Bevacizumab alone or in combination with TRC105 for patients with refractory metastatic renal cell cancer - PubMed (original) (raw)
Randomized Controlled Trial
. 2017 Dec 1;123(23):4566-4573.
doi: 10.1002/cncr.30942. Epub 2017 Aug 22.
Affiliations
- PMID: 28832978
- PMCID: PMC5726867
- DOI: 10.1002/cncr.30942
Randomized Controlled Trial
Bevacizumab alone or in combination with TRC105 for patients with refractory metastatic renal cell cancer
Tanya B Dorff et al. Cancer. 2017.
Abstract
Background: Targeting the vascular endothelial growth factor (VEGF) pathway has improved outcomes in metastatic renal cell carcinoma (RCC); however, resistance inevitably occurs. CD105 (endoglin) is an angiogenic pathway that is strongly upregulated after VEGF inhibition, potentially contributing to resistance. The authors tested whether TRC105, a monoclonal antibody against endoglin, impacted disease control in patients with previously treated RCC who were receiving bevacizumab.
Methods: Eligible patients with metastatic RCC who had previously received 1 to 4 prior lines of therapy, including VEGF-targeted agents, were randomized 1:1 to receive bevacizumab 10 mg/kg intravenously every 2 weeks (arm A) or the same plus TRC105 10 mg/kg intravenously every 2 weeks (arm B). The primary endpoint was progression-free survival (PFS) at 12 and 24 weeks. Correlative studies included serum transforming growth factor β (TGFβ) and CD105 levels as well as tissue immunostaining for TGFβ receptors.
Results: Fifty-nine patients were enrolled (28 on arm A and 31 on arm B), and 1 patient on each arm had a confirmed partial response. The median PFS for bevacizumab alone was 4.6 months compared with 2.8 for bevacizumab plus TRC105 (P = .09). Grade ≥ 3 toxicities occurred in 16 patients (57%) who received bevacizumab compared with 19 (61%) who received bevacizumab plus TRC105 (P = .9). Baseline serum TGFβ levels below the median (<10.6 ng/mL) were associated with longer median PFS (5.6 vs 2.1 months; P = .014).
Conclusions: TRC105 failed to improve PFS when added to bevacizumab. TGFβ warrants further study as a biomarker in RCC. Cancer 2017;123:4566-4573. © 2017 American Cancer Society.
Keywords: angiogenesis; renal cancer; targeted therapy; transforming growth factor β (TGFβ); vascular endothelial growth factor (VEGF).
© 2017 American Cancer Society.
Conflict of interest statement
Conflict of interest disclosure:
TBD: consultant for Bayer, Genentech, Pfizer. Speaker for Astellas, Dendreon, Exelixis. JL: none. SKP: WS: MF: consultant to Bayer, Alkermes, Medivation; speaker for Dendreon. UV: AR: JP: consultant for Aeterna-Zentaris, Astellas, Bayer, Dendreon, Janssen, Millenium
Figures
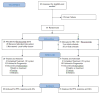
Figure 1
CONSORT diagram depicting subject accrual, randomization
Figure 2
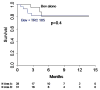
Kaplan-Meier Curves depicting (A) overall survival and (B) time to treatment failure for the study arms.
Figure 2
Kaplan-Meier Curves depicting (A) overall survival and (B) time to treatment failure for the study arms.
References
- Siegel RL, Miller KD, Jemal A. Cancer statistics 2016. CA Cancer J Clin. 2016;66:7–30. -PubMed
- Escudier B, Eisen T, Stadler WM, et al. Sorafenib in advanced renal cell carcinoma. New Engl J Med. 2007;356:125–34. -PubMed
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alpha in metastatic renal cell cancer. New Engl J Med. 2007;356:115–24. -PubMed
- Motzer RJ, Escudier B, Oudard S, et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomized, placebo-controlled trial. Lancet. 2008;372:449–56. -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P30 CA093373/CA/NCI NIH HHS/United States
- P30 CA014089/CA/NCI NIH HHS/United States
- UM1 CA186686/CA/NCI NIH HHS/United States
- P30 CA033572/CA/NCI NIH HHS/United States
- UM1 CA186717/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical