The calmodulin pathway and evolution of elongated beak morphology in Darwin's finches (original) (raw)
- Letter
- Published: 03 August 2006
- Winston P. Kuo1,2,3 nAff6,
- Christine Hartmann4,
- B. Rosemary Grant5,
- Peter R. Grant5 &
- …
- Clifford J. Tabin1
Nature volume 442, pages 563–567 (2006)Cite this article
- 15k Accesses
- 592 Citations
- 43 Altmetric
- Metrics details
Abstract
A classic textbook example of adaptive radiation under natural selection is the evolution of 14 closely related species of Darwin's finches (Fringillidae, Passeriformes), whose primary diversity lies in the size and shape of their beaks1,2,3,4,5,6. Thus, ground finches have deep and wide beaks, cactus finches have long and pointed beaks (low depth and narrower width), and warbler finches have slender and pointed beaks, reflecting differences in their respective diets6. Previous work has shown that even small differences in any of the three major dimensions (depth, width and length) of the beak have major consequences for the overall fitness of the birds3,4,5,6,7. Recently we used a candidate gene approach to explain one pathway involved in Darwin's finch beak morphogenesis8. However, this type of analysis is limited to molecules with a known association with craniofacial and/or skeletogenic development. Here we use a less constrained, complementary DNA microarray analysis of the transcripts expressed in the beak primordia to find previously unknown genes and pathways whose expression correlates with specific beak morphologies. We show that calmodulin (CaM), a molecule involved in mediating Ca2+ signalling, is expressed at higher levels in the long and pointed beaks of cactus finches than in more robust beak types of other species. We validated this observation with in situ hybridizations. When this upregulation of the CaM-dependent pathway is artificially replicated in the chick frontonasal prominence, it causes an elongation of the upper beak, recapitulating the beak morphology of the cactus finches. Our results indicate that local upregulation of the CaM-dependent pathway is likely to have been a component of the evolution of Darwin's finch species with elongated beak morphology and provide a mechanistic explanation for the independence of beak evolution along different axes. More generally, our results implicate the CaM-dependent pathway in the developmental regulation of craniofacial skeletal structures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Microarray analysis in different finch species.
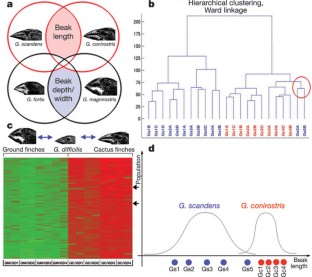
The alternative text for this image may have been generated using AI.
Figure 2: Comparative analysis of CaM expression in finches.
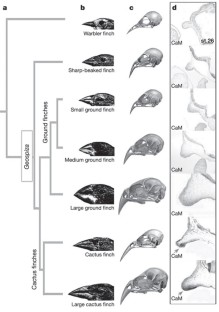
The alternative text for this image may have been generated using AI.
Figure 3: Functional analysis of CaM-dependent pathway in beak development.
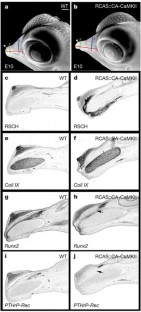
The alternative text for this image may have been generated using AI.
Figure 4: BMP- and CaM-dependent signalling regulates growth along different axes, facilitating the evolution of distinct beak morphologies in Darwin's finches.
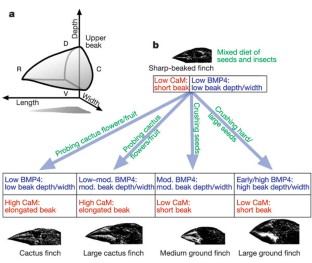
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Darwin, C. R. Journal of Researches into the geology and Natural History of the various countries visited during the voyage of H.M.S. Beagle, under the command of Captain FitzRoy, R.N. 2nd edn (John Murray, London, 1845)
Google Scholar - Lack, D. Darwin's Finches (Cambridge Univ. Press, Cambridge, 1947)
Google Scholar - Grant, P. R. The Ecology and Evolution of Darwin's Finches (Princeton Univ. Press, Princeton, New Jersey, 1999)
Google Scholar - Futuyma, D. J. Evolutionary Biology 3rd edn (Sinauer Associates, Sunderland, Massachusetts, 1998)
Google Scholar - Freeman, S. & Herron, J. C. Evolutionary Analysis 3rd edn (Prentice Hall, Englewood Cliffs, New Jersey, 2003)
Google Scholar - Bowman, R. I. Morphological differentiation and adaptation in the Galapagos finches. Univ. Calif. Publ. Zool. 58, 1–302 (1961)
Google Scholar - Grant, P. R. & Grant, B. R. Unpredictable evolution in a 30-year study of Darwin's finches. Science 296, 707–711 (2002)
Article ADS CAS Google Scholar - Abzhanov, A., Protas, M., Grant, R. B., Grant, P. R. & Tabin, C. J. Bmp4 and morphological variation of beaks in Darwin's finches. Science 305, 1462–1465 (2004)
Article ADS CAS Google Scholar - O'Day, D. H. CaMBOT: profiling and characterizing calmodulin-binding proteins. Cell. Signal. 15, 347–354 (2003)
Article CAS Google Scholar - Burke, A. C., Nelson, C. E., Morgan, B. A. & Tabin, C. Hox genes and the evolution of vertebrate axial morphology. Development 121, 333–346 (1996)
Google Scholar - Averof, M. & Patel, N. H. Crustacean appendage evolution associated with changes in Hox gene expression. Nature 388, 682–686 (1997)
Article ADS CAS Google Scholar - Abzhanov, A. & Kaufman, T. C. Novel regulation of the homeotic gene Scr associated with a crustacean leg-to-maxilliped appendage transformation. Development 126, 1121–1128 (1999)
CAS PubMed Google Scholar - Abzhanov, A. & Kaufman, T. C. Crustacean (malacostracan) Hox genes and the evolution of the arthropod trunk. Development 127, 2239–2249 (2000)
CAS PubMed Google Scholar - Shapiro, M. D. et al. Genetic and developmental basis of evolutionary pelvic reduction in threespine sticklebacks. Nature 428, 717–723 (2004)
Article ADS CAS Google Scholar - Kimmel, C. B. et al. Evolution and development of facial bone morphology in threespine sticklebacks. Proc. Natl. Acad. Sci. USA 102, 5791–5796 (2005)
Article ADS CAS Google Scholar - Grant, P. R. Inheritance of size and shape in a population of Darwin's finches. Proc. R. Soc. Lond. B 212, 403–432 (1983)
ADS Google Scholar - Price, T. D. & Grant, P. R. The evolution of ontogeny in Darwin's Finches: a quantitative genetics approach. Am. Nat. 125, 169–188 (1985)
Article Google Scholar - Grant, B. R. & Grant, P. R. Evolutionary Dynamics of a Natural Population. The Large Cactus Finch of the Galápagos (Univ. Chicago Press, Chicago, Illinois, 1989)
Google Scholar - Grant, P. R. & Grant, B. R. Phenotypic and genetic effects of hybridization in Darwin's finches. Evolution Int. J. Org. Evolution 48, 297–316 (1994)
Article Google Scholar - Kirschner, M. W. & Gerhart, J. C. The Plausibility of Life: Resolving Darwin's Dilemma (Yale Univ. Press, New Haven, Connecticut, 2005)
Google Scholar - Hamburger, V. & Hamilton, H. L. A series of normal stages in the development of the chick embryo. J. Morphol. 88, 49–92 (1951)
Article CAS Google Scholar - Dudoit, S., Yang, H., Callow, M. J. & Speed, T. P. Statistical methods for identifying genes with differential expression in replicated cDNA experiments. Statist. Sin. 12, 111–139 (2002)
MATH Google Scholar - Petren, K., Grant, B. R. & Grant, P. R. A phylogeny of Darwin's finches based on microsatellite DNA length variation. Proc. R. Soc. Lond. B 266, 321–329 (1999)
Article CAS Google Scholar
Acknowledgements
We thank all field assistants and participants of the field collecting trips—M. Protas, J. Chavez, G. Castaneda, O. Perez, F. Brown, A. Aitkhozhina, M. Gavilanes, M. Paez, K. Petren, J. Podos and S. Kleindorfer—for their advice with species identification and other advice; Charles Darwin Research Station on Santa Cruz Island and The Galápagos National Park for permits and logistical support; and M. Kirschner for discussions that led to the inception of this project. A.A. was supported by the Cancer Research Fund of a Damon Runyon–Walter Winchell Foundation Fellowship. This project was funded by a program project grant from the NIH to C.J.T. Author Contributions A.A. performed embryonic material collection, microarray probe preparation, microarray hybridizations and scanning, sectioning of material, in situ hybridizations and CaMKII functional analysis in chicken embryos. W.P.K. conducted all relevant bioinformatics analyses. C.H. constructed the RCAS virus carrying the constitutively active version of CaMKII. B.R.G. and P.R.G. provided logistics and secured permits for the fieldwork on the Galápagos Islands. C.J.T. conceived and supervised the project. A.A., B.R.G., P.R.G. and C.J.T. co-wrote the manuscript. All authors discussed the results and commented on the manuscript.
Author information
Author notes
- Arhat Abzhanov & Winston P. Kuo
Present address: Department of Developmental Biology, Harvard School of Dental Medicine, Boston, Massachusetts, 02115, USA
Authors and Affiliations
- Department of Genetics, Harvard Medical School, Massachusetts, 02115, Boston, USA
Arhat Abzhanov, Winston P. Kuo & Clifford J. Tabin - Decision Systems Group, Brigham and Women's Hospital, Harvard Medical School, 310 Thorn Building, 75 Francis Street, Massachusetts, 02115, Boston, USA
Winston P. Kuo - Department of Oral Medicine, Infection and Immunity, Harvard School of Dental Medicine, 188 Longwood Avenue, Massachusetts, 02115, Boston, USA
Winston P. Kuo - Institute of Molecular Pathology, Dr Bohr-Gasse 7, A-1030, Vienna, Austria
Christine Hartmann - Department of Ecology and Evolutionary Biology, Princeton University, New Jersey, 08544, Princeton, USA
B. Rosemary Grant & Peter R. Grant
Authors
- Arhat Abzhanov
- Winston P. Kuo
- Christine Hartmann
- B. Rosemary Grant
- Peter R. Grant
- Clifford J. Tabin
Corresponding author
Correspondence toClifford J. Tabin.
Ethics declarations
Competing interests
The sequence of Darwin's finch CaM has been deposited at GenBank under accession number DQ386479, and the microarray data have been filed with ArrayExpress under the accession number E-MEXP-702. Reprints and permission information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Abzhanov, A., Kuo, W., Hartmann, C. et al. The calmodulin pathway and evolution of elongated beak morphology in Darwin's finches.Nature 442, 563–567 (2006). https://doi.org/10.1038/nature04843
- Received: 15 February 2006
- Accepted: 25 April 2006
- Issue date: 03 August 2006
- DOI: https://doi.org/10.1038/nature04843
This article is cited by
Editorial Summary
The long and the short of it
A few years ago the depth and thickness of beaks of various species of Darwin's finches, the birds forever linked in the textbooks to adaptive radiation by natural selection, were found to correlate with expression of Bmp4 (bone morphogenetic protein 4). At that time a candidate screen did not yield any genes that could have been responsible for the evolution of different beak lengths. Now DNA microarray analysis of transcripts in the beak primordia of different Darwin's finch embryos shows that levels of calmodulin, a molecule involved in Ca2+ signalling, are correlated with beak length. With these two pathways modulating different dimensions of beak growth, a mechanism capable of producing a wide range of beak structures under different selective conditions can be envisaged.