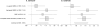The 19q12 bladder cancer GWAS signal: association with cyclin E function and aggressive disease - PubMed (original) (raw)
. 2014 Oct 15;74(20):5808-18.
doi: 10.1158/0008-5472.CAN-14-1531.
Indu Kohaar 1, Lee E Moore 2, Petra Lenz 3, Jonine D Figueroa 2, Wei Tang 1, Patricia Porter-Gill 1, Nilanjan Chatterjee 2, Alexandra Scott-Johnson 1, Montserrat Garcia-Closas 4, Brian Muchmore 1, Dalsu Baris 2, Ashley Paquin 1, Kris Ylaya 5, Molly Schwenn 6, Andrea B Apolo 7, Margaret R Karagas 8, McAnthony Tarway 1, Alison Johnson 9, Adam Mumy 1, Alan Schned 8, Liliana Guedez 10, Michael A Jones 11, Masatoshi Kida 12, G M Monawar Hosain 13, Nuria Malats 14, Manolis Kogevinas 15, Adonina Tardon 16, Consol Serra 17, Alfredo Carrato 18, Reina Garcia-Closas 19, Josep Lloreta 20, Xifeng Wu 21, Mark Purdue 2, Gerald L Andriole Jr 22, Robert L Grubb 3rd 22, Amanda Black 2, Maria T Landi 2, Neil E Caporaso 2, Paolo Vineis 23, Afshan Siddiq 24, H Bas Bueno-de-Mesquita 25, Dimitrios Trichopoulos 26, Börje Ljungberg 27, Gianluca Severi 28, Elisabete Weiderpass 29, Vittorio Krogh 30, Miren Dorronsoro 31, Ruth C Travis 32, Anne Tjønneland 33, Paul Brennan 34, Jenny Chang-Claude 35, Elio Riboli 24, Jennifer Prescott 36, Constance Chen 37, Immaculata De Vivo 36, Edward Govannucci 38, David Hunter 37, Peter Kraft 37, Sara Lindstrom 37, Susan M Gapstur 39, Eric J Jacobs 39, W Ryan Diver 39, Demetrius Albanes 2, Stephanie J Weinstein 2, Jarmo Virtamo 40, Charles Kooperberg 41, Chancellor Hohensee 41, Rebecca J Rodabough 41, Victoria K Cortessis 42, David V Conti 43, Manuela Gago-Dominguez 44, Mariana C Stern 43, Malcolm C Pike 45, David Van Den Berg 43, Jian-Min Yuan 46, Christopher A Haiman 43, Olivier Cussenot 47, Geraldine Cancel-Tassin 48, Morgan Roupret 49, Eva Comperat 49, Stefano Porru 50, Angela Carta 50, Sofia Pavanello 51, Cecilia Arici 50, Giuseppe Mastrangelo 51, H Barton Grossman 52, Zhaoming Wang 53, Xiang Deng 53, Charles C Chung 53, Amy Hutchinson 53, Laurie Burdette 53, William Wheeler 54, Joseph Fraumeni Jr 2, Stephen J Chanock 2, Stephen M Hewitt 5, Debra T Silverman 2, Nathaniel Rothman 2, Ludmila Prokunina-Olsson 55
Affiliations
- PMID: 25320178
- PMCID: PMC4203382
- DOI: 10.1158/0008-5472.CAN-14-1531
The 19q12 bladder cancer GWAS signal: association with cyclin E function and aggressive disease
Yi-Ping Fu et al. Cancer Res. 2014.
Abstract
A genome-wide association study (GWAS) of bladder cancer identified a genetic marker rs8102137 within the 19q12 region as a novel susceptibility variant. This marker is located upstream of the CCNE1 gene, which encodes cyclin E, a cell-cycle protein. We performed genetic fine-mapping analysis of the CCNE1 region using data from two bladder cancer GWAS (5,942 cases and 10,857 controls). We found that the original GWAS marker rs8102137 represents a group of 47 linked SNPs (with r(2) ≥ 0.7) associated with increased bladder cancer risk. From this group, we selected a functional promoter variant rs7257330, which showed strong allele-specific binding of nuclear proteins in several cell lines. In both GWASs, rs7257330 was associated only with aggressive bladder cancer, with a combined per-allele OR = 1.18 [95% confidence interval (CI), 1.09-1.27, P = 4.67 × 10(-5)] versus OR = 1.01 (95% CI, 0.93-1.10, P = 0.79) for nonaggressive disease, with P = 0.0015 for case-only analysis. Cyclin E protein expression analyzed in 265 bladder tumors was increased in aggressive tumors (P = 0.013) and, independently, with each rs7257330-A risk allele (P(trend) = 0.024). Overexpression of recombinant cyclin E in cell lines caused significant acceleration of cell cycle. In conclusion, we defined the 19q12 signal as the first GWAS signal specific for aggressive bladder cancer. Molecular mechanisms of this genetic association may be related to cyclin E overexpression and alteration of cell cycle in carriers of CCNE1 risk variants. In combination with established bladder cancer risk factors and other somatic and germline genetic markers, the CCNE1 variants could be useful for inclusion into bladder cancer risk prediction models.
©2014 American Association for Cancer Research.
Conflict of interest statement
Disclosure of Potential Conflicts of Interest: G.L. Andriole is a consultant/advisory board member of Augmenix, Bayer, GlaxoSmithKline, Myriad Genetics and Genomic Health. H.B. Grossman has ownership interest (including patents) in Abbott Molecular and is a consultant/board member for Nucleix. No potential conflicts of interest were disclosed by the other authors.
Figures
Figure 1. Genetic and genomic landscape of the CCNE1 region
Genomic plot of the CCNE1 region is based on information from the UCSC browser (
) and RNA-sequencing of 7 tumors and 5 adjacent normal bladder tissue samples. The plot shows the main CCNE1 transcripts detected by our RNA-sequencing – WT1, WT2, cyclin ES and cyclin ET. Alternative exons missing in forms cyclin ES and cyclin ET are marked by red boxes. Arrows indicate translation start sites and direction of protein synthesis. Genetic variants on the plot are coding non-synonymous CCNE1 variants (marked in red), GWAS marker rs8102137, promoter variant rs7257330, an ovarian cancer candidate variant rs3218036, and several variants previously associated with various cancers (Supplementary Table 7). Histone H3 Lysine 27 acetylation marks (H3K27Ac) in 7 cell lines are a part of ENCODE track presented by the UCSC browser.
Figure 2. Forest plot for association of rs8102137 and rs7257330 in bladder cancer subgroups in the combined set of NCI-GWAS1 and NCI-GWAS2 samples
The plot shows allelic odds ratios and 95% confidence intervals for SNPs. Analysis was performed only on subjects with genotype information available for both SNPs, where rs8102137 was from actual genotyping in NCI-GWAS1 and NCI-GWAS2, and rs7257330 was converted from imputed allelic dosage with ≥ 90% probability and validated by TaqMan genotyping in a subset of samples. Bladder cancer sub-groups were defined in three categories (Supplementary Table 4): low-grade NMIBC (Ta with G1/G2), high-grade NMIBC (Ta with G3/G4 or all T1) and MIBC (all T2-T4)(3). Incomplete clinical information category includes cases with missing or incomplete tumor stage or grade data.
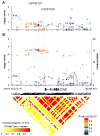
Figure 3. Association results, linkage disequilibrium (r2) and recombination plots of the CCNE1 region in combined NCI-GWAS1 and NCI-GWAS2 samples
Left y-axis (–log10 scale) presents p-values for GWAS (filled circles) and well-imputed markers (IMPUTE2-score ≥ 0.9, open circles), adjusted for age, gender, study sites, smoking status (ever/never) and top eigenvectors (EV1, EV5, EV6) from principal component analysis. The color scheme is based on r2 values between rs8102137 and corresponding markers. Right y-axis presents likelihood ratio of putative recombination hotspots based on five sets of 100 randomly selected controls from NCI-GWAS1 and shown as connected gray lines. The GWAS marker rs8102137 and the promoter variant rs7257330 are shown as diamonds. Pair-wise r2 values based on all control samples are displayed at the bottom of the plot for all 165 SNPs included in our analyses. Genomic coordinates are based on the NCBI Human Genome Build 37.1/UCSC hg19 assembly. (A) Association analysis in a subset of patients with non-aggressive bladder cancer (stage Ta with grade G1 or G2, n = 1,870) vs. all controls (n = 10,857). (B) Association analysis in a subset of patients with aggressive bladder cancer (stage Ta with grade G3+ or stages T1-T4 with any grade, n = 1,930) vs. all controls (n = 10,857).
Figure 4. Analysis of CCNE1 mRNA expression in bladder tumors and adjacent normal tissue and in relation to rs8102137 and rs7257330 genotypes
Expression of total CCNE1 (all transcripts), cyclin ES and cyclin ET transcripts was measured with TaqMan assays. CCNE1 expression was normalized to a geometric mean of endogenous controls (B2M, GAPDH and PPIA) and presented on the log 2 scale. Less negative values correspond to higher relative expression of CCNE1 transcripts. The association between normalized CCNE1 mRNA expression and risk allele counts (0, 1 and 2) of both SNPs was evaluated with multivariable linear regression models adjusting for age and gender. Most of bladder tumors used for expression analysis (30 of 41 tumors) are of aggressive type (Supplementary Table 2).
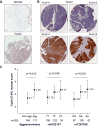
Figure 5. Immunohistochemistry (IHC) analysis of cyclin E expression in bladder tissues
(A) Images of cyclin E expression in one pair of tumor-adjacent normal bladder tissue samples, representative of 8 pairs tested. Immunostaining was done with an anti-CCNE1 rabbit polyclonal antibody (HPA018169, Sigma, dilution 1:85). Images are presented at 10× magnification with 0.1 mm scale bars. (B) Representative images of bladder tumors with nuclear cyclin E expression scores of 0, 2, 4 and 6, which is a sum of nuclear positivity (scored 0-3) and intensity (scored 0-3). The samples are a part of a TMA that includes 265 bladder tumors, immunostaining was done with an anti-CCNE1 rabbit monoclonal antibody (1655-1, Epitomics, dilution 1:250). (C) Association between IHC cyclin E nuclear scores in 265 bladder tumors, bladder cancer aggressiveness and genotypes of rs8102137 and rs7257330. The scores are shown as least squares mean values with 95% confidence intervals, based on multivariable linear regression models assuming additive effects of SNPs, with adjustment for age, gender, two study sub-sites, smoking, and categories of tumor aggressiveness, when appropriate: non-aggressive and aggressive.
References
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. -PubMed
- Stein JP, Lieskovsky G, Cote R, Groshen S, Feng AC, Boyd S, Skinner E, et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J Clin Oncol. 2001;19:666–675. -PubMed
- Hernandez S, Lopez-Knowles E, Lloreta J, Kogevinas M, Amoros A, Tardon A, Carrato A, et al. Prospective study of FGFR3 mutations as a prognostic factor in nonmuscle invasive urothelial bladder carcinomas. J Clin Oncol. 2006;24:3664–3671. -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- U01HG004446/HG/NHGRI NIH HHS/United States
- CA34627/CA/NCI NIH HHS/United States
- UM1 CA176726/CA/NCI NIH HHS/United States
- HHSN268201100046C/HL/NHLBI NIH HHS/United States
- DH_/Department of Health/United Kingdom
- R01CA65726/CA/NCI NIH HHS/United States
- R01 CA082838/CA/NCI NIH HHS/United States
- R01CA114665/CA/NCI NIH HHS/United States
- P01 CA87969/CA/NCI NIH HHS/United States
- HHSN268200782096C/HL/NHLBI NIH HHS/United States
- Z99 CA999999/ImNIH/Intramural NIH HHS/United States
- N01 CN045165/CN/NCI NIH HHS/United States
- 1R01CA114665/CA/NCI NIH HHS/United States
- CA082838/CA/NCI NIH HHS/United States
- R01 CA057494/CA/NCI NIH HHS/United States
- 16491/CRUK_/Cancer Research UK/United Kingdom
- U01HG004438/HG/NHGRI NIH HHS/United States
- N01-RC-45035/RC/CCR NIH HHS/United States
- P50 CA091846/CA/NCI NIH HHS/United States
- U01 HG004438/HG/NHGRI NIH HHS/United States
- T32 GM007814/GM/NIGMS NIH HHS/United States
- U01 HG004446/HG/NHGRI NIH HHS/United States
- P01 CA087969/CA/NCI NIH HHS/United States
- R01 CA67262/CA/NCI NIH HHS/United States
- R01 CA067262/CA/NCI NIH HHS/United States
- HHSN261201000006C/CP/NCI NIH HHS/United States
- P30 CA015704/CA/NCI NIH HHS/United States
- BHF_/British Heart Foundation/United Kingdom
- R01 CA065726/CA/NCI NIH HHS/United States
- N02 CP001037/CP/NCI NIH HHS/United States
- N02 CP011015/CP/NCI NIH HHS/United States
- P01 CA055075/CA/NCI NIH HHS/United States
- R01-CA089715/CA/NCI NIH HHS/United States
- HHSN268201100003C/WH/WHI NIH HHS/United States
- R01 CA089715/CA/NCI NIH HHS/United States
- R01 CA49449/CA/NCI NIH HHS/United States
- R01 CA114665/CA/NCI NIH HHS/United States
- N01 CN045035/CN/NCI NIH HHS/United States
- 1P01CA86871/CA/NCI NIH HHS/United States
- MRC_/Medical Research Council/United Kingdom
- HHSN271201100004C/AG/NIA NIH HHS/United States
- HHSN268201100002C/WH/WHI NIH HHS/United States
- R01HL091172-01/HL/NHLBI NIH HHS/United States
- P01 CA086871/CA/NCI NIH HHS/United States
- R01 CA034627/CA/NCI NIH HHS/United States
- UM1 CA167552/CA/NCI NIH HHS/United States
- N01 RC037004/RC/CCR NIH HHS/United States
- N02-CP-01037/CP/NCI NIH HHS/United States
- WT_/Wellcome Trust/United Kingdom
- R01 CA049449/CA/NCI NIH HHS/United States
- CA055075/CA/NCI NIH HHS/United States
- 001/WHO_/World Health Organization/International
- HHSN268201100001C/WH/WHI NIH HHS/United States
- HHSN268201100004C/WH/WHI NIH HHS/United States
- HHSN261200800001E/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical