Trance, a Tumor Necrosis Factor Family Member, Enhances the Longevity and Adjuvant Properties of Dendritic Cells in Vivo (original) (raw)
Abstract
Mature dendritic cells (DCs) are powerful antigen presenting cells that have the unique capacity to migrate to the T cell zone of draining lymph nodes after subcutaneous injection. Here we report that treatment of antigen-pulsed mature DCs with tumor necrosis factor (TNF)-related activation-induced cytokine (TRANCE), a TNF family member, before immunization enhances their adjuvant capacity and elicits improved T cell priming in vivo, such that both primary and memory T cell immune responses are enhanced. By enumerating migratory DCs in the draining lymph nodes and by studying their function in stimulating naive T cells, we show that one of the underlying mechanisms for enhanced T cell responses is an increase in the number of ex vivo antigen-pulsed DCs that are found in the T cell areas of lymph nodes. These results suggest that the longevity and abundance of mature DCs at the site of T cell priming influence the strength of the DC-initiated T cell immunity in situ. Our findings have the potential to improve DC-based immunotherapy; i.e., the active immunization of humans with autologous DCs that have been pulsed with clinically significant antigens ex vivo.
Keywords: TRANCE, dendritic cells, T cell, immunization
Introduction
Dendritic cells (DCs) are antigen-presenting cells specialized to initiate T cell immunity, and as such can be regarded as nature's adjuvant 1 2. DCs have the capacity in vivo to migrate from tissues, where they encounter antigens, to the T cell areas of lymphoid tissues, where immune responses begin 2 3 4 5. DCs reside in tissues as immature cells, specialized to capture and process antigens, and, after antigen capture, they mature in response to inflammatory stimuli characterized by an upregulation of MHC and T cell costimulatory molecules 2. DCs then migrate to the draining LN and stimulate antigen-specific T cells to initiate an immune response. However, several reports suggest that the life span of DCs in the LN is short. Although DCs migrate to the LNs via afferent lymphatics, they are not found in the efferent lymph 6 7. It is difficult to identify DCs in the LN >2 d after subcutaneous injection 4 5. In tissue culture, fully differentiated or mature DCs undergo rapid apoptosis, but this can be delayed substantially by treatment with several members of the TNF family, including CD40 ligand (CD40L), TNF-related activation-induced cytokine (TRANCE), and TNF 8.
TRANCE, a recently described member of the TNF family 9, is a regulator of the immune system and of bone development (for a review, see reference 10). TRANCE is expressed on activated T cells 11, and a major target for TRANCE in the immune system appears to be mature DCs that express a high level of TRANCE receptor (TRANCE-R) 8 12. In vitro, TRANCE promotes the survival of mature DCs, most likely by upregulating the expression of Bcl-XL 8, and induces the production of proinflammatory cytokines, such as IL-1 and IL-6, and cytokines that stimulate and induce differentiation of T cells, such as IL-12 and IL-15 11 13. Therefore, TRANCE is likely to act as a positive feedback regulator during productive T cell–DC interactions 10.
In this study, we have treated antigen-pulsed mature DCs with soluble TRANCE, injected the cells subcutaneously, and then evaluated their numbers and function in LNs. We have found that TRANCE treatment greatly enhances the number and persistence of antigen-presenting DCs in the draining LNs, and increases the ensuing primary and memory T cell responses. Thus, our results indicate that the longevity and abundance of antigen-pulsed DCs are critical factors in the magnitude of a T cell response to antigen in vivo, and provide a potential avenue to improve the efficacy of DC-based immunotherapy in humans.
Materials and Methods
Dendritic Cells.
DCs were generated from bone marrow progenitors, using GM-CSF as described 14 15. On day 6 of culture, the immature DCs were pulsed for 6 h with 10 μg/ml purified protein derivative (PPD) from Mycobacterium tuberculosis (Statens Serum Institute) or OVA (Sigma Chemical Co.) in complete medium (RPMI 1640 supplemented with 5% FCS, 50 μM 2-ME, and 20 μg/ml gentamicin). The cells were then dislodged and transferred to fresh 100-mm culture dishes at 106/ml to allow DC maturation. On day 7, the mature DCs were pulsed overnight with recombinant, soluble TRANCE (1 μg/ml) 8. On day 8, the DCs were washed twice in HBSS and resuspended in HBSS for injection into the footpads of mice (5 × 105 cells per footpad).
Proliferation Assays for T Cell Priming and Cytokine Production.
Draining LNs (popliteal LNs), cervical LNs, and spleen were harvested from different groups of mice (C57BL/6 or BALB/c; Taconic Farms), and single cell suspensions were prepared. Cells were restimulated in vitro with 10-fold serial dilutions of PPD (0.1–10 μg/ml) or OVA, for 3 d in triplicate in flat-bottomed 96-well plates at 3 × 105 LN cells/well in 200 μl of Click's medium (GIBCO BRL) supplemented with 0.5% normal mouse serum, l-glutamine, penicillin-streptomycin, Hepes, and 2-ME. [3H]thymidine was added for the last 8 h of culture, and thymidine incorporation was measured using standard scintillation procedures. For cytokine production, cells prepared as described above were cultured at 3 × 106/ml in complete Click's medium in the presence of 10 μg/ml of PPD or OVA for 48 h. Supernatants were then assessed for IFN-γ and IL-4 by ELISA (R&D Systems).
Presentation of Antigens by DCs That Have Migrated to LNs.
LN DCs were purified using CD11c-MACS beads (Miltenyi Biotec) after digestion in 400 U/ml collagenase following the manufacturer's instructions. DO11-10 CD4+ T cells were purified from LNs of DO11-10 TCR transgenic mice (provided by Dr. Dennis Loh) using CD4 magnetic beads (Miltenyi Biotec). 5 × 104 DO11-10 CD4+ T cells were cultured in RPMI 1640 supplemented with 10% FCS, 2-ME, l-glutamine, penicillin-streptomycin, with various numbers of CD11c+ cells in round-bottomed 96-well plates for 4 d and pulsed with [3H]thymidine during the last 8 h of culture.
Measurement of Delayed-Type Hypersensitivity.
4.5 or 9 wk after priming with antigen-pulsed DCs in the left hind footpad, mice were challenged by the injection of 30 μg PPD in PBS in the right hind footpad, and PBS only in the left footpad. Footpad swelling was measured after 24 and 48 h and recorded as the difference between right and left footpad thickness.
Labeling and Migration of DCs.
To follow the migration of bone marrow–derived DCs in draining LNs after footpad injection, cells were labeled before injection with 5-chloromethyl-fluorescein diacetate (CMFDA) or 5-(and 6)-(((4-chloromethyl)benzoyl)amino)tetramethylrhodamine (CMTMR) according to the manufacturer's protocol (Molecular Probes). DCs were incubated for 30 min at 37°C in 10 μM CMFDA or CMTMR in serum-free RPMI, and then another 30 min at 37°C in serum-free RPMI. The DCs were washed, counted, and injected in both hind footpads (5 × 105 per footpad). For confocal microscopy, the draining popliteal LNs were harvested at various time points after injection, processed, and analyzed by confocal microscopy as described previously 5. For FACS® analysis, popliteal LNs were harvested, then digested with 400 U/ml collagenase (Sigma Chemical Co.) for 30 min at 37°C in RPMI, 1% FCS. EDTA at a final concentration of 10 mM was added during the last 5 min. Total LN cells were counted and then stained with PE-conjugated anti-CD11c mAb (PharMingen). After gating on live cells, 106 events were acquired to quantify the absolute number of migrating DCs per LN (CD11c+ and FL1high).
Results
Enhancement of Antigen-specific Primary Th Cell Responses by TRANCE-treated DCs.
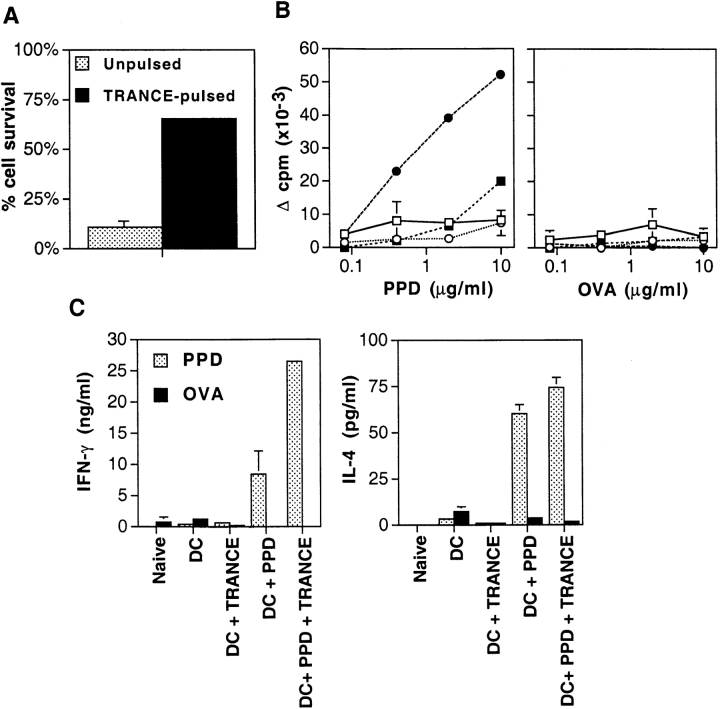
To determine whether the pretreatment of antigen-pulsed mature DCs ex vivo with TNF family members such as TRANCE would enhance the adjuvant effect of DCs in vivo, we used the PPD from mycobacteria, a classical antigen for T cell–mediated immunity and an antigen known to be presented by DCs in mice 14. DCs were prepared from bone marrow progenitors stimulated with GM-CSF 14 15. PPD was added for 6 h on day 6 of the cultures, when most of the DCs were in an immature state. These immature DCs display efficient endocytic activity 14 and sequester most of their antigen-presenting MHC class II products within intracellular endocytic compartments (MIICs 16), but lack strong cell surface expression of CD40, TRANCE-R, and CD86 10. After the 6-h exposure to low doses of PPD, DC maturation was induced by transfer to a new culture vessel as described 14 15. We confirmed that the DCs began to express high levels of CD40, TRANCE-R, and CD86 upon maturation 10. The mature DCs were then treated with or without soluble TRANCE during the last 12 h of the culture, washed, and injected subcutaneously in the hind footpads of naive mice. Aliquots of each DC preparation were further cultured in vitro to test whether a brief exposure to TRANCE was sufficient to increase DC survival. A brief exposure of mature DCs to TRANCE was indeed sufficient to enhance DC survival in vitro (Fig. 1 A), suggesting that cellular responses induced by TRANCE responsible for antiapoptotic effects on mature DCs are initiated at an early point after TRANCE stimulation 8.
Figure 1.
TRANCE enhances the capacity of DCs to act as adjuvants for T cell priming. (A) Antigen-pulsed mature DCs were treated with 1 μg/ml soluble TRANCE for 12 h, washed three times, and cultured for 2 d to determine the effect of TRANCE treatment on DC viability. The results represent the mean ± SD. (B) Immature bone marrow–derived DCs were pulsed for 6 h with (filled symbols) or without (open symbols) 10 μg/ml PPD. The cells were dislodged and transferred to new 100-mm culture dishes for 48 h to allow maturation. During the last 12 h, the cells were pulsed with 1 μg/ml soluble TRANCE (circles) or with medium alone (squares). The DCs were then extensively washed, and 2 × 105 cells were injected in the left hind footpad of anesthetized naive mice. 7 d later, draining popliteal LN cells were prepared and cultured for 3 d at 3 × 105 cells/well in 200 μl of complete Click's medium supplemented with 0.5% normal mouse serum in flat-bottomed 96-well plates in the presence of various concentrations of PPD. Proliferation was assessed during the last 8 h of culture by pulsing the cells with 0.5 μCi [3H]thymidine. Each point represents the mean ± SD of three mice per group at each time point. Results from one experiment representative of five are shown. (C) As in B, but supernatants of the LN cultures with the highest antigen concentration were harvested after 48 h to measure IFN-γ (left) and IL-4 (right) production by ELISA. Results of one experiment representative of three are shown. The results represent the mean ± SD.
The immunogenicity of the subcutaneously injected DCs was then assessed after in vitro restimulation and measurement of antigen-specific T cell proliferation and cytokine production. PPD-specific proliferative responses by T cells from draining popliteal LNs were induced when mice were immunized with PPD-pulsed DCs (Fig. 1 B), but not with PPD alone (data not shown), confirming that DCs act as adjuvants 3. The PPD-specific T cell responses from mice immunized with TRANCE-treated, PPD-pulsed DCs were much higher than those from mice immunized with PPD-pulsed DCs without prior treatment of TRANCE. Approximately 20-fold more PPD was required to show a similar level of proliferation by draining LN T cells from mice immunized with DCs that were PPD-pulsed without treatment with TRANCE (Fig. 1 B). In controls, the LN cells did not respond to a nonspecific protein, OVA (Fig. 1B and Fig. C). Primed popliteal LN cells from mice immunized with PPD-pulsed DCs also showed strong cytokine production (IFN-γ and IL-4) upon rechallenge with PPD (Fig. 1 C). Immunization with TRANCE-treated, PPD-pulsed DCs also increased the production of IFN-γ by severalfold, but the production of IL-4 was not affected (Fig. 1 C).
No priming was found in LNs that were distal to the site of DC injection, namely mesenteric and axillary LNs, whether DCs were treated with TRANCE or not (data not shown). However, when spleen cells were examined at a later time point (day 10), a small but significant response to PPD was noted (data not shown), suggesting a migration of primed T cells from the popliteal LNs to the spleen. Furthermore, this response was significantly higher in spleen cells from mice injected with TRANCE-treated, PPD-pulsed DCs compared with that from mice immunized with DCs that were PPD-pulsed without TRANCE treatment (data not shown). Similar results were also obtained when OVA was used as antigen (data not shown). Therefore, these results indicate that TRANCE-treated, antigen-pulsed DCs have an increased capacity to induce CD4+ T cell priming in vivo.
Enhancement of Antigen-specific Memory Responses by TRANCE-treated DCs.
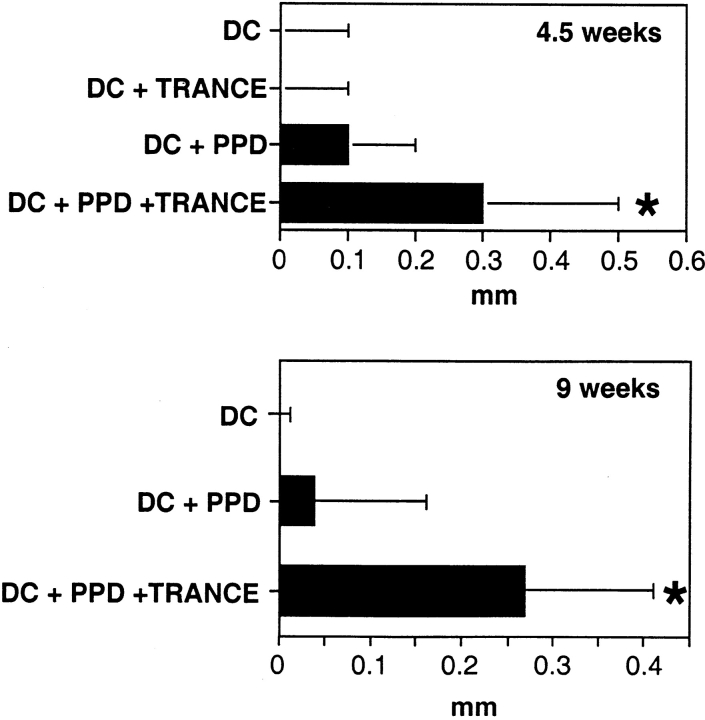
The effects of DCs on the establishment of memory was then tested using the induction of delayed-type hypersensitivity (DTH) as a criterion. When mice were immunized with PPD-pulsed DCs, no significant DTH was observed 4.5 wk later (Fig. 2). However, when mice were immunized with DCs that had been pulsed with PPD and exposed to TRANCE, DTH responses were evident 4.5 and 9 wk later (Fig. 2). Taken together, these results indicate that TRANCE treatment enhances the capacity of antigen-pulsed DCs to induce both primary and secondary immune responses in vivo.
Figure 2.
Induction of T cell memory by TRANCE-treated antigen-pulsed DCs. Mice were injected in the left hind footpad with 2 × 105 DCs that were either unpulsed, TRANCE treated, PPD pulsed, or both PPD pulsed and TRANCE treated as in the legend to Fig. 1. 4.5 or 9 wk later, mice were challenged by the injection of 30 μg of PPD in the right hind footpad and PBS only in the left. Footpad swelling was assessed 24 h later. Each bar represents the increase in thickness of right versus left footpad (mean ± SD, 9–12 mice per group). *P < 0.05, compared with the group of mice injected with PPD-pulsed DCs.
TRANCE Increases the Number of DCs in Draining LNs.
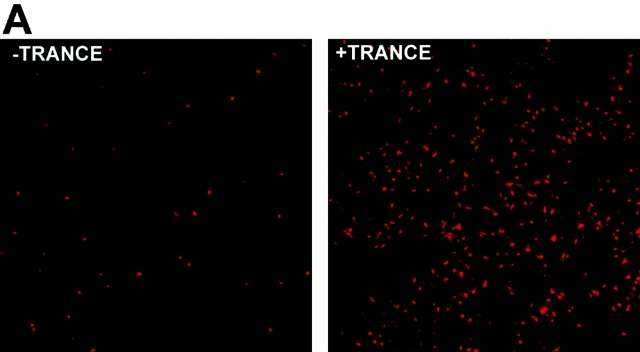
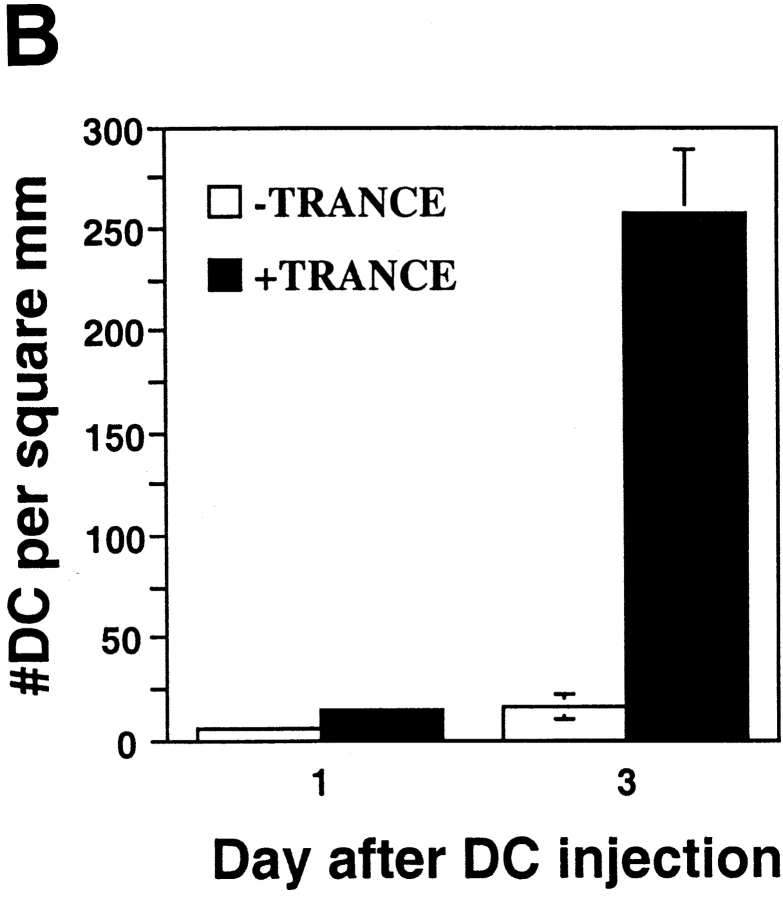
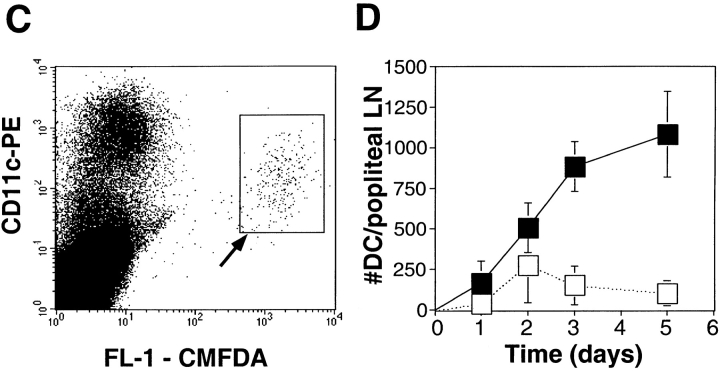
Prior results 8, confirmed repeatedly during the course of these experiments, had shown that TRANCE did not alter the expression of several cell surface molecules that contribute to the T cell stimulatory function of mature DCs, including MHC class II and costimulatory molecules such as B7-1 (CD80), B7-2 (CD86), intercellular adhesion molecule 1 (ICAM-1 [CD54]), and CD40. Therefore, we looked for other mechanisms whereby TRANCE might act to enhance the capacity of DCs as an adjuvant. As mentioned above, DCs die shortly after reaching the LN 5. Given the capacity of TRANCE to enhance DC survival in vitro (Fig. 1 A), we tested whether TRANCE treatment could enhance the longevity of DCs in the draining LNs. OVA-pulsed DCs were labeled with a fluorescent vital dye (CMTMR) before subcutaneous injection. Draining LNs were subsequently collected and analyzed by confocal microscopy as described previously 5. Whether DCs were pretreated with TRANCE or not, all of the subcutaneously injected DCs were found almost exclusively in the paracortical regions of the LN (Fig. 3 A), previously shown to be the site for antigen-specific T cell–DC interactions 5. However, when DCs were pretreated with TRANCE, there were considerably higher numbers of DCs detected in the draining LNs (Fig. 3a and Fig. b).
Figure 3.
TRANCE treatment enhances numbers of injected mature DCs in the LN draining the site of injection. OVA-pulsed mature DCs were treated with or without TRANCE, labeled with CMTMR or CMFDA, and 5 × 105 cells were injected subcutaneously in both hind footpads of anesthetized naive mice. For A and B, CMTMR-labeled DCs were injected, and draining popliteal LNs were harvested at various time points after subcutaneous injection, processed, and the sections were analyzed by confocal immunofluorescent microscopy as described (reference 5). (A) Images were taken from the paracortical region of the LNs of mice injected 3 d earlier with DCs with or without TRANCE. The optical thickness of each section is 4 μm. (B) The number of DCs was quantitated per unit area (mm2) of LN. The results represent the mean ± SD of two to three mice per group derived from a single experiment. Similar results were obtained in three other independent experiments. (C and D) CMFDA-labeled DCs were injected, and the draining popliteal LNs were harvested daily and digested with collagenase. The LN cells were counted, stained with PE-conjugated CD11c, and analyzed on a FACScan™. (C) FACS® data showing that the injected DCs are recovered as a small fraction (≤1%) of the CD11c+ subset (arrow). (D) The absolute number of injected DCs in each draining LN (CD11c+ and FL-1high) was calculated. Each point represents the mean ± SD of three mice in each group and at each time point. Similar results were obtained in three other independent experiments.
To further quantitate the increase in the number of TRANCE-treated DCs in the draining LNs, mice were injected subcutaneously with CMFDA-labeled DCs, and the draining LNs were then collected 1–5 d later. The LNs were dissociated by collagenase treatment, which has been shown to release most of the DCs from the T cell areas 17. All of the fluorescent cells were found to express CD11c (Fig. 3 C), a marker that is expressed at high levels on DCs, but not on lymphocytes and macrophages 18 19. As described previously 15, the bone marrow–derived DCs that migrated to the LNs expressed lower levels of CD11c than resident LN DCs (Fig. 3 C). Only a small number of fluorescent DCs were found in the LNs (Fig. 3 C), indicating that most of the DCs injected subcutaneously must have died rapidly in situ since it is known that most of those DCs leave the injection site 4. Nevertheless, the number of labeled DCs in draining LNs was markedly increased (up to 5–10-fold) when the DCs were treated before injection with TRANCE (Fig. 3 D). Moreover, the number of injected DCs remained at increased levels for at least 5 d, whereas few injected DCs were observed after 3 d when the DCs were not pretreated with TRANCE (Fig. 3 D).
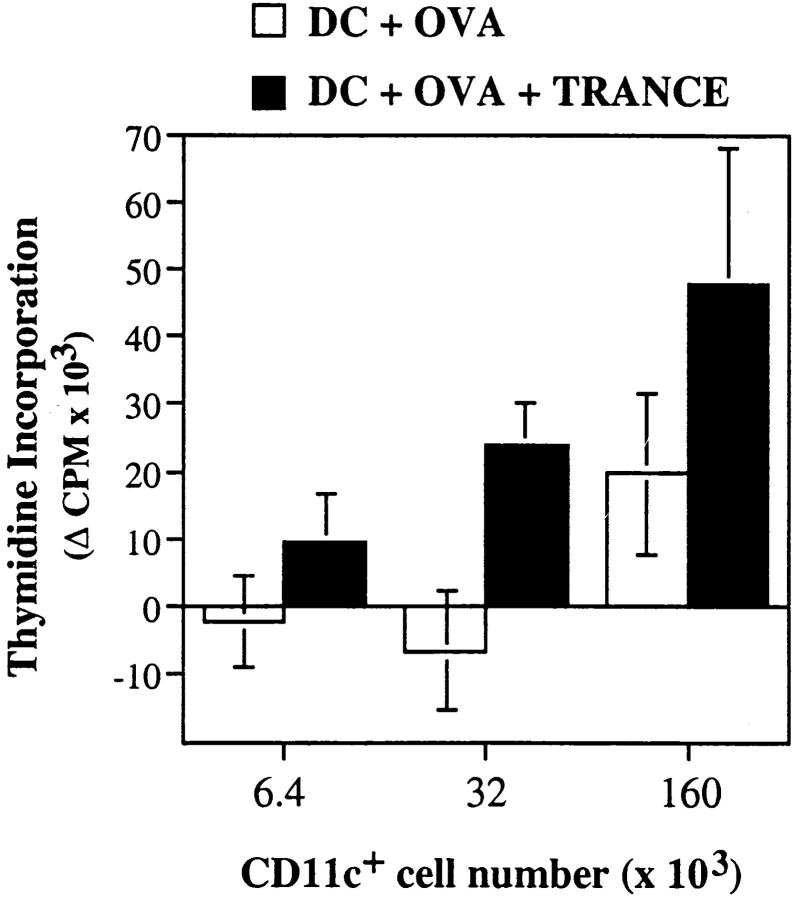
We then determined whether DCs found in draining LNs 3 d after subcutaneous injection retained their capacity to present antigen and stimulate T cells. Immature bone marrow–derived DCs were pulsed with OVA, and then treated with TRANCE as described above. 5 × 105 cells were then injected in each footpad of BALB/c mice, and draining LNs were harvested 3 d later and digested with collagenase. CD11c+ cells were then purified and used to stimulate resting CD4+ T cells from DO11-10 TCR transgenic mice 20. As shown in Fig. 4, CD11c+ cells from mice injected with TRANCE-treated, OVA-pulsed DCs induced significantly higher proliferative responses for DO11-10 CD4+ T cells than those from mice immunized with DCs pulsed with OVA only (Fig. 4): approximately three- to fivefold more CD11c+ cells from mice immunized with OVA-pulsed DCs were required to obtain a similar T cell proliferative response than from mice immunized with TRANCE-treated, OVA-pulsed DCs. Since we consistently observed a three- to fivefold difference in the number of injected DCs in the draining LNs 3 d after immunization (Fig. 3), the results suggest that the difference in the number of antigen-pulsed DCs found in the LN is responsible for differential T cell proliferation.
Figure 4.
Increased presentation of antigens by DCs from the draining LNs of mice immunized with TRANCE-treated DCs. Immature bone marrow–derived DCs were pulsed for 6 h with or without 10 μg/ml OVA. The cells were dislodged and transferred to new 100-mm culture dishes for 48 h to allow maturation. During the last 12 h, the cells were pulsed with 1 μg/ml soluble TRANCE (black bars) or with medium alone (white bars). The DCs were then washed, and 5 × 105 cells were injected in hind and front footpads of anesthetized naive mice. 3 d later, draining LNs were harvested, digested in collagenase, and DCs were purified using CD11c-MACS beads following the manufacturer's instructions (Miltenyi Biotec). Serial dilutions of purified CD11c+ DCs were mixed with 5 × 104 DO11-10 TCR transgenic resting CD4+ T cells in round-bottomed 96-well plates for 72 h. Proliferation was assessed during the last 8 h of culture. Background proliferation obtained with CD11c+ DCs prepared in a similar way from BALB/c mice immunized with TRANCE-treated DCs without OVA was subtracted.
Discussion
Mature DCs are powerful antigen-presenting cells thought to be short-lived both in vitro and in vivo 2. The short half-life of these cells could represent an important mechanism for controlling the normal immune response and for ensuring adequate space for the constant influx of fresh DCs loaded with different antigens. However, it is not known whether an increase in DC survival, and therefore in the longevity of antigen-pulsed DCs at the site of T cell priming in LNs, would affect the strength of an immune response.
In this study, we have shown that treatment of DCs with TRANCE before immunization enhances their adjuvant capacity, which results in better T cell priming in vivo. Although DCs without prior TRANCE treatment were able to prime Th cells in the draining LNs, as assessed by antigen-specific proliferation assay and cytokine production, this primary response was strongly enhanced when DCs were pretreated with TRANCE before subcutaneous injection. Moreover, a significant memory response was observed only in mice injected with TRANCE-treated DCs. How does TRANCE produce these consequences? One possibility would be that TRANCE enhances the adjuvant property of DCs via cytokine induction. We have recently shown that TRANCE induced multiple cytokines in DCs, including IL-1, IL-6, IL-12, and IL-15 11 13. Since IL-12 and IL-15 are important cytokines involved in the activation and differentiation of T cells 21 22, this effect may contribute to the increased T cell responses we observed with TRANCE-pulsed DCs. IL-12 production might also explain the substantial increase in IFN-γ priming and Th1 response 22 found in draining LNs of mice injected with TRANCE-pulsed DCs.
However, it is also possible that one of the underlying mechanisms for the enhanced T cell immune responses by TRANCE is an increase in the number of antigen-pulsed DCs that are found in the draining LNs. Similar to its effect on DC survival in vitro, TRANCE may have increased the survival of DCs at the site of injection, which would possibly increase the number of DCs migrating out of this site and into the draining LN. TRANCE may also enhance the survival of DCs that have migrated to the T cell area. Ingulli et al. 5 have shown that 24 h after subcutaneous injection, peptide-pulsed mature DCs are found in the draining LN and they interact with antigen-specific T cells. Since activated T cells express CD40L and TRANCE, which can increase the survival of DCs in situ, it is possible that the antiapoptotic molecules induced by TRANCE 8 before the subcutaneous injection may have protected antigen-presenting mature DCs against spontaneous apoptosis until they are able to fully communicate with antigen-specific T cells. In addition, TRANCE might modify the expression of those chemokine receptors, such as CCR7 23 24, that favor the entry of DCs into afferent lymphatics and homing to the T cell areas. Although several of these mechanisms may operate together, we favor the theory that the effect of TRANCE is on DC numbers as opposed to “function per cell,” because the efficacy of T cell stimulation by DCs isolated from the draining LNs is proportional to the number of surviving migrated DCs (Fig. 4).
Our results now suggest that the strength of DC–T cell interaction required for successful activation of antigen-specific T cells 25 can be influenced not only by the cytokine secretion profile of DCs, but also by their longevity and/or abundance at the site of T cell priming. Therefore, future experiments should determine whether the longevity and/or abundance of antigen-presenting DCs at the site of T cell priming are critical factors for the induction of T cell responses to antigens delivered by natural routes, e.g., viral infection or tumors.
Although this report focuses on the effects of TRANCE on DCs, CD40L can also enhance the survival and adjuvanticity of DCs (our unpublished data). This observation may explain previous reports that CD40L enhances DC function in vivo, allowing them to stimulate CD8+ T cells and induce CTL differentiation 26 27 28 29. However, while the expression of CD40L is restricted to CD4+ T cells, TRANCE is also expressed on activated CD8+ T cells, and may therefore confer the ability to enhance DC survival and function on CD8+ T cells as well 11. Since TNF family members are rapidly upregulated on T cells by TCR stimulation 11, our results suggest that activated T cells provide a powerful positive reinforcement to antigen-presenting DCs by enhancing their survival and cytokine production.
DC-mediated immunotherapy (i.e., the active immunization of humans with autologous DCs that have been pulsed with clinically significant antigens, including tumor, viral, and bacterial antigens [ 30, 31]) may be improved by the application of our findings. Initial studies in humans have shown that the subcutaneous injection of DCs pulsed ex vivo with protein antigen or peptide is an efficient way to prime and boost CD4+ and CD8+ T cells 32. However, recent reports suggest that current protocols result in very low rates of survival and/or migration of DCs in the draining LNs 33, indicating that the methodology is operating far below the adjuvant potential of DCs. We have also found that, despite a significant adjuvant effect, mature bone marrow–derived DCs migrated poorly to draining LN after subcutaneous injection. Exposure of DCs to TNF family members such as TRANCE or CD40L before injection is likely to improve the efficacy of DC-based immunotherapy by enhancing the in vivo survival and the number of antigen-presenting cells in draining LNs. This hypothesis is currently under investigation.
Acknowledgments
This study was supported in part by National Institutes of Health grants to R.M. Steinman (AI13013 and DK39672) and Y. Choi (AI44264). R. Josien was supported in part by the Association pour la Recherche contre le Cancer, and a Revson and Winston Biomedical Fellowship. B. Wong is supported by the Medical Scientist Training Program. Y. Choi is an associate investigator of the Howard Hughes Medical Institute.
Footnotes
R. Josien's present address is Service de Néphrologie-Immunologie Clinique, CHRU de Nantes, Immeuble Jean Monnet, 44035 Nantes Cedex 1, France.
Y. Choi, Laboratory of Immunology, Howard Hughes Medical Institute, The Rockefeller University, 1230 York Ave., New York, NY 10021. Phone: 212-327-7441; Fax: 212-327-7319; E-mail: choi@rockvax.rockefeller.edu
Abbreviations used in this paper: DC, dendritic cell; DTH, delayed-type hypersensitivity; PPD, purified protein derivative; TRANCE, TNF-related activation-induced cytokine.
References
- Hart D.N. Dendritic cellsunique leukocyte populations which control the primary immune response. Blood. 1997;90:3245–3287 . [PubMed] [Google Scholar]
- Banchereau J., Steinman R.M. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- Inaba K., Metlay J.P., Crowley M.T., Steinman R.M. Dendritic cells pulsed with protein antigens in vitro can prime antigen-specific, MHC-restricted T cells in situ. J. Exp. Med. 1990;172:631–640. doi: 10.1084/jem.172.2.631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kupiec-Weglinski J.W., Austyn J.M., Morris P.J. Migration patterns of dendritic cells in the mousetraffic from the blood, and T cell–dependent and –independent entry to lymphoid tissues. J. Exp. Med. 1988;167:632–645. doi: 10.1084/jem.167.2.632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingulli E., Mondino A., Khoruts A., Jenkins M.K. In vivo detection of dendritic cell antigen presentation to CD4+ T cells. J. Exp. Med. 1997;185:2133–2141. doi: 10.1084/jem.185.12.2133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fossum S. Lymph-borne dendritic leukocytes do not recirculate, but enter the lymph node paracortex to become interdigitating cells. Scand. J. Immunol. 1988;27:97–105. doi: 10.1111/j.1365-3083.1988.tb02326.x. [DOI] [PubMed] [Google Scholar]
- Pugh C.W., MacPherson G.G., Steer H.W. Characterization of nonlymphoid cells derived from rat peripheral lymph. J. Exp. Med. 1983;157:1758–1779. doi: 10.1084/jem.157.6.1758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong B.R., Josien R., Lee S.Y., Sauter B., Li H.-L., Steinman R.M., Choi Y. TRANCE (tumor necrosis factor [TNF]-related activation-induced cytokine), a new TNF family member predominantly expressed in T cells, is a dendritic cell–specific survival factor. J. Exp. Med. 1997;186:2075–2080. doi: 10.1084/jem.186.12.2075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong B.R., Rho J., Arron J.R., Robinson E., Orlinick J., Chao M., Kalachikov S., Cayani E., Bartlett F.S., III, Frankel W.N. TRANCE is a novel ligand of the tumor necrosis factor receptor family that activates c-Jun N-terminal kinase in T cells. J. Biol. Chem. 1997;272:25190–25194. doi: 10.1074/jbc.272.40.25190. [DOI] [PubMed] [Google Scholar]
- Wong B.R., Josien R., Choi Y. TRANCE is a TNF family member that regulates dendritic cell and osteoclast function. J. Leukoc. Biol. 1999;65:715–724. doi: 10.1002/jlb.65.6.715. [DOI] [PubMed] [Google Scholar]
- Josien R., Wong B.R., Li H.L., Steinman R.M., Choi Y. TRANCE, a TNF family member, is differentially expressed on T cell subsets and induces cytokine production in dendritic cells. J. Immunol. 1999;162:2562–2568. [PubMed] [Google Scholar]
- Anderson D.M., Maraskovsky E., Billingsley W.L., Dougall W.C., Tometsko M.E., Roux E.R., Teepe M.C., DuBose R.F., Cosman D., Galibert L.A. A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature. 1997;390:175–179. doi: 10.1038/36593. [DOI] [PubMed] [Google Scholar]
- Bachmann M.F., Wong B.R., Josien R., Steinman R.M., Oxenius A., Choi Y. TRANCE, a tumor necrosis factor family member critical for CD40 ligand–independent T helper cell activation. J. Exp. Med. 1999;189:1025–1031. doi: 10.1084/jem.189.7.1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inaba K., Inaba M., Naito M., Steinman R.M. Dendritic cell progenitors phagocytose particulates, including bacillus Calmette-Guérin organisms, and sensitize mice to mycobacterial antigens in vivo. J. Exp. Med. 1993;178:479–488. doi: 10.1084/jem.178.2.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inaba K., Inaba M., Romani N., Aya H., Deguchi M., Ikehara S., Muramatsu S., Steinman R.M. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J. Exp. Med. 1992;176:1693–1702. doi: 10.1084/jem.176.6.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierre P., Turley S.J., Gatti E., Hull M., Meltzer J., Mirza A., Inaba K., Steinman R.M., Mellman I. Developmental regulation of MHC class II transport in mouse dendritic cells. Nature. 1997;388:787–792. doi: 10.1038/42039. [DOI] [PubMed] [Google Scholar]
- Inaba K., Pack M., Inaba M., Sakuta H., Isdell F., Steinman R.M. High levels of a major histocompatibility complex II–self peptide complex on dendritic cells from the T cell areas of lymph nodes. J. Exp. Med. 1997;186:665–672. doi: 10.1084/jem.186.5.665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metlay J.P., Witmer-Pack M.D., Agger R., Crowley M.T., Lawless D., Steinman R.M. The distinct leukocyte integrins of mouse spleen dendritic cells as identified with new hamster monoclonal antibodies. J. Exp. Med. 1980;171:1753–1772. doi: 10.1084/jem.171.5.1753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witmer-Pack M.D., Crowley M.T., Inaba K., Steinman R.M. Macrophages, but not dendritic cells, accumulate colloidal carbon following administration in situ . J. Cell Sci. 1993;105:965–973. doi: 10.1242/jcs.105.4.965. [DOI] [PubMed] [Google Scholar]
- Murphy K.M., Heimberger A.B., Loh D.Y. Induction by antigen of intrathymic apoptosis of CD4+CD8+TCRlo thymocytes in vivo . Science. 1990;250:1720–1723. doi: 10.1126/science.2125367. [DOI] [PubMed] [Google Scholar]
- Zhang X., Sun S., Hwang I., Tough D.F., Sprent J. Potent and selective stimulation of memory-phenotype CD8+ T cells in vivo by IL-15. Immunity. 1998;8:591–599. doi: 10.1016/s1074-7613(00)80564-6. [DOI] [PubMed] [Google Scholar]
- Macatonia S.E., Hosken N.A., Litton M., Vieira P., Hsieh C.S., Culpepper J.A., Wysocka M., Trinchieri G., Murphy K.M., O'Garra A. Dendritic cells produce IL-12 and direct the development of Th1 cells from naive CD4+ T cells. J. Immunol. 1995;154:5071–5079. [PubMed] [Google Scholar]
- Yanagihara S., Komura E., Nagafune J., Watarai H., Yamaguchi Y. EBI1/CCR7 is a new member of dendritic cell chemokine receptor that is up-regulated upon maturation. J. Immunol. 1998;161:3096–3102. [PubMed] [Google Scholar]
- Sallusto F., Schaerli P., Loetscher P., Schaniel C., Lenig D., Mackay C.R., Qin S., Lanzavecchia A. Rapid and coordinated switch in chemokine receptor expression during dendritic cell maturation. Eur. J. Immunol. 1998;28:2760–2769. doi: 10.1002/(SICI)1521-4141(199809)28:09<2760::AID-IMMU2760>3.0.CO;2-N. [DOI] [PubMed] [Google Scholar]
- Lanzavecchia A., Lezzi G., Viola A. From TCR engagement to T cell activationa kinetic view of T cell behavior. Cell. 1999;96:1–4. doi: 10.1016/s0092-8674(00)80952-6. [DOI] [PubMed] [Google Scholar]
- Mackey M.F., Gunn J.R., Maliszewsky C., Kikutani H., Noelle R.J., Barth R.J., Jr. Dendritic cells require maturation via CD40 to generate protective antitumor immunity. J. Immunol. 1998;161:2094–2098. [PubMed] [Google Scholar]
- Ridge J.P., Di Rosa F., Matzinger P. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer. Nature. 1998;393:474–478. doi: 10.1038/30989. [DOI] [PubMed] [Google Scholar]
- Schoenberger S.P., Toes R.E., van der Voort E.I., Offringa R., Melief C.J. T-cell help for cytotoxic T lymphocytes is mediated by CD40-CD40L interactions. Nature. 1998;393:480–483. doi: 10.1038/31002. [DOI] [PubMed] [Google Scholar]
- Bennett S.R., Carbone F.R., Karamalis F., Flavell R.A., Miller J.F., Heath W.R. Help for cytotoxic-T-cell responses is mediated by CD40 signalling. Nature. 1998;393:478–480. doi: 10.1038/30996. [DOI] [PubMed] [Google Scholar]
- Fernandez N., Duffour M.T., Perricaudet M., Lotze M.T., Tursz T., Zitvogel L. Active specific T-cell–based immunotherapy for cancernucleic acids, peptides, whole native proteins, recombinant viruses, with dendritic cell adjuvants or whole tumor cell-based vaccines. Principles and future prospects. Cytokines Cell. Mol. Ther. 1998;4:53–65. [PubMed] [Google Scholar]
- Schuler G., Steinman R.M. Dendritic cells as adjuvants for immune-mediated resistance to tumors. J. Exp. Med. 1997;186:1183–1187. doi: 10.1084/jem.186.8.1183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhodapkar M.V., Steinman R.M., Sapp M., Desai H., Fossella C., Krasovsky J., Donahoe S.M., Dunbar P.R., Cerundolo V., Nixon D.F., Bhardwaj N. Rapid generation of broad T-cell immunity in humans after a single injection of mature dendritic cells. J. Clin. Invest. 1999;104:173–180. doi: 10.1172/JCI6909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morse M.A., Coleman R.E., Akabani G., Niehaus N., Coleman D., Lyerly H.K. Migration of human dendritic cells after injection in patients with metastatic malignancies. Cancer Res. 1999;59:56–58. [PubMed] [Google Scholar]