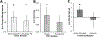Gamma neuromodulation improves episodic memory and its associated network in amnestic mild cognitive impairment: a pilot study - PubMed (original) (raw)
doi: 10.1016/j.neurobiolaging.2023.04.005. Epub 2023 Apr 20.
Courtney L Gallen 2, Avery E Ostrand 2, Julio C Rojas 3, Peter Wais 2, James Rini 2, Brandon Chan 3, Argentina Lario Lago 3, Adam Boxer 3, Min Zhao 4, Adam Gazzaley 5, Theodore P Zanto 6
Affiliations
- PMID: 37276822
- PMCID: PMC10583532
- DOI: 10.1016/j.neurobiolaging.2023.04.005
Gamma neuromodulation improves episodic memory and its associated network in amnestic mild cognitive impairment: a pilot study
Kevin T Jones et al. Neurobiol Aging. 2023 Sep.
Abstract
Amnestic mild cognitive impairment (aMCI) is a predementia stage of Alzheimer's disease associated with dysfunctional episodic memory and limited treatment options. We aimed to characterize feasibility, clinical, and biomarker effects of noninvasive neurostimulation for aMCI. 13 individuals with aMCI received eight 60-minute sessions of 40-Hz (gamma) transcranial alternating current stimulation (tACS) targeting regions related to episodic memory processing. Feasibility, episodic memory, and plasma Alzheimer's disease biomarkers were assessed. Neuroplastic changes were characterized by resting-state functional connectivity (RSFC) and neuronal excitatory/inhibitory balance. Gamma tACS was feasible and aMCI participants demonstrated improvement in multiple metrics of episodic memory, but no changes in biomarkers. Improvements in episodic memory were most pronounced in participants who had the highest modeled tACS-induced electric fields and exhibited the greatest changes in RSFC. Increased RSFC was also associated with greater hippocampal excitability and higher baseline white matter integrity. This study highlights initial feasibility and the potential of gamma tACS to rescue episodic memory in an aMCI population by modulating connectivity and excitability within an episodic memory network.
Keywords: Amnestic mild cognitive impairment; Episodic memory; Gamma stimulation; Neurostimulation; Resting-state functional connectivity.
Published by Elsevier Inc.
Conflict of interest statement
Disclosure statement A.G. is a scientific advisor for Neuroelectrics, which is the company that makes the neurostimulation device used in the current study.
Figures
Fig. 1.
(A) Weekly timeline for the study protocol. For Week 2, participants received tACS while completing the mnemonic discrimination task (MDT), fluency task, and a tablet game, collectively referred to as the ‘Stimulation Tasks.’ (B) The California Verbal Learning Test (CVLT) paradigm and timing. Following encoding of the words participants completed a distractor list and then the short-delay free recall (SDFR). Following a 20-minute break participant completed the long-delay free recall (LDFR). Following the next 10-minute break participants completed the forced-choice recognition section. (C) Paired Associated Task (PAT) paradigm and timing. Participants completed the PAT during the 20-minute break of the CVLT. (D) The Mnemonic Discrimination Task (MDT) paradigm and timing. Eight unique sets of stimuli were used during each MDT task conducted concurrently with each tACS session.
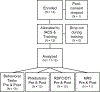
Fig. 2.. Flow chart of participant enrollment, data collection, and analysis stages. One participant withdrew prior to completing any tasks.
One participant had excessive movement during the RSFC at follow-up. One participant had no DTI scan due to time constraints at baseline. Two participants had an error in the MRS protocol at baseline.
Fig. 3.. Behavioral change following 8 sessions of gamma tACS over 4 weeks.
(A) Change from baseline on the CVLT SDFR and LDFR. (B) Change from baseline on the PAT recall and recognition portions completed during the 20-minute break in the CVLT. (C) Change from baseline on verbal and semantic fluency tasks. Parentheses represent the standard deviation of the mean. * = p < 0.05, ~ = p < 0.10.
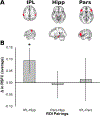
Fig. 4.. Change in RSFC between ROI pairings from pre-tACS to post-tACS MRI sessions.
(A) Axial (top) and sagittal (bottom) view of ROI masks in MNI space for the inferior parietal lobule (IPL), hippocampi (Hipp), and Pars triangularis (Pars), marked in red. (B) Changes in average RSFC between ROI pairings from pre-tACS to post-tACS MRI sessions. Data presented with ‘outlier’ data point included. * = p < .05.
Fig. 5.
Spearman’s correlations between the change in IPL-Hipp RSFC and (A) the change in SDFR on the CVLT, (B) change in E/I ratio within the Hipp and (C) baseline FA in the Fornix (black diamonds), Arcuate Fasciculus (dark gray diamonds), SLF (light gray circles), and Uncinate (white squares) white matter tracts.
Fig. 6.
Spearman’s correlations between the modeled EF and (A) the change in SDFR on the CVLT and (B) the change in verbal fluency. Color represents significant correlation.
References
- Aslaksen PM, Bystad MK, Ørbo MC, Vangberg TR, 2018. The relation of hippocampal subfield volumes to verbal episodic memory measured by the California Verbal Learning Test II in healthy adults. Behav. Brain Res. 351, 131–137. . https://www.sciencedirect.com/science/article/pii/S0166432818300597. -DOI -PubMed
Publication types
MeSH terms
Grants and funding
- K24 AG045333/AG/NIA NIH HHS/United States
- R21 AG060335/AG/NIA NIH HHS/United States
- U19 AG063911/AG/NIA NIH HHS/United States
- P01 AG019724/AG/NIA NIH HHS/United States
- K23 AG059888/AG/NIA NIH HHS/United States