Metastatic gastric cancer target lesion complete response with Claudin18.2-CAR T cells - PubMed (original) (raw)
. 2024 Feb 5;12(2):e007927.
doi: 10.1136/jitc-2023-007927.
Joseph Chao 3, Hong Ma 4, Michael Hahn 5, Gloria Sierra 4, Jie Jia 4, Amanda Y Hendrix 4, Joy V Nolte Fong 4, Audrey Ween 6, Peter Vu 6 2, Aaron Miller 6, Michael Choi 6 2, Benjamin Heyman 6 2, Gregory A Daniels 6 2, Dan Kaufman 6 2, Catriona Jamieson 6 2, Zonghai Li 4, Ezra Cohen 6
Affiliations
- PMID: 38316518
- PMCID: PMC10860094
- DOI: 10.1136/jitc-2023-007927
Metastatic gastric cancer target lesion complete response with Claudin18.2-CAR T cells
Gregory P Botta et al. J Immunother Cancer. 2024.
Abstract
Treatment of hematologic malignancies with patient-derived anti-CD19 chimeric antigen receptor (CAR) T-cells has demonstrated long-term remissions for patients with otherwise treatment-refractory advanced leukemia and lymphoma. Conversely, CAR T-cell treatment of solid tumors, including advanced gastric cancer (GC), has proven more challenging due to on-target off-tumor toxicities, poor tumor T-cell infiltration, inefficient CAR T-cell expansion, immunosuppressive tumor microenvironments, and demanding preconditioning regimens. We report the exceptional results of autologous Claudin18.2-targeted CAR T cells (CT041) in a patient with metastatic GC, who had progressed on four lines of combined systemic chemotherapy and immunotherapy. After two CT041 infusions, the patient had target lesion complete response and sustained an 8-month overall partial response with only minimal ascites. Moreover, tumor-informed circulating tumor DNA (ctDNA) reductions coincided with rapid CAR T-cell expansion and radiologic response. No severe toxicities occurred, and the patient's quality of life significantly improved. This experience supports targeting Claudin18.2-positive GC with CAR T-cell therapy and helps to validate ctDNA as a biomarker in CAR T-cell therapy. Clinical Insight: Claudin18.2-targeted CAR T cells can safely provide complete objective and ctDNA response in salvage metastatic GC.
Keywords: Gastrointestinal Neoplasms; Receptors, Chimeric Antigen; Therapies, Investigational; Translational Medical Research.
© Author(s) (or their employer(s)) 2024. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: HM, GS, JJ, AYH, JVNF and ZL are employees of CARsgen Therapeutics Corporation.
Figures
Figure 1
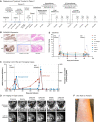
Clinical timeline of Patient 1 treated with CT041. (A) Initial diagnosis, treatment history, and follow-up visits for Patient 1, who received two CT041 infusions, 250×106 CAR-T cells each. (B) CLDN18.2 expression in gastric biopsy via immunohistochemistry, 5× and 40× magnification. (C) Tumor-informed circulating tumor DNA (ctDNA) in plasma measured in mean tumor molecules (MTM)/mL via qPCR and CAR-transgene copies/μg genomic DNA in peripheral blood via qPCR. Open spheres, ctDNA negative; closed spheres, ctDNA positive. (D) Complete response of target lesion at peritoneal nodules (arrows, top) and decreasing loculated ascites from left midabdomen (arrows, bottom) via contrast-enhanced axial CT scans. (E) Serum cytokines including interleukins, TNF-α, VEGF-A, and IFN-γ via enzyme-linked immunoassays. Fold changes normalized to Day 0 value (first infusion). (F) Skin rash following first CT041 infusion. CAR, chimeric antigen receptor; CLDN18.2, Claudin18.2; CT041, Claudin18.2-targeted CAR T cells; DCF, docetaxel, cisplatin, 5-fluorouracil; dx, diagnosis; FOLFIRI, 5, fluorouracil, leucovorin, irinotecan; FOLFOX, 5, fluorouracil, leucovorin, oxaliplatin; HIPEC, hyperthermic intraperitoneal chemotherapy; IFN, interferon; IL, interleukin; m, months qPCR, quantitative PCR; TNF, tumor necrosis factor; tx, treatment; VEGF, vascular endothelial growth factor, w/wks, weeks.
Figure 2
Characterization of CT041 CAR-T-cell product. (A) Schematic of an autologous CT041 CAR-CLDN18.2 T cell consisting of an extracellular humanized anti-CLDN18.2 scFv (blue) and a CD8α hinge (purple), connected to an intracellular CD28 costimulatory domain (cyan) and a CD3ζ activating domain (brown) by a CD28 transmembrane domain (dark green). (B) Cell viability (black) and growth rate (orange) of CT041 during the 10-day manufacturing process, determined daily using NC 200 cell counter. (C) T-cell subpopulations, characterized and quantified by flow cytometry analysis using different cell-surface markers, including total T cells (CD3+CD45+) (upper left); T helper cells (CD4+CD3+CD45+) and cytotoxic T cells (CD8+CD3+CD45+) (upper right); central memory T cells (CD62L+CD45RA−CD3+CD45+), naïve T cells (CD62L+CD45RA+CD3+CD45+), effector memory T cells (CD62L−CD45RA−CD3+CD45+), effector T cells (CD62L−CD45RA+CD3+CD45+) (bottom left); and natural killer T cells (CD56+CD3+CD45+) (bottom right). (D) Transduction efficiency of CT041 as quantified by flow cytometry with anti-CD3 antibody plus PE-CAR. (E) T-cell subpopulations as determined at different time points by flow cytometry during manufacturing.
References
- Stomach cancer — cancer STAT facts. n.d. Available: https://seer.cancer.gov/statfacts/html/stomach.html
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous