Pembrolizumab plus dinaciclib in patients with hematologic malignancies: the phase 1b KEYNOTE-155 study - PubMed (original) (raw)
Clinical Trial
. 2022 Feb 22;6(4):1232-1242.
doi: 10.1182/bloodadvances.2021005872.
Shaji Kumar 2, Ding Wang 3, Daruka Mahadevan 4, Patricia Walker 5, Nina Wagner-Johnston 6, Carolina Escobar 7, Rajat Bannerji 8, Divaya Bhutani 9, Julie Chang 10, Francisco J Hernandez-Ilizaliturri 11, Andreas Klein 12, John M Pagel 13, Witold Rybka 14, Andrew J Yee 15, Anne Mohrbacher 16, Mo Huang 17, Mohammed Farooqui 17, Patricia Marinello 17, Hang Quach 18
Affiliations
- PMID: 34972202
- PMCID: PMC8864641
- DOI: 10.1182/bloodadvances.2021005872
Clinical Trial
Pembrolizumab plus dinaciclib in patients with hematologic malignancies: the phase 1b KEYNOTE-155 study
Gareth P Gregory et al. Blood Adv. 2022.
Abstract
Preclinical data demonstrated that combining an anti-programmed cell death 1 (PD-1) inhibitor with a cyclin-dependent kinase 9 (CDK9) inhibitor provided enhanced antitumor activity with no significant toxicities, suggesting this combination may be a potential therapeutic option. The multicohort, phase 1 KEYNOTE-155 study evaluated the safety and antitumor activity of the PD-1 inhibitor pembrolizumab plus the CDK9 inhibitor dinaciclib in patients with relapsed or refractory (rr) chronic lymphocytic leukemia (CLL), diffuse large B-cell lymphoma (DLBCL) and multiple myeloma (MM). Patients enrolled were ≥18 years of age with a confirmed diagnosis of CLL, DLBCL, or MM. The study included 2 phases: a dose-evaluation phase to determine dose-limiting toxicities and a signal-detection phase. Patients received pembrolizumab 200 mg every 3 weeks plus dinaciclib 7 mg/m2 on day 1 and 10 mg/m2 on day 8 of cycle 1 and 14 mg/m2 on days 1 and 8 of cycles 2 and later. Primary endpoint was safety, and a key secondary endpoint was objective response rate (ORR). Seventy-two patients were enrolled and received ≥1 dose of study treatment (CLL, n = 17; DLBCL, n = 38; MM, n = 17). Pembrolizumab plus dinaciclib was generally well tolerated and produced no unexpected toxicities. The ORRs were 29.4% (5/17, rrCLL), 21.1% (8/38, rrDLBCL), and 0% (0/17, rrMM), respectively. At data cutoff, all 72 patients had discontinued treatment, 38 (52.8%) because of progressive disease. These findings demonstrate activity with combination pembrolizumab plus dinaciclib and suggest that a careful and comprehensive approach to explore anti-PD-1 and CDK9 inhibitor combinations is warranted. This trial was registered at www.clinicaltrials.gov as NCT02684617.
© 2022 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Figures
Graphical abstract
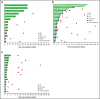
Figure 1.
Time on study and response duration* per investigator assessment. Patients with rrCLL (A), rrDLBCL (B), and rrMM (C). CR, complete response; PR, partial response. *Tumor response as assessed by investigator review according to International Workshop on CLL guidelines (2008) for the rrCLL cohort, revised International Working Group response criteria for malignant lymphoma (2007) for the rrDLBCL cohort, and International Myeloma Working Group Uniform Response Criteria (2006) for the rrMM cohort.
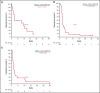
Figure 2.
Kaplan-Meier estimates of PFS. Patients with rrCLL (A), rrDLBCL (B), and rrMM (C).
Figure 3.
Kaplan-Meier estimates of overall survival. Patients with relapsed or refractory rrCLL (A), rrDLBCL (B), and rrMM (C).
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Version 1.2021. Chronic Lymphocytic leukemia/Small Lymphocytic Lymphoma. Vol. 2020. Plymouth Meeting, PA: National Comprehensive Cancer Network; 2020.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous