Virally mediated optogenetic excitation and inhibition of pain in freely moving nontransgenic mice (original) (raw)
- Letter
- Published: 16 February 2014
- Kate L Montgomery1 na1,
- Chris Towne1,
- Soo Yeun Lee1,
- Charu Ramakrishnan1,
- Karl Deisseroth1,2 &
- …
- Scott L Delp1,3
Nature Biotechnology volume 32, pages 274–278 (2014) Cite this article
- 19k Accesses
- 211 Citations
- 116 Altmetric
- Metrics details
Subjects
Abstract
Primary nociceptors are the first neurons involved in the complex processing system that regulates normal and pathological pain1. Because of constraints on pharmacological and electrical stimulation, noninvasive excitation and inhibition of these neurons in freely moving nontransgenic animals has not been possible. Here we use an optogenetic2 strategy to bidirectionally control nociceptors of nontransgenic mice. Intrasciatic nerve injection of adeno-associated viruses encoding an excitatory opsin enabled light-inducible stimulation of acute pain, place aversion and optogenetically mediated reductions in withdrawal thresholds to mechanical and thermal stimuli. In contrast, viral delivery of an inhibitory opsin enabled light-inducible inhibition of acute pain perception, and reversed mechanical allodynia and thermal hyperalgesia in a model of neuropathic pain. Light was delivered transdermally, allowing these behaviors to be induced in freely moving animals. This approach may have utility in basic and translational pain research, and enable rapid drug screening and testing of newly engineered opsins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Intrasciatic injection of AAV6-hSyn-ChR2-eYFP results in transduction of unmyelinated nociceptors projecting to spinal cord lamina I/IIo.
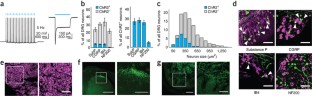
The alternative text for this image may have been generated using AI.
Figure 2: Transdermal illumination of AAV6-hSyn-ChR2-eYFP–injected mice results in tunable pain-like behavior and sensitizes mice to mechanical and thermal stimuli.
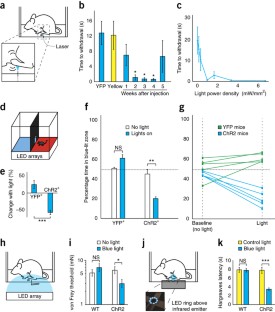
The alternative text for this image may have been generated using AI.
Figure 3: Transdermal illumination of AAV6-hSyn-NpHR-eYFP–injected mice desensitizes mice to mechanical and thermal stimuli and reverses mechanical allodynia and thermal hyperalgesia caused by a chronic constriction injury (CCI).
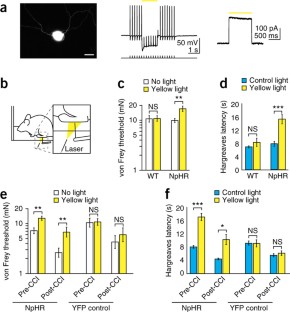
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Dubin, A.E. & Patapoutian, A. Review series: nociceptors: the sensors of the pain pathway. J. Clin. Invest. 120, 3760–3772 (2010).
Article CAS PubMed PubMed Central Google Scholar - Fenno, L., Yizhar, O. & Deisseroth, K. The development and application of optogenetics. Annu. Rev. Neurosci. 34, 389–412 (2011).
Article CAS PubMed PubMed Central Google Scholar - Liske, H. et al. Optical inhibition of motor nerve and muscle activity in vivo. Muscle Nerve 47, 916–921 (2013).
Article PubMed PubMed Central Google Scholar - Llewellyn, M.E., Thompson, K.R., Deisseroth, K. & Delp, S.L. Orderly recruitment of motor units under optical control in vivo. Nat. Med. 16, 1161–1165 (2010).
Article CAS PubMed PubMed Central Google Scholar - Ji, Z.-G. et al. Light-evoked somatosensory perception of transgenic rats that express channelrhodopsin-2 in dorsal root ganglion cells. PLoS ONE 7, e32699 (2012).
Article CAS PubMed PubMed Central Google Scholar - Wang, H. & Zylka, M.J. Mrgprd-expressing polymodal nociceptive neurons innervate most known classes of substantia gelatinosa neurons. J. Neurosci. 29, 13202–13209 (2009).
Article CAS PubMed PubMed Central Google Scholar - Mourot, A. et al. Rapid optical control of nociception with an ion-channel photoswitch. Nat. Methods 9, 396–402 (2012).
Article CAS PubMed PubMed Central Google Scholar - Kokel, D. et al. Photochemical activation of TRPA1 channels in neurons and animals. Nat. Chem. Biol. 9, 257–263 (2013).
Article CAS PubMed PubMed Central Google Scholar - Anonymous. Enlightened engineering. Nat. Biotechnol. 29, 849 (2011).
- Towne, C., Montgomery, K.L., Iyer, S.M., Deisseroth, K. & Delp, S.L. Optogenetic control of targeted peripheral axons in freely moving animals. PLoS ONE 8, e72691 (2013).
Article CAS PubMed PubMed Central Google Scholar - Daou, I. et al. Remote optogenetic activation and sensitization of pain pathways in freely moving mice. J. Neurosci. 33, 18631–18640 (2013).
Article CAS PubMed PubMed Central Google Scholar - Mattis, J. et al. Principles for applying optogenetic tools derived from direct comparative analysis of microbial opsins. Nat. Methods 9, 159–172 (2012).
Article CAS Google Scholar - Williams, J.C. & Denison, T. From optogenetic technologies to neuromodulation therapies. Sci. Transl. Med. 5, 177ps6 (2013).
Article PubMed Google Scholar - Chow, B.Y. & Boyden, E.S. Optogenetics and translational medicine. Sci. Transl. Med. 5, 177ps5 (2013).
PubMed Google Scholar - Towne, C., Schneider, B.L., Kieran, D., Redmond, D.E. & Aebischer, P. Efficient transduction of non-human primate motor neurons after intramuscular delivery of recombinant AAV serotype 6. Gene Ther. 17, 141–146 (2010).
Article CAS PubMed Google Scholar - San Sebastian, W. et al. Adeno-associated virus type 6 is retrogradely transported in the non-human primate brain. Gene Ther. 20, 1178–1183 (2013).
Article CAS PubMed Google Scholar - Asokan, A., Schaffer, D.V. & Samulski, R.J. The AAV vector toolkit: poised at the clinical crossroads. Mol. Ther. 20, 699–708 (2012).
Article CAS PubMed PubMed Central Google Scholar - Towne, C., Pertin, M., Beggah, A.T., Aebischer, P. & Decosterd, I. Recombinant adeno-associated virus serotype 6 (rAAV2/6)-mediated gene transfer to nociceptive neurons through different routes of delivery. Mol. Pain 5, 52 (2009).
Article PubMed PubMed Central Google Scholar - Mogil, J.S. Animal models of pain: progress and challenges. Nat. Rev. Neurosci. 10, 283–294 (2009).
Article CAS PubMed Google Scholar - Mason, M.R.J. et al. Comparison of AAV serotypes for gene delivery to dorsal root ganglion neurons. Mol. Ther. 18, 715–724 (2010).
Article CAS PubMed PubMed Central Google Scholar - Bennett, G.J. & Xie, Y.K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 33, 87–107 (1988).
Article CAS PubMed Google Scholar - Lin, J.Y., Knutsen, P.M., Muller, A., Kleinfeld, D. & Tsien, R.Y. ReaChR: a red-shifted variant of channelrhodopsin enables deep transcranial optogenetic excitation. Nat. Neurosci. 16, 1499–1508 (2013).
Article CAS PubMed PubMed Central Google Scholar - Bashkatov, A.N., Genina, E.A., Kochubey, V.I. & Tuchin, V.V. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J. Phys. D Appl. Phys. 38, 2543–2555 (2005).
Article CAS Google Scholar - Yizhar, O. et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature 477, 171–178 (2011).
Article CAS PubMed PubMed Central Google Scholar - Konermann, S. et al. Optical control of mammalian endogenous transcription and epigenetic states. Nature 500, 472–476 (2013).
Article CAS PubMed PubMed Central Google Scholar - Sohal, V.S., Zhang, F., Yizhar, O. & Deisseroth, K. Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 459, 698–702 (2009).
Article CAS PubMed PubMed Central Google Scholar - Tillman, D.B., Treede, R.D., Meyer, R.A. & Campbell, J.N. Response of C fibre nociceptors in the anaesthetized monkey to heat stimuli: estimates of receptor depth and threshold. J. Physiol. (Lond.) 485, 753–765 (1995).
Article CAS Google Scholar - Gao, G.-P. et al. Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc. Natl. Acad. Sci. USA 99, 11854–11859 (2002).
Article CAS PubMed PubMed Central Google Scholar - Lee, J.H. et al. Global and local fMRI signals driven by neurons defined optogenetically by type and wiring. Nature 465, 788–792 (2010).
Article CAS PubMed PubMed Central Google Scholar - Kügler, S., Kilic, E. & Bähr, M. Human synapsin 1 gene promoter confers highly neuron-specific long-term transgene expression from an adenoviral vector in the adult rat brain depending on the transduced area. Gene Ther. 10, 337–347 (2003).
Article PubMed Google Scholar - Gold, M.S. Whole-cell recording in isolated primary sensory neurons. Methods Mol. Biol. 851, 73–97 (2012).
Article CAS PubMed Google Scholar - Hentschke, H. & Stüttgen, M.C. Computation of measures of effect size for neuroscience data sets. Eur. J. Neurosci. 34, 1887–1894 (2011).
Article PubMed Google Scholar - Chaplan, S.R., Bach, F.W., Pogrel, J.W., Chung, J.M. & Yaksh, T.L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 53, 55–63 (1994).
Article CAS PubMed Google Scholar - Dixon, W.J. Efficient analysis of experimental observations. Annu. Rev. Pharmacol. Toxicol. 20, 441–462 (1980).
Article CAS PubMed Google Scholar - Sommer, C. & Schäfers, M. Painful mononeuropathy in C57BL/Wld mice with delayed Wallerian degeneration: differential effects of cytokine production and nerve regeneration on thermal and mechanical hypersensitivity. Brain Res. 784, 154–162 (1998).
Article CAS PubMed Google Scholar
Acknowledgements
We thank H. Liske, X. Qian, S. Mackey, A. Weitz, D.J. Clark, D.C. Yeomans, P. Sabhaie, C. Gorini, S. Young and H. Scutt for useful discussions and assistance with experiments. This study was supported by the US National Institutes of Health (National Institute of Neurological Disorders and Stroke grant R01-NS080954), the Stanford Bio-X NeuroVentures program and the Stanford Bio-X Interdisciplinary Initiatives program. S.M.I. was supported by an Office of Technology Licensing Stanford Graduate Fellowship and by a Howard Hughes Medical Institute International Student Research Fellowship. K.L.M. was supported by a Bio-X Bioengineering Graduate Fellowship and by a Stanford Interdisciplinary Graduate Fellowship. C.T. was supported by a Swiss National Science Foundation Fellowship.
Author information
Author notes
- Shrivats Mohan Iyer and Kate L Montgomery: These authors contributed equally to this work.
- Department of Psychiatry and Behavioral Sciences, Stanford University, Stanford, California, USA.
Authors and Affiliations
- Department of Bioengineering, Stanford University, Stanford, California, USA
Shrivats Mohan Iyer, Kate L Montgomery, Chris Towne, Soo Yeun Lee, Charu Ramakrishnan, Karl Deisseroth & Scott L Delp - Howard Hughes Medical Institute, Stanford University, Stanford, California, USA
Karl Deisseroth - Department of Mechanical Engineering, Stanford University, Stanford, California, USA
Scott L Delp
Authors
- Shrivats Mohan Iyer
- Kate L Montgomery
- Chris Towne
- Soo Yeun Lee
- Charu Ramakrishnan
- Karl Deisseroth
- Scott L Delp
Contributions
S.M.I., K.L.M., C.T. and S.L.D. designed the experiments. S.M.I. and K.L.M. performed the experiments. C.R. performed cell culture and created vectors. S.Y.L. performed electrophysiology. K.D. contributed reagents and tools. S.M.I., K.L.M. and S.L.D. wrote and edited the paper, with comments from all other authors.
Corresponding author
Correspondence toScott L Delp.
Ethics declarations
Competing interests
C.T., K.D., and S.L.D. have a financial interest in Circuit Therapeutics, Inc., which, however, did not support this work. S.M.I., K.L.M., C.T., K.D. and S.L.D. have disclosed these findings to the Stanford Office of Technology Licensing for potential use in the identification of new treatments for pain.
Supplementary information
Supplementary Video 1 (download MOV )
Video demonstration of transdermal optogenetic activation of nociceptors. A mouse with bilateral ChR2 expression is placed in a clear cylinder on a transparent glass plate, and allowed to habituate to its environment. When blue light (473 nm, 1 mW/mm2) is shone on the plantar surface of the mouse's skin it immediately withdraws its paw, engages in prolonged licking behavior, and shakes its paw. However, when yellow light (593 nm, 1 mW/mm2) is shone on the skin, there is no observable change in behavior, and the mouse moves around normally. Also, when blue light (473 nm, 1 mW/mm2) is shone on the paw of a bilaterally injected YFP+ mouse, no change in behavior is seen, and the mouse moves around normally. (MOV 8136 kb)
Rights and permissions
About this article
Cite this article
Iyer, S., Montgomery, K., Towne, C. et al. Virally mediated optogenetic excitation and inhibition of pain in freely moving nontransgenic mice.Nat Biotechnol 32, 274–278 (2014). https://doi.org/10.1038/nbt.2834
- Received: 13 October 2013
- Accepted: 17 January 2014
- Published: 16 February 2014
- Issue date: March 2014
- DOI: https://doi.org/10.1038/nbt.2834