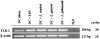Lactobacilli activate human dendritic cells that skew T cells toward T helper 1 polarization - PubMed (original) (raw)
Lactobacilli activate human dendritic cells that skew T cells toward T helper 1 polarization
Mansour Mohamadzadeh et al. Proc Natl Acad Sci U S A. 2005.
Abstract
Professional antigen-presenting dendritic cells (DCs) are critical in regulating T cell immune responses at both systemic and mucosal sites. Many Lactobacillus species are normal members of the human gut microflora and most are regarded as safe when administered as probiotics. Because DCs can naturally or therapeutically encounter lactobacilli, we investigated the effects of several well defined strains, representing three species of Lactobacillus on human myeloid DCs (MDCs) and found that they modulated the phenotype and functions of human MDCs. Lactobacillus-exposed MDCs up-regulated HLA-DR, CD83, CD40, CD80, and CD86 and secreted high levels of IL-12 and IL-18, but not IL-10. IL-12 was sustained in MDCs exposed to all three Lactobacillus species in the presence of LPS from Escherichia coli, whereas LPS-induced IL-10 was greatly inhibited. MDCs activated with lactobacilli clearly skewed CD4(+) and CD8(+) T cells to T helper 1 and Tc1 polarization, as evidenced by secretion of IFN-gamma, but not IL-4 or IL-13. These results emphasize a potentially important role for lactobacilli in modulating immunological functions of DCs and suggest that certain strains could be particularly advantageous as vaccine adjuvants, by promoting DCs to regulate T cell responses toward T helper 1 and Tc1 pathways.
Figures
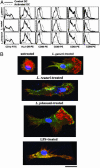
Fig. 1.
FACS and confocal analysis of MDCs treated with different Lactobacillus species. (A) MDCs were treated with Lactobacillus cells (1,000 colony-forming units per DC) for 3 d. Treated (dotted line) and untreated (solid line) MDCs were harvested, washed extensively with PBS, stained with specific Abs for 1 h at 4°C, and analyzed by FACS. Experiments were repeated at least three times with similar results. (B) Treated or untreated MDCs with Lactobacillus species or E. coli LPS were stained with Ab to HLA-DR (green), a fluorescent marker of actin polymer (red), and Hoechst DNA stain (blue). (Scale bar, 20 μm.)
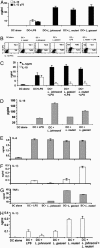
Fig. 2.
Induction of bioactive IL-12, IL-18, and proinflammatory cytokines in MDCs treated with Lactobacillus cells. MDCs were treated with Lactobacillus species, E. coli LPS, or no supplement for 3 d at 37°C. (A) Supernatants of MDCs treated with Lactobacillus species, E. coli LPS, or no supplement were harvested after 72 h and analyzed for IL-10 and IL-12 by ELISA. Experiments were performed at least three times with similar results. (B) MDCs were treated with Lactobacillus species, E. coli LPS, L. reuteri and E. coli LPS, or no supplement for 48 h. MDCs were harvested and treated with Golgi inhibitor for 4 h. Cells were stained with anti-IL-10 allophycocyanin or anti-IL-12 FITC for 1 h on ice. Cells were washed and analyzed by flow cytometry. (C) DCs were treated with LPS alone or in combination with Lactobacillus species for 72 h. Cytokines were then analyzed by ELISA. (D) Supernatants of MDCs treated with Lactobacillus species, E. coli LPS, or no supplement were harvested after 72 h and analyzed for IL-18 by ELISA. Experiments were performed at least three times with similar results. (E_–_H) MDCs supernatants were harvested and analyzed for proinflammatory cytokines by ELISA. Results are representative of three independent experiments.
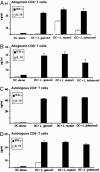
Fig. 3.
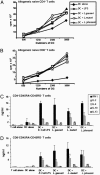
Induction of T cell proliferation by MDCs treated with live Lactobacillus species. MDCs were treated with live Lactobacillus species, E. coli LPS (100 ng/ml), or no supplement for 72 h at 37°C. Treated and untreated MDCs were harvested, washed extensively with PBS, and at graded doses cocultured with purified allogeneic or autologous CD4+ or CD8+ T cells (105 cells per well in a 96-well plate) for 4 d at 37°C. [3H]Thymidine incorporation was measured in triplicate by using a β-counter. Results are representative of three independent experiments.
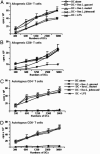
Fig. 4.
MDCs treated with Lactobacillus species skew T cells toward Th1 immune responses. MDCs were treated with live Lactobacillus species, E. coli LPS (100 ng/ml), or no supplement for 3 d. MDCs were harvested and cocultured with allogeneic or autologous CD4+ or CD8+ T cells for 4 d at 37°C. Supernatants of T cells cocultured with MDCs treated with Lactobacillus species, E. coli LPS, or no supplement were collected and assayed for cytokines by ELISA. All experiments were repeated at least three times.
Fig. 5.
Activation of naive T cells by _Lactobacillus_-activated MDCs. Sorted naive CD4+ and CD8+ T cells (50 × 103 cells per well in a 96-well plate) were cocultured for 4 d with allogeneic or autologous MDCs, which were treated with live Lactobacillus species or with E. coli LPS. Cells were pulsed for the last 16 h with 0.5 μCi of [3H]thymidine per well. [3H]Thymidine incorporation was measured by using a β-counter. Cytokines released in the supernatants of the cocultures were assayed by ELISA.
Fig. 6.
Total RNA was isolated from MDC activated with Lactobacillus species, E. coli LPS, or untreated MDC. RT-PCR for TLR-2 and β-actin was performed. Data shown are representative of three experiments.
Similar articles
- Specific Lactobacillus species differentially activate Toll-like receptors and downstream signals in dendritic cells.
Mohamadzadeh M, Klaenhammer TR. Mohamadzadeh M, et al. Expert Rev Vaccines. 2008 Oct;7(8):1155-64. doi: 10.1586/14760584.7.8.1155. Expert Rev Vaccines. 2008. PMID: 18844590 - Polarization of naive T cells into Th1 or Th2 by distinct cytokine-driven murine dendritic cell populations: implications for immunotherapy.
Feili-Hariri M, Falkner DH, Morel PA. Feili-Hariri M, et al. J Leukoc Biol. 2005 Sep;78(3):656-64. doi: 10.1189/jlb.1104631. Epub 2005 Jun 16. J Leukoc Biol. 2005. PMID: 15961574 - Heat-killed cells of lactobacilli skew the immune response toward T helper 1 polarization in mouse splenocytes and dendritic cell-treated T cells.
Chuang L, Wu KG, Pai C, Hsieh PS, Tsai JJ, Yen JH, Lin MY. Chuang L, et al. J Agric Food Chem. 2007 Dec 26;55(26):11080-6. doi: 10.1021/jf071786o. Epub 2007 Nov 27. J Agric Food Chem. 2007. PMID: 18038979 - Diverse functional activity of CD83+ monocyte-derived dendritic cells and the implications for cancer vaccines.
Czerniecki BJ, Cohen PA, Faries M, Xu S, Roros JG, Bedrosian I. Czerniecki BJ, et al. Crit Rev Immunol. 2001;21(1-3):157-78. Crit Rev Immunol. 2001. PMID: 11642602 Review. - Induction of protective immunity against microbial challenge by targeting antigens expressed by probiotic bacteria to mucosal dendritic cells.
Mohamadzadeh M. Mohamadzadeh M. Curr HIV Res. 2010 Jun;8(4):323-9. doi: 10.2174/157016210791208668. Curr HIV Res. 2010. PMID: 20353394 Review.
Cited by
- Probiotic modulation of dendritic cells co-cultured with intestinal epithelial cells.
Kim JY, Park MS, Ji GE. Kim JY, et al. World J Gastroenterol. 2012 Mar 28;18(12):1308-18. doi: 10.3748/wjg.v18.i12.1308. World J Gastroenterol. 2012. PMID: 22493544 Free PMC article. - Differential interleukin-10 (IL-10) and IL-23 production by human blood monocytes and dendritic cells in response to commensal enteric bacteria.
Manuzak J, Dillon S, Wilson C. Manuzak J, et al. Clin Vaccine Immunol. 2012 Aug;19(8):1207-17. doi: 10.1128/CVI.00282-12. Epub 2012 Jun 13. Clin Vaccine Immunol. 2012. PMID: 22695160 Free PMC article. - Immunological evaluation of Lactobacillus casei Zhang: a newly isolated strain from koumiss in Inner Mongolia, China.
Ya T, Zhang Q, Chu F, Merritt J, Bilige M, Sun T, Du R, Zhang H. Ya T, et al. BMC Immunol. 2008 Nov 19;9:68. doi: 10.1186/1471-2172-9-68. BMC Immunol. 2008. PMID: 19019236 Free PMC article. - Role of natural killer and dendritic cell crosstalk in immunomodulation by commensal bacteria probiotics.
Rizzello V, Bonaccorsi I, Dongarrà ML, Fink LN, Ferlazzo G. Rizzello V, et al. J Biomed Biotechnol. 2011;2011:473097. doi: 10.1155/2011/473097. Epub 2011 May 3. J Biomed Biotechnol. 2011. PMID: 21660136 Free PMC article. Review. - Regulation of induced colonic inflammation by Lactobacillus acidophilus deficient in lipoteichoic acid.
Mohamadzadeh M, Pfeiler EA, Brown JB, Zadeh M, Gramarossa M, Managlia E, Bere P, Sarraj B, Khan MW, Pakanati KC, Ansari MJ, O'Flaherty S, Barrett T, Klaenhammer TR. Mohamadzadeh M, et al. Proc Natl Acad Sci U S A. 2011 Mar 15;108 Suppl 1(Suppl 1):4623-30. doi: 10.1073/pnas.1005066107. Epub 2011 Jan 31. Proc Natl Acad Sci U S A. 2011. PMID: 21282652 Free PMC article.
References
- Macpherson, A. J. & Harris, N. L. (2004) Nat. Rev. Immunol. 4, 478–485. - PubMed
- Klaenhammer, T. R. & Kullen, M. J. (1999) Int. J. Food Microbiol. 50, 45–57. - PubMed
- Erickson, K. L. & Hubbard, N. E. (2000) J. Nutr. 130, 403S–409S. - PubMed
- Ahrne, S., Nobaek, S., Jeppsson, B., Adlerberth, I., Wold, A. E. & Molin, G. (1998) J. Appl. Microbiol. 85, 88–94. - PubMed
- Guarner, F. & Schaafsma, G. J. (1998) Int. J. Food Microbiol. 39, 237–238. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous