Charcot–Marie–Tooth disease (original) (raw)
In brief
- The cardinal clinical feature is peripheral neuropathy: lower motor neuron-type motor deficits and sensory signs and symptoms. As neuropathy can be associated with many multisytemic disorders, it is required to be the predominant manifestation.
- Clinical phenotypes are established by age of onset, neurophysiological findings and in some cases by neuropathology.
- Clinical phenotypes include Charcot–Marie–Tooth disease type 1 (CMT1), Charcot–Marie–Tooth disease type 2 (CMT2), Dejerine–Sottas neuropathy (DSN), congenital hypomyelinating neuropathy (CHN) and Roussy–Levy syndrome (RLS).
- Genetic heterogeneity, age-dependent penetrance and variable expressivity are key characteristics of CMT and related peripheral neuropathies.
- Thirty-six loci and more than two dozen genes are involved in CMT, implicating pathways in myelination, radial and axonal transport, Schwann cell differentiation, signal transduction, mitochondrial function, endosome, protein translation and single-stranded DNA break repair.
- The plethora of genetic information necessitates a rational approach to genetic testing.
- CMT is one of the genetic conditions in which molecular-based therapies are progressing to the clinical trial phase.
Introduction
The prevalence of Charcot–Marie–Tooth disease (CMT) is 1 per 2500 population, which results in 125 000 patients in the United States alone, making it the most common inherited neurological disease. Over the last 15 years, molecular genetics research identified over two dozen genes in which mutations cause the CMT phenotype. In vitro functional assays and experiments in animal models of specific genetic alterations elucidated the pathomechanisms by which mutations in certain genes cause disease and delineated pathways involved in peripheral nerve biology. Some of these genes/mutations contribute to a significant fraction of inherited peripheral neuropathy cases and thus molecular analysis can have a substantial function in establishing a precise molecular diagnosis. Population-based cohorts established the contribution of the individual genes to disease burden, allowing evidence-based prioritization of genetic testing. Compounds have been shown to ameliorate symptoms in animal models progressing the field toward clinical trials. This in turn stimulated clinical research to establish the natural history of the disease and to develop tools to assess outcome in prospective clinical trials.
Clinical overview
Cardinal features of CMT
The main features of CMT are a combination of lower motor neuron-type motor deficits and sensory signs and symptoms, reflecting the sensory-motor neuropathy. Length-dependent paresis and muscle atrophy develops, with areflexia, although a subset of patients will retain deep tendon reflexes, especially in the axonal forms. The chronic nature of the motor neuropathy will result in foot deformity (eg, pes cavus), hammertoes and high-arched feet. Involvement of the hands may follow as the disease progresses. Sensory symptoms are less frequent than in acquired chronic neuropathies, but may point to specific gene involvement. Signs of sensory system dysfunction are common (70%) and include loss of vibration and joint position sense followed by decreased pain and temperature sensation in stocking and glove distribution. Clinical features do not distinguish between the demyelinating or axonal forms.
Ancillary diagnostic tests include electrophysiological studies and sural nerve biopsy. Recently, peripheral nerve MRI and skin biopsy have emerged as potential diagnostic aids in certain types of hereditary neuropathies, though further research studies are needed. EMG and nerve conduction studies (NCS) are extremely helpful in the clinical classification of hereditary peripheral neuropathies and in guiding genetic testing. Electrophysiological studies distinguish two major types – the demyelinating form, which is characterized by symmetrically slowed nerve conduction velocity (NCV; usually <38 m/s), and the axonal form, which is associated with normal or subnormal NCV and reduced compound muscle action potential. The term intermediate CMT is used without consensus in the literature. It identifies the group of patients who cannot be classified readily as either CMT1 or CMT2, as they tend to have features of both demyelination and axonopathy. The NCV falls in the 30–45 m range, with overlap with both the demyelinating and the axonal form.1 If this pattern is recognized, certain genes are more likely to be involved than others (eg, GJB1 and MPZ).
Sural nerve biopsies from patients with the demyelinating type reveal segmental demyelination and onion bulb formation, whereas the nerve biopsies from patients with the axonal form show axonal loss, absent or few onion bulbs and no evidence of demyelination. With the advent of genetic testing, invasive diagnostic tests such as nerve biopsy are reserved for patients in whom genetic testing does not yield to a molecular diagnosis, patients with atypical presentation or patients in whom inflammatory neuropathy is suspected.
Depending on age of onset and neurophysiological findings, several clinical phenotypes have been described historically. As molecular characterization of phenotypes became available, genetic and clinical heterogeneity of the hereditary motor and sensory neuropathies (HMSNs) became apparent.
Disease phenotypes
Charcot–Marie–Tooth Disease (MIM 118200, 118220)
As CMT1 and CMT2 present with similar clinical features, distinction on the basis of the neurological exam is often impossible. The onset of clinical symptoms is in the first or second decade of life. Weakness starts distally in the feet and progresses proximally in an ascending pattern. Neuropathic bony deformities develop including pes cavus (high-arched feet) and hammer toes. With further progression the hands become weak. Muscle stretch reflexes disappear early in the ankles and later in the patella and upper limbs. Mild sensory loss to pain, temperature or vibration sensation in the legs is consistent with the phenotype. Patients also complain of numbness and tingling in their feet and hands, but paresthesias are not as common as in acquired neuropathies. Restless leg syndrome occurs in nearly 40% of patients with the axonal form.2
Hereditary neuropathy with liability to pressure palsies (MIM 162500)
The clinical phenotype is characterized by recurrent nerve dysfunction at compression sites. Asymmetric palsies occur after relatively minor compression or trauma. Repeated attacks result in the inability of full reversal. Thus with ageing the patients with hereditary neuropathy with liability to pressure palsies (HNPP) can have significant clinical overlap with CMT1. Electrophysiological findings include mildly slowed NCV, increased distal motor latencies and conduction blocks.3 The neuropathological hallmark is sausage-like thickening of myelin sheaths (tomacula).
Dejerine–Sottas neuropathy (MIM 145900)
Dejerine–Sottas neuropathy (DSN) is a clinically distinct entity defined by delayed motor milestones. Signs of lower motor neuron-type lesion accompany the delayed motor milestones. Neurophysiological studies reveal severe slowing of NCV (<10 m/s). Neuropathology reveals pronounced demyelination, and a greater number of onion bulbs are present compared to CMT. Cerebrospinal fluid proteins may be elevated. Most patients have significant disability.
Congentital hypomyelinating neuropathy (MIM 605253)
Congentital hypomyelinating neuropathy (CHN) is usually present at birth, although frequently the delayed motor development draws the first attention to the peripheral neuropathy. The distinction between DSN and CHN is often difficult by clinical examination as they both may present as a hypotonic infant. The differentiation of CHN and DSN is based on pathology: the presence of onion bulbs suggest DSN whereas their absence indicate CHN. CHN may present as arthrogryposis multiplex congenita.4
Roussy–Levy syndrome (MIM 180800)
Roussy–Levy syndrome (RLS) was originally described as demyelinating CMT associated with sensory ataxia and tremor. As molecular data became available, it was shown that these patients have the same molecular abnormalities as observed in patients clinically classified as demyelinating CMT. RLS represents the spectrum of CMT.
Differential diagnosis of CMT
Peripheral neuropathy has a broad differential diagnosis: it can be the only manifestation, part of a complex neurological phenotype or part of a multisystemic disorder. Careful search for other affected organ systems or central nervous system (CNS) involvement during the history and physical examination is of utmost importance. Laboratory screening for correctable causes should always be performed, including screening for diabetes, vitamin B deficiency and serum immunofixation electrophoresis, especially in the adult population. Marked CNS involvement makes CMT less likely; in these cases leukodystrophies, mitochondrial disorders, the hereditary ataxias with neuropathy (Friedreich ataxia, abetalipoproteinemia), Refsum disease, Pelizeaus–Merzbacher disease and amyloid neuropathies should be considered. Hereditary sensory neuropathies lack motor symptoms and are associated with autonomic dysfunction. The lower motor neuron-type weakness may mimic a distal myopathy; however, electrophysiology is useful in differentiating between the two.
CMT is predominantly a peripheral neuropathy phenotype; however, certain features are consistent with the disease and in fact may even help guide the molecular genetic testing. Sensorineuronal hearing loss is present in 5% of the patients. Adie's pupil is almost pathognomic for the Thr124Met mutation in MPZ.5 Ophthalmoparesis, facial weakness, vocal cord paralysis and bulbar signs reflect cranial nerve involvement; these are common in EGR2 mutations.6 Hyperkeratosis and juvenile glaucoma are associated with mutations in NEFL7 and MTMR138 genes, respectively. Scoliosis is present in 20% of the cases and is a secondary phenomenon caused by the neuromuscular weakness.
Inheritance pattern
All forms of Mendelian inheritance – autosomal dominant (AD), autosomal recessive (AR) and X-linked (XL) – can be seen in CMT families. The AD demyelinating form is the most frequent pattern observed.9 Out of the 36 linked loci, 14 are AD, 13 AR and 3 XL. HNPP and RLS show AD inheritance whereas CHN is AR or sporadic. DSN has both AD and AR forms. Genotype–phenotype correlation studies suggest that genetic heterogeneity, age-dependent penetrance and variable expressivity significantly contribute to the genetics of CMT. It is estimated that about one-third of the point mutations and 5–24% of the duplication mutation may occur de novo;10, 11, 12 thus, the absence of family history does not preclude genetic testing.
Classification
The classification system for CMT and related peripheral neuropathies was initially developed on the basis of the clinical phenotype, electrophysiological and inheritance patterns (Table 1). This classification was derived from clinical data on large pedigrees and served as an invaluable tool in identifying genes responsible for certain types of CMT. The molecular classification added further refinement and introduced ambiguities. Genes identified as a specific locus-associated and type-associated gene were found to be responsible for other types of CMT, or with a different inheritance pattern depending on the specific mutant allele.
Table 1 Genetic classification of Charcot–Marie–Tooth disease and related peripheral neuropathies
Genetics
The more than two dozen genes (Table 1) implicated in the HMSNs belong to various functional classes, all involved in the biology of peripheral nerve development and function. They include structural proteins that are important in myelination (eg, PMP22, MPZ), radial transport proteins (eg, Cx32), proteins of axonal transport (eg, NEFL), transcription factors involved in Schwann cell differentiation (EGR2), members of signal transduction pathways (eg, PRX, MTMR2, SBF2, NDGR1), proteins related to mitochondrial function (eg, MFN2, GDAP1), proteins related to the endosome (RAB7, SIMPLE) and molecular chaperones (HSP22, HSP27), a gene involved in DNA single-stranded break repair (TDP1), and genes involved in protein translation (GARS, YARS), in nuclear envelope function (LMNA) and in the actin cytoskeleton (DNM2). A detailed summary of all the contributing genes is beyond the scope of this clinical review and has been summarized.13, 14 Table 2 summarizes the genes, their functions and the associated phenotypes.
Table 2 Genotype–phenotype correlation
Genetic testing
The genetic complexity of CMT necessitates a rational approach for clinical genetic testing. Factors to consider when initiating genetic testing should include careful evaluation of (1) the availability of clinical testing, (2) the yield of a specific molecular test, (3) the aim of establishing a molecular diagnosis and (4) the frequency of de novo mutations.
Evidence-based data from 12 population-based studies from various ethnic backgrounds10, 11, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 established the contribution of 5 genes/genomic rearrangements to disease burden: PMP22 duplication/deletion; MPZ, Cx32 and PMP22 point mutations. Electrophysiological classification (demyelinating versus axonal neuropathy) markedly improves the diagnostic yield (Table 3). In families, with informative pedigrees to determine the inheritance pattern, further targeting of the diagnostic testing can be achieved.19, 25
Table 3 Mutation frequencies for CMT and related neuropathies
Duplication of a chromosomal segment harboring PMP22 (ie, the CMT1A duplication)26 represents 43% of the total CMT cases, whereas the yield of duplication detection rises to 70% in CMT1. The deletion of the same chromosomal segment results in HNPP.27 Although the deletion has not been reported in any other phenotype, the yield of deletion testing is over 90% in this distinctive phenotype.
Cx32 mutations are the next most common culprits in inherited neuropathy. In informative pedigrees a dominant inheritance pattern and lack of male-to-male transmission points to this gene on the X chromosome. Because electrophysiology frequently suggests the intermediate form, molecular testing for Cx32 is appropriate in both CMT1 (after duplication testing) and CMT2. In the CMT1 group, MPZ and PMP22 mutations are the next most common, followed by the rare genes.25 In the CMT2 group, Cx32 mutations are followed by MPZ mutations in frequency; however, recent data, though not population based, suggest that MFN2 mutations may be one of the most common causes of CMT2.28, 29, 30
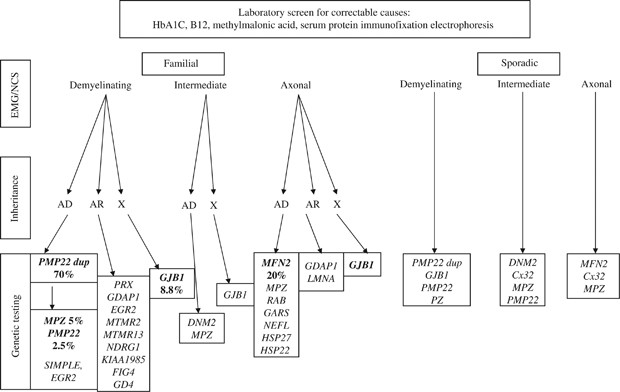
The high frequency of de novo mutations in duplication/deletion (37–90%)10, 12 and in point mutations11 illustrates that genetic disease is commonly sporadic in presentation. The absence of a family history does not exclude CMT and related peripheral neuropathies. In fact, in a patient presenting with chronic polyneuropathy in the absence of other signs and symptoms, after the most common systemic and treatable causes, such as diabetes, uremia and nutritional deficiency, genetic causes are more common than autoimmune or paraneoplastic neuropathy. A rational diagnostic approach is presented in Figure 1. In pediatric cases, which are more severe and when reproductive plans may depend on the genetic information, complete evaluation with panel testing is warranted.
Figure 1
Suggested testing scheme in hereditary sensory and motor polyneuropathy for patients with and without a family history of CMT based on the genotype–phenotype correlations and frequency data in 12 population-based studies.
Management
Treatment approaches to the HMSNs can be supportive or etiologic. As CMT is a slowly progressive neurodegenerative disease, patients require periodic assessments. Physiotherapy and occupational therapy aid in maintaining range of motion and thus help in functioning appropriately.31, 32 The application of orthotic devices and assistive equipment can be made if safety or function requires them. In some instances, surgical interventions for the hands and feet are necessary.33, 34
Symptomatic treatment may have a substantial impact on the quality of life. Nonsteroidal anti-inflammatory drugs may help to relieve lower back or leg pain. Neuropathic pain can be treated with antiepileptic drugs (gabapentin, pregabalin, topiramate) or tricyclic antidepressants (amitriptyline).35, 36 The tremor may respond to _β_-blockers or primidone.37 Caffeine and nicotine can aggravate the fine intentional tremor, thus avoidance of these substances is recommended. Neurotoxic drugs (http://www.charcot-marie-tooth.org/) and excessive alcohol should be avoided. A small dose of vincristine can produce a devastating effect in patients with CMT, thus early detection of HMSN can avoid life-threatening vincristine neurotoxicity.38
Potential therapeutic approaches aiming at normalizing dosage by small molecules in the CMT1A duplication models include vitamin C and onapristone, a progesterone antagonist.39, 40, 41 An alternate molecular mechanism, point mutations in Pmp22 in the Trembler and Trembler J mouse models cause peripheral neuropathy; the disease was modified by the administration of curcumin likely by alleviating the unfolded protein response.42 These treatments have been shown to be effective only in animal models thus far; however, vitamin C has progressed to a phase 2 clinical trial.
Genetic counseling
Because CMT follows the principles of Mendelian inheritance, genetic counseling for recurrence of CMT1 and CMT2 is relatively straightforward if the family history for an affected individual is defined. Because of intrafamilial variability in disease expression, definition of parental disease status requires either testing for a mutation defined in the propositus or, if the mutation is not identifiable, a thorough neurological exam with objective NCS.
An affected parent with AD or XL-dominant CMT1 or CMT2 has a 50% risk of having a child with the same mutation. At what age a child with a mutation will be clinically affected is not known because the penetrance has not been determined prospectively for genetically well-defined patient populations. In general only a few patients with AD CMT1 or CMT2 have substantial difficulty walking before age 50 years, and almost all patients express some symptoms by the sixth decade of life.43 For fathers with XL-dominant CMT, the risk of having an affected son is negligible but the risk of having an affected daughter is 100%, whereas for mothers with XL-dominant CMT, the risk of having an affected son or daughter is 50%.
In the absence of a molecular diagnosis in AD CMT1, NCV slowing is detectable by age 2–5 years;44, 45 therefore, if a young adult has normal NCVs, their risk of developing AD CMT1 is negligible, whereas if the NCVs are abnormal, the patient has at least a 90% lifetime risk of developing symptoms. Electrophysiological changes associated with AD CMT2 develop with disease progression, thus only about half of patients can be identified by age 20 years.43 In one study performed before the molecular era in 15 unrelated families, the average age of onset was 12.2 years. The penetrance was 28% in the first decade, but almost complete by the third decade.46
When unaffected parents have a child affected with CMT1 or CMT2, four possibilities exist: a de novo dominant mutation in the affected child, AR inheritance, XL inheritance or nonpaternity. Distinction between these possibilities requires either the identification of the causative mutation(s) or the identification of affected siblings. The identification of a de novo heterozygous presumed dominant mutation suggests a low recurrence risk for the parents; however, the risk is higher than that for the general population because of the possibility of germ-line mosaicism.47 A proband with a heterozygous presumed dominant mutation has a 50% risk of having affected children. For AR inheritance, the parental risk of an affected child is 25% because penetrance is nearly complete.
Summary
CMT is one of the most prevalent neurogenetic conditions, with a plethora of accumulated knowledge of the genes and pathways implicated in peripheral nerve function and dysfunction. Although a lot remains to be learnt, clinical research has aided the estimation of the contribution of specific genes to disease burden. Animal models provide the basis for preclinical treatment trials in which small compounds modifying gene expression to normalize gene dosage and potentially modulating protein misfolding have been identified. Clinical research has developed tools to assess outcome in clinical trials48 and data on disease progression are accumulating. Thus, we have all the tools to move to the exciting translational research phase, where patients can potentially benefit from the translation of laboratory discoveries at the bedside.
References
- Jani-Acsadi A, Krajewski K, Shy ME : Charcot–Marie–Tooth neuropathies: diagnosis and management. Semin Neurol 2008; 28: 185–194.
Article Google Scholar - Gemignani F, Marbini A, Di Giovanni G, Salih S, Terzano MG : Charcot–Marie–Tooth disease type 2 with restless legs syndrome. Neurology 1999; 52: 1064–1066.
Article CAS Google Scholar - Li J, Krajewski K, Shy ME, Lewis RA : Hereditary neuropathy with liability to pressure palsy: the electrophysiology fits the name. Neurology 2002; 58: 1769–1773.
Article Google Scholar - Boylan KB, Ferriero DM, Greco CM, Sheldon RA, Dew M : Congenital hypomyelination neuropathy with arthrogryposis multiplex congenita. Ann Neurol 1992; 31: 337–340.
Article CAS Google Scholar - De Jonghe P, Timmerman V, Ceuterick C et al: The Thr124Met mutation in the peripheral myelin protein zero (MPZ) gene is associated with a clinically distinct Charcot–Marie–Tooth phenotype. Brain 1999; 122 (Part 2): 281–290.
Article Google Scholar - Szigeti K, Wiszniewski W, Saifi GM et al: Functional, histopathologic and natural history study of neuropathy associated with EGR2 mutations. Neurogenetics 2007; 8: 257–262.
Article Google Scholar - Jordanova A, De Jonghe P, Boerkoel CF et al: Mutations in the neurofilament light chain gene (NEFL) cause early onset severe Charcot–Marie–Tooth disease. Brain 2003; 126: 590–597.
Article CAS Google Scholar - Azzedine H, Bolino A, Taieb T et al: Mutations in MTMR13, a new pseudophosphatase homologue of MTMR2 and Sbf1, in two families with an autosomal recessive demyelinating form of Charcot–Marie–Tooth disease associated with early-onset glaucoma. Am J Hum Genet 2003; 72: 1141–1153.
Article CAS Google Scholar - Szigeti K, Lupski JR : Charcot-Marie-Tooth peripheral neuropathies and related disorders; in: Scriver CR, Beaudet AL, Valle D et al. (eds): The Metabolic and Molecular Bases of Inherited Disease. New York: McGraw-Hill, 2006.
- Nelis E, Van Broeckhoven C, De Jonghe P et al: Estimation of the mutation frequencies in Charcot–Marie–Tooth disease type 1 and hereditary neuropathy with liability to pressure palsies: a European collaborative study. Eur J Hum Genet 1996; 4: 25–33.
Article CAS Google Scholar - Boerkoel CF, Takashima H, Garcia CA et al: Charcot–Marie–Tooth disease and related neuropathies: mutation distribution and genotype-phenotype correlation. Ann Neurol 2002; 51: 190–201.
Article CAS Google Scholar - Hoogendijk JE, Hensels GW, Gabreels-Festen AA et al: De-novo mutation in hereditary motor and sensory neuropathy type I. Lancet 1992; 339: 1081–1082.
Article CAS Google Scholar - Nave KA, Sereda MW, Ehrenreich H : Mechanisms of disease: inherited demyelinating neuropathies – from basic to clinical research. Nat Clin Pract Neurol 2007; 3: 453–464.
Article CAS Google Scholar - Zuchner S, Vance JM : Mechanisms of disease: a molecular genetic update on hereditary axonal neuropathies. Nat Clin Pract Neurol 2006; 2: 45–53.
Article CAS Google Scholar - Wise CA, Garcia CA, Davis SN et al: Molecular analyses of unrelated Charcot–Marie–Tooth (CMT) disease patients suggest a high frequency of the CMTIA duplication. Am J Hum Genet 1993; 53: 853–863.
CAS PubMed PubMed Central Google Scholar - Choi BO, Lee MS, Shin SH et al: Mutational analysis of PMP22, MPZ, GJB1, EGR2 and NEFL in Korean Charcot–Marie–Tooth neuropathy patients. Hum Mutat 2004; 24: 185–186.
Article Google Scholar - Janssen EA, Kemp S, Hensels GW et al: Connexin32 gene mutations in X-linked dominant Charcot–Marie–Tooth disease (CMTX1). Hum Genet 1997; 99: 501–505.
Article CAS Google Scholar - Nicholson GA : Mutation testing in Charcot–Marie–Tooth neuropathy. Ann NY Acad Sci 1999; 883: 383–388.
Article CAS Google Scholar - Mostacciuolo ML, Righetti E, Zortea M et al: Charcot–Marie–Tooth disease type I and related demyelinating neuropathies: mutation analysis in a large cohort of Italian families. Hum Mutat 2001; 18: 32–41.
Article CAS Google Scholar - Silander K, Meretoja P, Juvonen V et al: Spectrum of mutations in Finnish patients with Charcot–Marie–Tooth disease and related neuropathies. Hum Mutat 1998; 12: 59–68.
Article CAS Google Scholar - Bort S, Nelis E, Timmerman V et al: Mutational analysis of the MPZ, PMP22 and Cx32 genes in patients of Spanish ancestry with Charcot–Marie–Tooth disease and hereditary neuropathy with liability to pressure palsies. Hum Genet 1997; 99: 746–754.
Article CAS Google Scholar - Leonardis L, Zidar J, Ekici A, Peterlin B, Rautenstrauss B : Autosomal dominant Charcot–Marie–Tooth disease type 1A and hereditary neuropathy with liability to pressure palsies: detection of the recombination in Slovene patients and exclusion of the potentially recessive Thr118Met PMP22 point mutation. Int J Mol Med 1998; 1: 495–501.
CAS PubMed Google Scholar - Mersiyanova IV, Ismailov SM, Polyakov AV et al: Screening for mutations in the peripheral myelin genes PMP22, MPZ and Cx32 (GJB1) in Russian Charcot–Marie–Tooth neuropathy patients. Hum Mutat 2000; 15: 340–347.
Article CAS Google Scholar - Marques Jr W, Freitas MR, Nascimento OJ et al: 17p duplicated Charcot–Marie–Tooth 1A: characteristics of a new population. J Neurol 2005; 252: 972–979.
Article CAS Google Scholar - Szigeti K, Garcia CA, Lupski JR : Charcot–Marie–Tooth disease and related hereditary polyneuropathies: molecular diagnostics determine aspects of medical management. Genet Med 2006; 8: 86–92.
Article Google Scholar - Lupski JR, de Oca-Luna RM, Slaugenhaupt S et al: DNA duplication associated with Charcot–Marie–Tooth disease type 1A. Cell 1991; 66: 219–232.
Article CAS Google Scholar - Chance PF, Alderson MK, Leppig KA et al: DNA deletion associated with hereditary neuropathy with liability to pressure palsies. Cell 1993; 72: 143–151.
Article CAS Google Scholar - Zuchner S, Mersiyanova IV, Muglia M et al: Mutations in the mitochondrial GTPase mitofusin 2 cause Charcot–Marie–Tooth neuropathy type 2A. Nat Genet 2004; 36: 449–451.
Article Google Scholar - Kijima K, Numakura C, Izumino H et al: Mitochondrial GTPase mitofusin 2 mutation in Charcot–Marie–Tooth neuropathy type 2A. Hum Genet 2005; 116: 23–27.
Article CAS Google Scholar - Verhoeven K, Claeys KG, Zuchner S et al: MFN2 mutation distribution and genotype/phenotype correlation in Charcot–Marie–Tooth type 2. Brain 2006; 129: 2093–2102.
Article Google Scholar - Lindeman E, Spaans F, Reulen J, Leffers P, Drukker J : Progressive resistance training in neuromuscular patients. Effects on force and surface EMG. J Electromyogr Kinesiol 1999; 9: 379–384.
Article CAS Google Scholar - Njegovan ME, Leonard EI, Joseph FB : Rehabilitation medicine approach to Charcot–Marie–Tooth disease. Clin Podiatr Med Surg 1997; 14: 99–116.
CAS PubMed Google Scholar - Mann DC, Hsu JD : Triple arthrodesis in the treatment of fixed cavovarus deformity in adolescent patients with Charcot–Marie–Tooth disease. Foot Ankle 1992; 13: 1–6.
Article CAS Google Scholar - Guyton GP, Mann RA : The pathogenesis and surgical management of foot deformity in Charcot–Marie–Tooth disease. Foot Ankle Clin 2000; 5: 317–326.
CAS PubMed Google Scholar - Backonja MM : Use of anticonvulsants for treatment of neuropathic pain. Neurology 2002; 59: S14–S17.
Article Google Scholar - Szigeti K, Lupski JR : Hereditary Motor and Sensory Neuropathies; in: Rimoin DL, Connor JM, Pyeritz RE, Korf BR (eds): Principles and Practice of Medical Genetics, Philadelphia: Churchill Livingstone Elsevier, 2007.
Google Scholar - Koller WC, Hristova A, Brin M : Pharmacologic treatment of essential tremor. Neurology 2000; 54: S30–S38.
Article CAS Google Scholar - Naumann R, Mohm J, Reuner U, Kroschinsky F, Rautenstrauss B, Ehninger G : Early recognition of hereditary motor and sensory neuropathy type 1 can avoid life-threatening vincristine neurotoxicity. Br J Haematol 2001; 115: 323–325.
Article CAS Google Scholar - Sereda MW, Meyer zu Horste G, Suter U, Uzma N, Nave KA : Therapeutic administration of progesterone antagonist in a model of Charcot–Marie–Tooth disease (CMT-1A). Nat Med 2003; 9: 1533–1537.
Article CAS Google Scholar - Passage E, Norreel JC, Noack-Fraissignes P et al: Ascorbic acid treatment corrects the phenotype of a mouse model of Charcot–Marie–Tooth disease. Nat Med 2004; 10: 396–401.
Article CAS Google Scholar - Meyer zu Horste G, Prukop T, Liebetanz D, Mobius W, Nave KA, Sereda MW : Antiprogesterone therapy uncouples axonal loss from demyelination in a transgenic rat model of CMT1A neuropathy. Ann Neurol 2007; 61: 61–72.
Article CAS Google Scholar - Khajavi M, Shiga K, Wiszniewski W et al: Oral curcumin mitigates the clinical and neuropathologic phenotype of the Trembler-J mouse: a potential therapy for inherited neuropathy. Am J Hum Genet 2007; 81: 438–453.
Article CAS Google Scholar - Harding AE, Thomas PK : The clinical features of hereditary motor and sensory neuropathy types I and II. Brain 1980; 103: 259–280.
Article CAS Google Scholar - Kaku DA, Parry GJ, Malamut R, Lupski JR, Garcia CA : Uniform slowing of conduction velocities in Charcot–Marie–Tooth polyneuropathy type 1. Neurology 1993; 43: 2664–2667.
Article CAS Google Scholar - Kaku DA, Parry GJ, Malamut R, Lupski JR, Garcia CA : Nerve conduction studies in Charcot–Marie–Tooth polyneuropathy associated with a segmental duplication of chromosome 17. Neurology 1993; 43: 1806–1808.
Article CAS Google Scholar - Bird TD, Kraft GH : Charcot–Marie–Tooth disease: data for genetic counseling relating age to risk. Clin Genet 1978; 14: 43–49.
Article CAS Google Scholar - Takashima H, Nakagawa M, Kanzaki A et al: Germline mosaicism of MPZ gene in Dejerine–Sottas syndrome (HMSN III) associated with hereditary stomatocytosis. Neuromuscul Disord 1999; 9: 232–238.
Article CAS Google Scholar - Shy ME, Blake J, Krajewski K et al: Reliability and validity of the CMT neuropathy score as a measure of disability. Neurology 2005; 64: 1209–1214.
Article CAS Google Scholar
Author information
Authors and Affiliations
- Department of Neurology, Baylor College of Medicine, Houston, TX, USA
Kinga Szigeti - Department of Molecular and Human Genetics, Baylor College of Medicine, Texas Children Hospital, Houston, TX, USA
Kinga Szigeti & James R Lupski - Department of Pediatrics, Baylor College of Medicine, Houston, TX, USA
James R Lupski
Authors
- Kinga Szigeti
You can also search for this author inPubMed Google Scholar - James R Lupski
You can also search for this author inPubMed Google Scholar
Corresponding author
Correspondence toJames R Lupski.
Additional information
Disclosure
JRL is a co-inventor of patented molecular diagnostic test for CMT (US patents 5 306 616; 5 599 920; 5 780 223; 7 273 698) and a consultant for Athena Diagnostics.
Rights and permissions
About this article
Cite this article
Szigeti, K., Lupski, J. Charcot–Marie–Tooth disease.Eur J Hum Genet 17, 703–710 (2009). https://doi.org/10.1038/ejhg.2009.31
- Received: 02 October 2008
- Accepted: 27 January 2009
- Published: 11 March 2009
- Issue Date: June 2009
- DOI: https://doi.org/10.1038/ejhg.2009.31