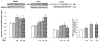A Combination of the Aerosolized PPAR-γ Agonist Pioglitazone and a Synthetic Surfactant Protein B Peptide Mimic Prevents Hyperoxia-Induced Neonatal Lung Injury in Rats - PubMed (original) (raw)
A Combination of the Aerosolized PPAR-γ Agonist Pioglitazone and a Synthetic Surfactant Protein B Peptide Mimic Prevents Hyperoxia-Induced Neonatal Lung Injury in Rats
Reiko Sakurai et al. Neonatology. 2018.
Abstract
Background: Despite improvements in perinatal care, bronchopulmonary dysplasia (BPD) in extremely premature infants has not decreased. Postnatal surfactant therapy provides symptomatic relief from respiratory distress syndrome, but does not translate into a reduction in BPD. Therefore, the search for effective interventions to prevent BPD continues.
Objectives: Since PPAR-γ agonists have been demonstrated to promote neonatal lung maturation and injury repair, we hypothesized that a formulation of a PPAR-γ agonist, pioglitazone (PGZ) and a synthetic lung surfactant (a surfactant protein B peptide mimic, B-YL) combined would stimulate lung maturation and block hyperoxia-induced neonatal lung injury more effectively than either modality alone.
Methods: One-day-old Sprague-Dawley rat pups were administered PGZ + B-YL via nebulization every 24 h for up to 72 h. The pups were exposed to either 21 or 95% O2, and then sacrificed. Their lungs were examined for markers of lung maturation (levels of PPAR-γ, SP-C and choline-phosphate cytidylyltransferase [CCT-α] and [3H]triolein uptake) and injury repair (bronchoalveolar lavage cell count and protein content, and levels of LEF-1, fibronectin, ALK5, and β-catenin) by Western blot analysis.
Results: Markers of alveolar epithelial/mesenchymal maturation (PPAR-γ, SP-C, CCT-α, and triolein uptake) increased significantly in the PGZ + B-YL group, more than with either drug alone. Similarly, markers of hyperoxia-induced lung injury were blocked effectively with PGZ + B-YL treatment.
Conclusions: Nebulized PPAR-γ agonist PGZ with a synthetic lung surfactant accelerates lung maturation and prevents neonatal hyperoxia-induced lung injury more than either modality alone, with the potential to provide more effective prevention of BPD.
Keywords: Bronchopulmonary dysplasia; Chronic lung disease; Lung maturation; Prematurity; Respiratory distress syndrome.
© 2018 S. Karger AG, Basel.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Fig. 1. Predicted molecular model of B-YL derived from the primary amino acid sequence using the homology templating program i-Tasser [26]
The secondary structure of the SP-B peptide mimic B-YL is that of a helix hairpin with the helical elements shown as green ribbons connected by a bend domain shown as a green tube. The helix hairpin structure is stabilized by the interaction of the tyrosine side chains in red highlight at the N and C terminal domains that emulate the disulfide interactions and the Saposin protein fold of the native SP-B sequence.
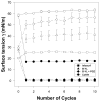
Fig. 2. Comparison of B-YL, PGZ+B-YL, lipids alone, and Infasurf® on surface tension reduction activity
Surface activity of the B-YL (100 mg/kg) was measured with captive bubble surfactometry in the presence and absence of PGZ (1 mg/kg) and compared with a clinical surfactant (Infasurf®) as positive control and lipids alone (DPPC:POPC:POPG 5:3:2 wt:wt:wt) as negative control. Minimum surface tension values are indicated with black symbols and maximum values with similar white symbols. Minimum surface tension values of B-YL surfactant ± PGZ were similar to those of Infasurf®. Values are mean ± SEM of N=4–5.
Fig. 3. Effect of B-YL on PGZ’s PPARγ agonist activity
Embryonic day 19 fetal rat lung explants, cultured for 24 h, under either control condition (without B-YL or PGZ supplementation) or treated with B-YL (100 mg/kg BW) alone or with PGZ (0.5, 1 or 2 mg/kg BW) + B-YL (100 mg/kg BW), demonstrated significant increases in PPARγ, SP-C, and CCT-α protein levels, as determined by Western analysis, along with a significant increase in [3H]choline incorporation into disaturated phosphatidylcholine (p <0.05 vs. control; N=3).
