Local migration promotes competitive restraint in a host–pathogen 'tragedy of the commons' (original) (raw)
- Letter
- Published: 06 July 2006
Nature volume 442, pages 75–78 (2006)Cite this article
- 5322 Accesses
- 369 Citations
- 13 Altmetric
- Metrics details
Abstract
Fragmented populations possess an intriguing duplicity: even if subpopulations are reliably extinction-prone, asynchrony in local extinctions and recolonizations makes global persistence possible1,2,3,4,5,6,7,8. Migration is a double-edged sword in such cases: too little migration prevents recolonization of extinct patches, whereas too much synchronizes subpopulations, raising the likelihood of global extinction. Both edges of this proverbial sword have been explored by manipulating the rate of migration within experimental populations1,3,4,5,6,8. However, few experiments have examined how the evolutionary ecology of fragmented populations depends on the pattern of migration5. Here, we show that the migration pattern affects both coexistence and evolution within a community of bacterial hosts (Escherichia coli) and viral pathogens (T4 coliphage) distributed across a large network of subpopulations. In particular, different patterns of migration select for distinct pathogen strategies, which we term 'rapacious' and 'prudent'. These strategies define a 'tragedy of the commons'9: rapacious phage displace prudent variants for shared host resources, but prudent phage are more productive when alone. We find that prudent phage dominate when migration is spatially restricted, while rapacious phage evolve under unrestricted migration. Thus, migration pattern alone can determine whether a de novo tragedy of the commons is resolved in favour of restraint.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Stochastic cellular automata predictions.
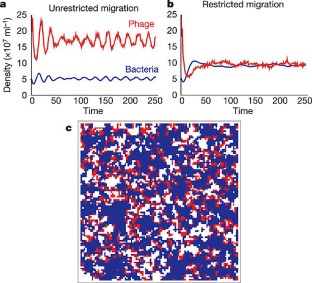
The alternative text for this image may have been generated using AI.
Figure 2: Ecological and evolutionary results for experimental metapopulations.
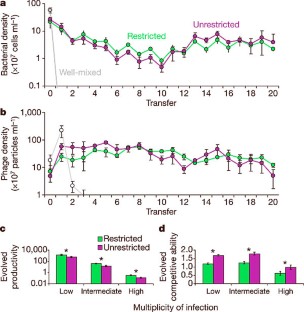
The alternative text for this image may have been generated using AI.
Figure 3: Evolutionary stochastic cellular automata.
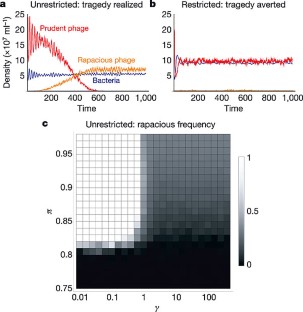
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Bonsall, M. B., French, D. R. & Hassell, M. P. Metapopulation structures affect persistence of predator–prey interactions. J. Anim. Ecol. 71, 1075–1084 (2002)
Article Google Scholar - Briggs, C. J. & Hoopes, M. F. Stabilizing effects in spatial parasitoid–host and predator–prey models: a review. Theor. Popul. Biol. 65, 299–315 (2004)
Article PubMed Google Scholar - Ellner, S. P. et al. Habitat structure and population persistence in an experimental community. Nature 412, 538–543 (2001)
Article ADS CAS PubMed Google Scholar - Holyoak, M. & Lawler, S. P. Persistence of an extinction-prone predator–prey interaction through metapopulation dynamics. Ecology 77, 1867–1879 (1996)
Article Google Scholar - Holyoak, M. Habitat patch arrangement and metapopulation persistence of predators and prey. Am. Nat. 156, 378–389 (2000)
Article Google Scholar - Huffaker, C. B. Experimental studies on predation: dispersion factors and predator–prey oscillations. Hilgardia 27, 343–383 (1958)
Article Google Scholar - Johst, K. & Schops, K. Persistence and conservation of a consumer-resource metapopulation with local overexploitation of resources. Biol. Conserv. 109, 57–65 (2003)
Article Google Scholar - Thrall, P. H., Godfree, R. & Burdon, J. J. Influence of spatial structure on pathogen colonization and extinction: a test using an experimental metapopulation. Plant Pathol. 52, 350–361 (2003)
Article Google Scholar - Hardin, G. The tragedy of the commons. Science 162, 1243–1248 (1968)
Article ADS CAS PubMed Google Scholar - Holyoak, M., Leibold, M. A. & Holt, R. D. Metacommunities: Spatial Dynamics and Ecological Communities (Univ. Chicago Press, Chicago, 2005)
Google Scholar - Antonovics, J. in Ecology, Genetics, and Evolution of Metapopulations (eds Hanski, I. & Gaggiotti, O. E.) 471–488 (Elsevier, Oxford, 2004)
Book Google Scholar - Grenfell, B., Bjørnstad, O. N. & Kappey, J. Travelling waves and spatial hierarchies in measles epidemics. Nature 414, 716–723 (2001)
Article ADS CAS PubMed Google Scholar - Keeling, M., Bjørnstad, O. N. & Grenfell, B. in Ecology, Genetics, and Evolution of Metapopulations (eds Hanski, I. & Gaggiotti, O. E.) 415–445 (Elsevier, Oxford, 2004)
Book Google Scholar - Abedon, S. T., Hyman, P. & Thomas, C. Experimental examination of bacteriophage latent-period evolution as a response to bacterial availability. Appl. Environ. Microbiol. 69, 7499–7506 (2003)
Article CAS PubMed PubMed Central Google Scholar - Bohannan, B. J. M. & Lenski, R. E. Linking genetic change to community evolution: insights from studies of bacteria and bacteriophage. Ecol. Lett. 3, 362–377 (2000)
Article Google Scholar - Lenski, R. E. & Levin, B. R. Constraints on the coevolution of bacteria and virulent phage: a model, some experiments, and predictions for natural communities. Am. Nat. 125, 585–602 (1985)
Article Google Scholar - Paddison, P. et al. The roles of the bacteriophage T4 r genes in lysis inhibition and fine-structure genetics: A new perspective. Genetics 148, 1539–1550 (1998)
CAS PubMed PubMed Central Google Scholar - Keeling, M. Evolutionary trade-offs at two time-scales: competition versus persistence. Proc. R. Soc. Lond. B 267, 385–391 (2000)
Article CAS Google Scholar - Kreft, J. U. Biofilms promote altruism. Microbiology 150, 2751–2760 (2004)
Article CAS PubMed Google Scholar - Bergstrom, C. T., McElhany, P. & Real, L. A. Transmission bottlenecks as determinants of virulence in rapidly evolving pathogens. Proc. Natl Acad. Sci. USA 96, 5095–5100 (1999)
Article ADS CAS PubMed PubMed Central Google Scholar - Boots, M. & Sasaki, A. 'Small worlds' and the evolution of virulence: infection occurs locally and at a distance. Proc. R. Soc. Lond. B 266, 1933–1938 (1999)
Article CAS Google Scholar - Bull, J. J., Molineux, I. J. & Rice, W. R. Selection of benevolence in a host–parasite system. Evolution 45, 875–882 (1991)
Article CAS PubMed Google Scholar - Cooper, V. S. et al. Timing of transmission and the evolution of virulence of an insect virus. Proc. R. Soc. Lond. B 269, 1161–1165 (2002)
Article Google Scholar - Ebert, D. Virulence and local adaptation of a horizontally transmitted parasite. Science 265, 1084–1086 (1994)
Article ADS CAS PubMed Google Scholar - Galvani, A. P. Epidemiology meets evolutionary ecology. Trends Ecol. Evol. 18, 132–139 (2003)
Article Google Scholar - Herre, E. A. Population structure and the evolution of virulence in nematode parasites of fig wasps. Science 259, 1442–1445 (1993)
Article ADS CAS PubMed Google Scholar - Lipsitch, M., Herre, E. A. & Nowak, M. A. Host population structure and the evolution of virulence: a 'law of diminishing returns'. Evolution 49, 743–748 (1995)
PubMed Google Scholar - Nowak, M. A. & May, R. M. Superinfection and the evolution of parasite virulence. Proc. R. Soc. Lond. B 255, 81–89 (1994)
Article ADS CAS Google Scholar - O'Keefe, K. J. & Antonovics, J. Playing by different rules: The evolution of virulence in sterilizing pathogens. Am. Nat. 159, 597–605 (2002)
Article PubMed Google Scholar - Thrall, P. H. & Burdon, J. J. Evolution of virulence in a plant host–pathogen metapopulation. Science 299, 1735–1737 (2003)
Article ADS CAS PubMed Google Scholar
Acknowledgements
We thank Y. Dang for help in the laboratory and the BioTechnology Resource Center at the University of Minnesota for robot access. We thank S. Abedon, C. Bergstrom, J. Bull, J. Fletcher, K. Koelle, C. Lehman, B. Levin and D. Stephens for useful feedback on this project and manuscript. This work was partially supported by an NSF grant to C.N. and an NIH grant to A.M.D. Author Contributions B.K., A.M.D. and B.J.M.B. designed the experiments. B.K. and A.M.D. worked out the robotic protocols. B.K. programmed the robot, executed the experiments, and conducted the assays. B.K. and C.N. coded and analysed the empirically calibrated and evolutionary models. B.K., C.N. and A.M.D. conducted the statistical analysis. All authors contributed to the writing of the manuscript.
Author information
Authors and Affiliations
- Department of Biology, University of Washington, Seattle, Box 351800, Washington, 98195-1800, USA
Benjamin Kerr - Department of Ecology, Evolution and Behaviour, University of Minnesota, 100 Ecology, 1987 Buford Circle, St Paul, Minnesota, 55108, USA
Claudia Neuhauser & Antony M. Dean - Department of Biological Sciences, Stanford University, Stanford, California, 94305, USA
Brendan J. M. Bohannan
Authors
- Benjamin Kerr
- Claudia Neuhauser
- Brendan J. M. Bohannan
- Antony M. Dean
Corresponding author
Correspondence toBenjamin Kerr.
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Methods (download PDF )
This file contains experimental and theoretical methods and results, giving a detailed description of the experimental methods used, presentation of experimental data, and its statistical analysis as well as the details concerning the construction and analysis of the theoretical models. (PDF 600 kb)
Rights and permissions
About this article
Cite this article
Kerr, B., Neuhauser, C., Bohannan, B. et al. Local migration promotes competitive restraint in a host–pathogen 'tragedy of the commons'.Nature 442, 75–78 (2006). https://doi.org/10.1038/nature04864
- Received: 12 March 2006
- Accepted: 04 May 2006
- Issue date: 06 July 2006
- DOI: https://doi.org/10.1038/nature04864
This article is cited by
Identifying Targets of Selection in Laboratory Evolution Experiments
- Artemiza A. Martínez
- Gregory I. Lang
Journal of Molecular Evolution (2023)
The scale of competition impacts parasite virulence evolution
- Elsa Noël
- Sophie Lefèvre
- Alison B. Duncan
Evolutionary Ecology (2023)
Multistep diversification in spatiotemporal bacterial-phage coevolution
- Einat Shaer Tamar
- Roy Kishony
Nature Communications (2022)
Shoaling guppies evade predation but have deadlier parasites
- Jason C. Walsman
- Mary J. Janecka
- Jessica F. Stephenson
Nature Ecology & Evolution (2022)
The evolution of cheating in viruses
- Asher Leeks
- Stuart A. West
- Melanie Ghoul
Nature Communications (2021)
Editorial Summary
Changing places
These T4 page and their E. coli hosts are the model for a typical 'victim-exploiter' interaction in a study of the role of migration patterns in a 'tragedy of the commons' competition for limited resources within fragmented communities. In this host-pathogen system, growing in 96-well microtitre plates, coexistence, stability and evolution within the separated communities depend critically on migration: restricted migration can promote restraint in the use of the common resource. In this experiment and in theory, highly connected social networks favour virulence.