Stem Cells in Teeth and Craniofacial Bones - PubMed (original) (raw)
Review
Stem Cells in Teeth and Craniofacial Bones
H Zhao et al. J Dent Res. 2015 Nov.
Abstract
Stem cells are remarkable, and stem cell-based tissue engineering is an emerging field of biomedical science aiming to restore damaged tissue or organs. In dentistry and reconstructive facial surgery, it is of great interest to restore lost teeth or craniofacial bone defects using stem cell-mediated therapy. In the craniofacial region, various stem cell populations have been identified with regeneration potential. In this review, we provide an overview of the current knowledge concerning the various types of tooth- and craniofacial bone-related stem cells and discuss their in vivo identities and regulating mechanisms.
Keywords: Gli1 protein; hedgehogs; mesenchymal stromal cells; neural crest; skull; stem cell niche.
© International & American Associations for Dental Research 2015.
Conflict of interest statement
The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
Figures
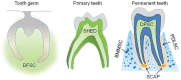
Figure 1.
The mesenchymal stem cell populations residing in the tooth–alveolar bone complex. Several mesenchymal stem cell populations have been identified from teeth of different developmental stages and associated alveolar bone, including dental pulp stem cells (DPSC), dental follicle stem cells (DFSC), stem cells from human exfoliated deciduous teeth (SHED), periodontal ligament stem cells (PDLSC), stem cells from the apical papilla (SCAP), and bone marrow mesenchymal stem cells (BMMSC).
Figure 2.
The neurovascular bundle niche and the in vivo origin of incisor mesenchymal stem cells. SHH is secreted by the sensory neurons in the trigeminal ganglion and transported through nerve axons into the incisor mesenchyme. SHH activates Gli1 expression in the stem cells surrounding the arterioles and regulates their differentiation. _Gli1_+ stem cells give rise to actively dividing transit amplifying cells and then differentiate into odontoblasts to support incisor mesenchyme turnover.
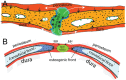
Figure 3.
The suture mesenchyme provides a niche for adult craniofacial bone mesenchymal stem cells (MSCs). (A) _Gli1_+ MSCs within the suture mesenchyme contribute to the periosteum, osteogenic front, and dura. (B) The osteogenic front secretes IHH to regulate the differentiation of _Gli1_+ MSCs in the suture mesenchyme.
References
- Akintoye SO, Lam T, Shi S, Brahim J, Collins MT, Robey PG. 2006. Skeletal site-specific characterization of orofacial and iliac crest human bone marrow stromal cells in same individuals. Bone. 38(6):758–768. -PubMed
- Aslan H, Zilberman Y, Kandel L, Liebergall M, Oskouian RJ, Gazit D, Gazit Z. 2006. Osteogenic differentiation of noncultured immunoisolated bone marrow-derived CD105+ cells. Stem Cells. 24(7):1728–1737. -PubMed
- Awad HA, Butler DL, Boivin GP, Smith FN, Malaviya P, Huibregtse B, Caplan AI. 1999. Autologous mesenchymal stem cell-mediated repair of tendon. Tissue Eng. 5(3):267–277. -PubMed
Publication types
MeSH terms
Grants and funding
- R01 DE022503/DE/NIDCR NIH HHS/United States
- R37 DE012711/DE/NIDCR NIH HHS/United States
- R90 DE022528/DE/NIDCR NIH HHS/United States
- U01 DE024421/DE/NIDCR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical