Capacity limit of visual short-term memory in human posterior parietal cortex (original) (raw)
- Letter
- Published: 15 April 2004
Nature volume 428, pages 751–754 (2004) Cite this article
- 12k Accesses
- 1334 Citations
- 43 Altmetric
- Metrics details
Abstract
At any instant, our visual system allows us to perceive a rich and detailed visual world. Yet our internal, explicit representation of this visual world is extremely sparse: we can only hold in mind a minute fraction of the visual scene1,2. These mental representations are stored in visual short-term memory (VSTM). Even though VSTM is essential for the execution of a wide array of perceptual and cognitive functions3,4,5, and is supported by an extensive network of brain regions6,7,8,9, its storage capacity is severely limited10,11,12,13. With the use of functional magnetic resonance imaging, we show here that this capacity limit is neurally reflected in one node of this network: activity in the posterior parietal cortex is tightly correlated with the limited amount of scene information that can be stored in VSTM. These results suggest that the posterior parietal cortex is a key neural locus of our impoverished mental representation of the visual world.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Trial design.
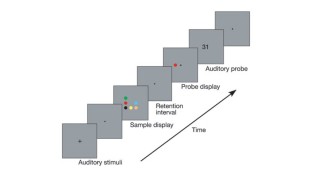
The alternative text for this image may have been generated using AI.
Figure 2: Behavioural performance and IPS/IOS response functions in VSTM and IM experiments.
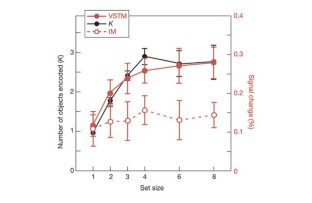
The alternative text for this image may have been generated using AI.
Figure 3: Brain activation time courses.
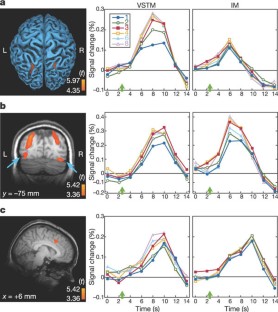
The alternative text for this image may have been generated using AI.
Figure 4: Response time courses during the encoding, maintenance and retrieval phases of a VSTM task with extended retention interval (9,200 ms).

The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Rensink, R. A. Change detection. Annu. Rev. Psychol. 53, 245–277 (2002)
Article Google Scholar - Simons, D. & Levin, D. Change blindness. Trends Cogn. Sci. 1, 261–267 (1997)
Article CAS Google Scholar - Chun, M. M. & Potter, M. C. A two-stage model for multiple target detection in rapid serial visual presentation. J. Exp. Psychol. Hum. Percept. Perform. 21, 109–127 (1995)
Article CAS Google Scholar - Jolicoeur, P., Dell' Acqua, R. & Crebolder, J. M. in The Limits of Attention: Temporal Constraints in Human Information Processing (ed. Shapiro, K.) 82–99 (Oxford Univ. Press, 2001)
Book Google Scholar - Wheeler, M. E. & Treisman, A. M. Binding in short-term visual memory. J. Exp. Psychol. Gen. 131, 48–64 (2002)
Article Google Scholar - Goldman-Rakic, P. S. in Handbook of Physiology: The Nervous System, Higher Functions of the Brain (eds Mountcastle, V. B. & Plum, F.) 373–417 (American Physiological Society, Bethesda, Maryland, 1987)
Google Scholar - Ungerleider, L. G., Courtney, S. M. & Haxby, J. V. A neural system for human visual working memory. Proc. Natl Acad. Sci. USA 95, 883–890 (1998)
Article ADS CAS Google Scholar - Callicott, J. H. et al. Physiological characteristics of capacity constraints in working memory as revealed by functional MRI. Cereb. Cortex 9, 20–26 (1999)
Article CAS Google Scholar - Linden, D. E. et al. Cortical capacity constraints for visual working memory: Dissociation of fMRI load effects in a fronto-parietal network. Neuroimage 20, 1518–1530 (2003)
Article Google Scholar - Duncan, J. et al. Systematic analysis of deficits in visual attention. J. Exp. Psychol. Gen. 128, 450–478 (1999)
Article CAS Google Scholar - Cowan, N. The magical number 4 in short-term memory: A reconsideration of mental storage capacity. Behav. Brain Sci. 24, 87–114 (2001)
Article CAS Google Scholar - Pashler, H. Familiarity and visual change detection. Percept. Psychophys. 44, 369–378 (1988)
Article CAS Google Scholar - Vogel, E. K., Woodman, G. F. & Luck, S. J. Storage of features, conjunctions and objects in visual working memory. J. Exp. Psychol. Hum. Percept. Perform. 27, 92–114 (2001)
Article CAS Google Scholar - Cohen, J. D. et al. Temporal dynamics of brain activation during a working memory task. Nature 386, 604–608 (1997)
Article ADS CAS Google Scholar - Phillips, W. A. On the distinction between sensory storage and short-term visual memory. Percept. Psychophys. 16, 283–290 (1974)
Article Google Scholar - Coltheart, M. in New Horizons in Psychology (ed. Dodwell, P. C.) 62–85 (Harmondsworth, Penguin, 1972)
Google Scholar - Baddeley, A. Working memory. Science 255, 556–559 (1992)
Article ADS CAS Google Scholar - Smith, E. E. & Jonides, J. Neuroimaging analyses of human working memory. Proc. Natl Acad. Sci. USA 95, 12061–12068 (1998)
Article ADS CAS Google Scholar - Rosnow, R. L. & Rosenthal, R. Contrasts and interactions redux: Five easy pieces. Psychol. Sci. 7, 253–257 (1996)
Article Google Scholar - Courtney, S. M., Ungerleider, L. G., Keil, K. & Haxby, J. V. Transient and sustained activity in a distributed neural system for human working memory. Nature 386, 608–611 (1997)
Article ADS CAS Google Scholar - Pessoa, K., Gutierrez, E., Bandettini, P. A. & Ungerleider, L. G. Neural correlates of visual working memory: fMRI amplitude predicts task performance. Neuron 35, 975–987 (2002)
Article CAS Google Scholar - Zarahn, E., Aguirre, G. & D'Esposito, M. A trial-based experimental design for fMRI. Neuroimage 6, 122–138 (1997)
Article CAS Google Scholar - Friedman-Hill, S. R., Robertson, L. C. & Treisman, A. Parietal contributions to visual feature binding: Evidence from a patient with bilateral lesions. Science 269, 853–855 (1995)
Article ADS CAS Google Scholar - Shafritz, K. M., Gore, J. C. & Marois, R. The role of the parietal cortex in visual feature binding. Proc. Natl Acad. Sci. USA 99, 10917–10922 (2002)
Article ADS CAS Google Scholar - Haxby, J. V. et al. Dissociation of object and spatial visual processing pathways in human extrastriate cortex. Proc. Natl Acad. Sci. USA 88, 1621–1625 (1991)
Article ADS CAS Google Scholar - McKeefry, D. J. & Zeki, S. The position and topography of the human colour centre as revealed by functional magnetic resonance imaging. Brain 120, 2229–2242 (1997)
Article Google Scholar - Curtis, C. E. & D'Esposito, M. Persistent activity in the prefrontal cortex during working memory. Trends Cogn. Sci. 7, 415–423 (2003)
Article Google Scholar - Beck, D. M., Rees, G., Frith, C. D. & Lavie, N. Neural correlates of change detection and change blindness. Nature Neurosci. 4, 645–650 (2001)
Article CAS Google Scholar - Gottlieb, J. P., Kusunoki, M. & Goldberg, M. E. The representation of visual salience in monkey parietal cortex. Nature 391, 481–484 (1998)
Article ADS CAS Google Scholar - Kourtzi, Z. & Kanwisher, N. Representation of perceived object shape by the human lateral occipital complex. Science 293, 1506–1509 (2001)
Article ADS CAS Google Scholar
Acknowledgements
We thank I. Gauthier, M. Chun, G. Logan and J. Schall for comments on earlier versions of this manuscript, and D. Nikolaiczyk-Stocks and A. Snyder for expert technical assistance. This work was supported by a grant from the NSF to R.M.
Author information
Authors and Affiliations
- Vanderbilt Vision Research Center, Department of Psychology, Vanderbilt University, 530 Wilson Hall, Nashville, Tennessee, 37203, USA
J. Jay Todd & René Marois
Authors
- J. Jay Todd
- René Marois
Corresponding author
Correspondence toRené Marois.
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Todd, J., Marois, R. Capacity limit of visual short-term memory in human posterior parietal cortex.Nature 428, 751–754 (2004). https://doi.org/10.1038/nature02466
- Received: 11 January 2004
- Accepted: 09 March 2004
- Issue date: 15 April 2004
- DOI: https://doi.org/10.1038/nature02466