Fig. 4. Nebulized PGZ+B-YL blocks hyperoxia-induced alterations in selective markers of neonatal lung maturation more effectively than either modality alone
Postnatal day one rat pups were exposed to hyperoxia ± B-YL, PGZ, or PGZ+B-YL for 72 h. Hyperoxia-induced decrease in PPARγ (A), SP-C (B), CCT-α (C) and increase in LEF-1 (D) and Fibronectin (E) protein levels were blocked by concomitant administration of either PGZ or PGZ+B-YL. This was also true for hyperoxia-induced decrease in triolein uptake (F). PGZ+B-YL administration had a more robust effect, as evidenced by the effects on the protein levels of SP-C and CCT-α and the triolein uptake; however, B-YL administration alone had no effect on any of these parameters. *, p<0.05 vs. control; #, p<0.05 vs. 95% O2; control; $, p< 0.05 vs. PGZ; N=3.
Fig. 4. Nebulized PGZ+B-YL blocks hyperoxia-induced alterations in selective markers of neonatal lung maturation more effectively than either modality alone
Postnatal day one rat pups were exposed to hyperoxia ± B-YL, PGZ, or PGZ+B-YL for 72 h. Hyperoxia-induced decrease in PPARγ (A), SP-C (B), CCT-α (C) and increase in LEF-1 (D) and Fibronectin (E) protein levels were blocked by concomitant administration of either PGZ or PGZ+B-YL. This was also true for hyperoxia-induced decrease in triolein uptake (F). PGZ+B-YL administration had a more robust effect, as evidenced by the effects on the protein levels of SP-C and CCT-α and the triolein uptake; however, B-YL administration alone had no effect on any of these parameters. *, p<0.05 vs. control; #, p<0.05 vs. 95% O2; control; $, p< 0.05 vs. PGZ; N=3.
Fig. 5. Nebulized PGZ+B-YL blocks hyperoxia-induced alterations in selective markers of neonatal lung injury more effectively than either modality alone
Postnatal day one rat pups were exposed to hyperoxia ± B-YL, PGZ, or PGZ+B-YL for 72 h. Hyperoxia-induced alterations in bronchoalveolar lavage total cell count(A) and protein content (B), apoptosis marker BcL2/Bax ratio (C) and inflammatory markers IL-6 and interferon-γ (D) were blocked by concomitant administration of either PGZ or PGZ+B-YL. *, p<0.05 vs. cont; #, p<0.05 vs. 95% O2; cont; $, p< 0.05 vs. PGZ N=3.
Fig. 5. Nebulized PGZ+B-YL blocks hyperoxia-induced alterations in selective markers of neonatal lung injury more effectively than either modality alone
Postnatal day one rat pups were exposed to hyperoxia ± B-YL, PGZ, or PGZ+B-YL for 72 h. Hyperoxia-induced alterations in bronchoalveolar lavage total cell count(A) and protein content (B), apoptosis marker BcL2/Bax ratio (C) and inflammatory markers IL-6 and interferon-γ (D) were blocked by concomitant administration of either PGZ or PGZ+B-YL. *, p<0.05 vs. cont; #, p<0.05 vs. 95% O2; cont; $, p< 0.05 vs. PGZ N=3.
Fig. 6. Nebulized PGZ+B-YL blocks hyperoxia-induced activations of TGF-β and Wnt signaling pathways
Postnatal day one rat pups were exposed to hyperoxia ± PGZ+B-YL for 72h. Hyperoxia-induced activation in TGF-β [determined by ALK5 protein levels, red staining) and Wnt (determined by β-catenin protein levels, green staining) signaling was blocked by concomitant PGZ+B-YL administration. Representative immunostaining pictures and relative mean fluorescence values are shown; N=3.
Fig. 6. Nebulized PGZ+B-YL blocks hyperoxia-induced activations of TGF-β and Wnt signaling pathways
Postnatal day one rat pups were exposed to hyperoxia ± PGZ+B-YL for 72h. Hyperoxia-induced activation in TGF-β [determined by ALK5 protein levels, red staining) and Wnt (determined by β-catenin protein levels, green staining) signaling was blocked by concomitant PGZ+B-YL administration. Representative immunostaining pictures and relative mean fluorescence values are shown; N=3.
References
- Martin RJ, Fanaroff AA. The preterm lung and airway: past, present, and future. Pediatr Neonatol. 2013;54(4):228–234. -PubMed
- Silva DM, Nardiello C, Pozarska A, Morty RE. Recent advances in the mechanisms of lung alveolarization and the pathogenesis of bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol. 2015;309(11):L1239–72. -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R21 HL107118/HL/NHLBI NIH HHS/United States
- R21 HD071731/HD/NICHD NIH HHS/United States
- R01 HD051857/HD/NICHD NIH HHS/United States
- R01 HL075405/HL/NHLBI NIH HHS/United States
- R01 HL127237/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources