Asterless is a scaffold for the onset of centriole assembly (original) (raw)
- Letter
- Published: 19 September 2010
- Quan D. Yu1 na1,
- Kipp Weiskopf1 nAff5,
- George Tzolovsky1,
- Ines Cunha-Ferreira1,2,
- Maria Riparbelli3,
- Ana Rodrigues-Martins1,2,
- Monica Bettencourt-Dias2,
- Giuliano Callaini3 &
- …
- David M. Glover1
Nature volume 467, pages 714–718 (2010)Cite this article
- 5920 Accesses
- 288 Citations
- Metrics details
Subjects
Abstract
Centrioles are found in the centrosome core and, as basal bodies, at the base of cilia and flagella. Centriole assembly and duplication is controlled by Polo-like-kinase 4 (Plk4): these processes fail if Plk4 is downregulated and are promoted by Plk4 overexpression1,2,3,4,5,6,7. Here we show that the centriolar protein Asterless (Asl; human orthologue CEP152) provides a conserved molecular platform, the amino terminus of which interacts with the cryptic Polo box of Plk4 whereas the carboxy terminus interacts with the centriolar protein Sas-4 (CPAP in humans). Drosophila Asl and human CEP152 are required for the centrosomal loading of Plk4 in Drosophila and CPAP in human cells, respectively. Depletion of Asl or CEP152 caused failure of centrosome duplication; their overexpression led to de novo centriole formation in Drosophila eggs, duplication of free centrosomes in Drosophila embryos, and centrosome amplification in cultured Drosophila and human cells. Overexpression of a Plk4-binding-deficient mutant of Asl prevented centriole duplication in cultured cells and embryos. However, this mutant protein was able to promote microtubule organizing centre (MTOC) formation in both embryos and oocytes. Such MTOCs had pericentriolar material and the centriolar protein Sas-4, but no centrioles at their core. Formation of such acentriolar MTOCs could be phenocopied by overexpression of Sas-4 in oocytes or embryos. Our findings identify independent functions for Asl as a scaffold for Plk4 and Sas-4 that facilitates self-assembly and duplication of the centriole and organization of pericentriolar material.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Asl recruits Plk4 to centrosomes for centriole duplication.

The alternative text for this image may have been generated using AI.
Figure 2: Asl overexpression causes de novo formation and amplification of centrioles.

The alternative text for this image may have been generated using AI.
Figure 3: Overexpression of Asl-M1 mutant or the Asl interactor Sas-4 leads to loss of centrioles.

The alternative text for this image may have been generated using AI.
Figure 4: CEP152 interacts with human PLK4 and CPAP and controls centrosome duplication in human cells.
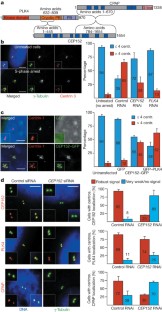
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Kleylein-Sohn, J. et al. Plk4-induced centriole biogenesis in human cells. Dev. Cell 13, 190–202 (2007)
Article CAS Google Scholar - Cunha-Ferreira, I. et al. The SCF/Slimb ubiquitin ligase limits centrosome amplification through degradation of SAK/PLK4. Curr. Biol. 19, 43–49 (2009)
Article CAS Google Scholar - Bettencourt-Dias, M. et al. SAK/PLK4 is required for centriole duplication and flagella development. Curr. Biol. 15, 2199–2207 (2005)
Article CAS Google Scholar - Rodrigues-Martins, A., Riparbelli, M., Callaini, G., Glover, D. M. & Bettencourt-Dias, M. Revisiting the role of the mother centriole in centriole biogenesis. Science 316, 1046–1050 (2007)
Article ADS CAS Google Scholar - Rogers, G. C., Rusan, N. M., Roberts, D. M., Peifer, M. & Rogers, S. L. The SCF Slimb ubiquitin ligase regulates Plk4/Sak levels to block centriole reduplication. J. Cell Biol. 184, 225–239 (2009)
Article CAS Google Scholar - Habedanck, R., Stierhof, Y. D., Wilkinson, C. J. & Nigg, E. A. The Polo kinase Plk4 functions in centriole duplication. Nature Cell Biol. 7, 1140–1146 (2005)
Article CAS Google Scholar - Peel, N., Stevens, N. R., Basto, R. & Raff, J. W. Overexpressing centriole-replication proteins in vivo induces centriole overduplication and de novo formation. Curr. Biol. 17, 834–843 (2007)
Article CAS Google Scholar - Varmark, H. et al. Asterless is a centriolar protein required for centrosome function and embryo development in Drosophila . Curr. Biol. 17, 1735–1745 (2007)
Article CAS Google Scholar - Blachon, S. et al. Drosophila asterless and vertebrate Cep152 are orthologs essential for centriole duplication. Genetics 180, 2081–2094 (2008)
Article CAS Google Scholar - Dobbelaere, J. et al. A genome-wide RNAi screen to dissect centriole duplication and centrosome maturation in Drosophila . PLoS Biol. 6, e224 (2008)
Article Google Scholar - Rodrigues-Martins, A. et al. DSAS-6 organizes a tube-like centriole precursor, and its absence suggests modularity in centriole assembly. Curr. Biol. 17, 1465–1472 (2007)
Article CAS Google Scholar - Leung, G. C. et al. The Sak polo-box comprises a structural domain sufficient for mitotic subcellular localization. Nature Struct. Biol. 9, 719–724 (2002)
Article CAS Google Scholar - Elia, A. E., Cantley, L. C. & Yaffe, M. B. Proteomic screen finds pSer/pThr-binding domain localizing Plk1 to mitotic substrates. Science 299, 1228–1231 (2003)
Article ADS CAS Google Scholar - Elia, A. E. et al. The molecular basis for phosphodependent substrate targeting and regulation of Plks by the Polo-box domain. Cell 115, 83–95 (2003)
Article CAS Google Scholar - Lowery, D. M. et al. Proteomic screen defines the Polo-box domain interactome and identifies Rock2 as a Plk1 substrate. EMBO J. 26, 2262–2273 (2007)
Article CAS Google Scholar - Lowery, D. M., Lim, D. & Yaffe, M. B. Structure and function of Polo-like kinases. Oncogene 24, 248–259 (2005)
Article CAS Google Scholar - Chang, J., Cizmecioglu, O., Hoffmann, I. & Rhee, K. PLK2 phosphorylation is critical for CPAP function in procentriole formation during the centrosome cycle. EMBO J. 29, 2395–2406 (2010)
Article CAS Google Scholar - D’Avino, P. P. et al. Isolation of protein complexes involved in mitosis and cytokinesis from Drosophila cultured cells. Methods Mol. Biol. 545, 99–112 (2009)
Article Google Scholar - Fox, J. D., Routzahn, K. M., Bucher, M. H. & Waugh, D. S. Maltodextrin-binding proteins from diverse bacteria and archaea are potent solubility enhancers. FEBS Lett. 537, 53–57 (2003)
Article CAS Google Scholar - D’Avino, P. P., Savoian, M. S., Capalbo, L. & Glover, D. M. RacGAP50C is sufficient to signal cleavage furrow formation during cytokinesis. J. Cell Sci. 119, 4402–4408 (2006)
Article Google Scholar - Basto, R. et al. Flies without centrioles. Cell 125, 1375–1386 (2006)
Article CAS Google Scholar - Mottier-Pavie, V. & Megraw, T. L. Drosophila Bld10 is a centriolar protein that regulates centriole, basal body, and motile cilium assembly. Mol. Biol. Cell 20, 2605–2614 (2009)
Article CAS Google Scholar - Warn, R. M. & Warn, A. Microtubule arrays present during the syncytial and cellular blastoderm stages of the early Drosophila embryo. Exp. Cell Res. 163, 201–210 (1986)
Article CAS Google Scholar - Frangioni, J. V. & Neel, B. G. Solubilization and purification of enzymatically active glutathione S-transferase (pGEX) fusion proteins. Anal. Biochem. 210, 179–187 (1993)
Article CAS Google Scholar
Acknowledgements
We thank E. Wegel for technical help. We thank P. Coelho, G. Mao, P. Gönczy, T. Megraw and J. Raff for antibodies. We thank Cancer Research UK for a Programme Grant to D.M.G. and a studentship to Q.D.Y. K.W. was a visiting scholar of the Winston Churchill Foundation of the United States. Fundação Calouste Gulbenkian, Fundação para a Ciência e Tecnologia (FCT) provided support to M.B.-D. and scholarships to I.C.-F. and A.R.-M. The Royal Society provided an International Joint Project Grant for collaboration between M.B.-D. and D.M.G.
Author information
Author notes
- Kipp Weiskopf
Present address: Present Address: Stanford University School of Medicine, Stanford, California 94305, USA., - Nikola S. Dzhindzhev and Quan D. Yu: These authors contributed equally to this work.
Authors and Affiliations
- Department of Genetics, Cancer Research UK Cell Cycle Genetics Group, University of Cambridge, Downing Street, Cambridge, CB2 3EH, UK
Nikola S. Dzhindzhev, Quan D. Yu, Kipp Weiskopf, George Tzolovsky, Ines Cunha-Ferreira, Ana Rodrigues-Martins & David M. Glover - Instituto Gulbenkian de Ciencia, Oeiras, P-2780-156, Portugal
Ines Cunha-Ferreira, Ana Rodrigues-Martins & Monica Bettencourt-Dias - Dipartimento di Biologia Evolutiva, Università di Siena, via Aldo Moro, 2-53100 Siena, Italy,
Maria Riparbelli & Giuliano Callaini
Authors
- Nikola S. Dzhindzhev
- Quan D. Yu
- Kipp Weiskopf
- George Tzolovsky
- Ines Cunha-Ferreira
- Maria Riparbelli
- Ana Rodrigues-Martins
- Monica Bettencourt-Dias
- Giuliano Callaini
- David M. Glover
Contributions
N.S.D. undertook interaction assays, mutagenesis and Drosophila cell culture work; Q.D.Y. worked on Drosophila embryos/eggs and de novo centriole formation; K.W. and I.C.-F. performed PrA–Plk4/Asl purifications; K.W. studied Asl depletion/overexpression in Drosophila cell culture. G.T. performed the human cell culture experiments. A.R.-M. and M.B.-D. overexpressed Sas-4 in embryos/eggs. M.R. and G.C. performed EM. N.S.D. and D.M.G. planned experiments and wrote the paper that was discussed by all authors.
Corresponding authors
Correspondence toNikola S. Dzhindzhev or David M. Glover.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
PowerPoint slides
Rights and permissions
About this article
Cite this article
Dzhindzhev, N., Yu, Q., Weiskopf, K. et al. Asterless is a scaffold for the onset of centriole assembly.Nature 467, 714–718 (2010). https://doi.org/10.1038/nature09445
- Received: 23 December 2009
- Accepted: 27 August 2010
- Published: 19 September 2010
- Issue date: 07 October 2010
- DOI: https://doi.org/10.1038/nature09445
This article is cited by
Editorial Summary
On the scaffold
Centrioles, cellular organelles composed of cylindrical arrays of nine microtubules, are essential for the formation of cilia, flagella and the centrosomes that organize microtubule structures within animal cells. Abnormal regulation of centriole duplication can lead to cancers and a number of ciliary diseases, and the centriolar protein Plk4 (Polo-like-kinase 4) is known to be a key regulator of centriole assembly. Now, David Glover and colleagues identify a single centriolar component, Asterless, as a scaffold for the binding of both Plk4 and another protein essential for centriole formation, Sas-4, with a key role in promoting centriole duplication.