applied to discover potential serum glycoprotein biomarkers that distinguish pancreatic cancer from other pancreas related conditions (diabetes, cyst, chronic pancreatitis, obstructive jaundice) and healthy controls. Aleuria aurantia lectin (AAL) was used to extract fucosylated glycoproteins and then both TMT protein-level labeling and label-free quantitative analysis were performed to analyze glycoprotein differences from 179 serum samples across the six different conditions. A total of 243 and 354 serum proteins were identified and quantified by label-free and TMT protein-level quantitative strategies, respectively. Nineteen and 25 proteins were found to show significant differences in samples between the pancreatic cancer and other conditions using the label-free and TMT strategies, respectively, with 7 proteins considered significant in both methods. Significantly different glycoproteins were further validated by lectin-ELISA and ELISA assays. Four candidates were identified as potential markers with profiles found to be highly complementary with CA 19-9 (p < 0.001). Obstructive jaundice (OJ) was found to have a significant impact on the performance of every marker protein, including CA 19-9. The combination of α-1-antichymotrypsin (AACT), thrombospondin-1 (THBS1), and haptoglobin (HPT) outperformed CA 19-9 in distinguishing pancreatic cancer from normal controls (AUC = 0.95), diabetes (AUC = 0.89), cyst (AUC = 0.82), and chronic pancreatitis (AUC = 0.90). A marker panel of AACT, THBS1, HPT, and CA 19-9 showed a high diagnostic potential in distinguishing pancreatic cancer from other conditions with OJ (AUC = 0.92) or without OJ (AUC = 0.95).
Figures
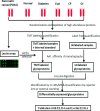
Figure 1
Study design for identification of serum glycoprotein markers for pancreatic cancer. Depleted sera from pancreatic cancer, diabetes, cyst, chronic pancreatitis, obstructive jaundice, and healthy controls were first applied to a lectin array. On the basis of the results of lectin array analysis, glycoproteins were extracted using AAL lectin, which were quantified by MS-based quantitative proteomics (TMT protein-level labeling and spectral counting methods). Potential candidates were validated by ELISA and lectin-ELISA.
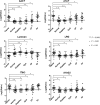
Figure 2
Scatter plots of quantitative ratios from TMT labeling for the six most significant proteins in each of the studied groups: pancreatic cancer, normal, diabetes, cyst, CP, OJ. The p value of the 6 significant proteins are <0.001 between cancer and other controls.
Figure 3
Serum concentration profiles of the four protein candidates (AACT, A1AT, LRG, THBS1) in each studied group: pancreatic cancer, normal, diabetes, cyst, CP, OJ based on ELISA assay.
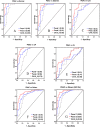
Figure 4
Performance of biomarker panels based on ELISA results, the ROC curve, and AUC value of panel 1 and panel 2 were shown. Panel 1 includes AACT, THBS1, and HPT; panel 2 is the combination of AACT, THBS1, HPT, and CA 19–9.
References
- Jemal A.; Siegel R.; Ward E.; Hao Y.; Xu J.; Thun M. J. Cancer statistics, 2009. Ca–Cancer J. Clin. 2009, 594225–49. -PubMed
- Chu D.; Kohlmann W.; Adler D. G. Identification and screening of individuals at increased risk for pancreatic cancer with emphasis on known environmental and genetic factors and hereditary syndromes. JOP 2010, 113203–12. -PubMed
- Sohn T. A.; Lillemoe K. D.; Cameron J. L.; Huang J. J.; Pitt H. A.; Yeo C. J. Surgical palliation of unresectable periampullary adenocarcinoma in the 1990s. J. Am. Coll. Surg. 1999, 1886658–66discussion 666–9.. -PubMed
- Singh P.; Srinivasan R.; Wig J. D. Major molecular markers in pancreatic ductal adenocarcinoma and their roles in screening, diagnosis, prognosis, and treatment. Pancreas 2011, 405644–52. -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 GM49500/GM/NIGMS NIH HHS/United States
- R01 CA154455/CA/NCI NIH HHS/United States
- P50 CA130810/CA/NCI NIH HHS/United States
- R01 GM049500/GM/NIGMS NIH HHS/United States
- 1 P50CA130810/CA/NCI NIH HHS/United States
- K23 DK082097/DK/NIDDK NIH HHS/United States
- 1 R01 CA154455/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous