Tunable metallic-like conductivity in microbial nanowire networks (original) (raw)
- Letter
- Published: 07 August 2011
- Madeline Vargas2,3,
- Kelly P. Nevin2,
- Ashley E. Franks2,
- Ching Leang2,
- Byoung-Chan Kim2 nAff5,
- Kengo Inoue2 nAff5,
- Tünde Mester2 nAff5,
- Sean F. Covalla2 nAff5,
- Jessica P. Johnson2 nAff5,
- Vincent M. Rotello4,
- Mark T. Tuominen1 &
- …
- Derek R. Lovley2
Nature Nanotechnology volume 6, pages 573–579 (2011) Cite this article
- 16k Accesses
- 872 Citations
- 105 Altmetric
- Metrics details
Subjects
Abstract
Electronic nanostructures made from natural amino acids are attractive because of their relatively low cost, facile processing and absence of toxicity1,2,3. However, most materials derived from natural amino acids are electronically insulating1,2,3,4,5,6. Here, we report metallic-like conductivity in films of the bacterium Geobacter sulfurreducens7 and also in pilin nanofilaments (known as microbial nanowires8,9) extracted from these bacteria. These materials have electronic conductivities of ∼5 mS cm−1, which are comparable to those of synthetic metallic nanostructures2. They can also conduct over distances on the centimetre scale, which is thousands of times the size of a bacterium. Moreover, the conductivity of the biofilm can be tuned by regulating gene expression, and also by varying the gate voltage in a transistor configuration. The conductivity of the nanofilaments has a temperature dependence similar to that of a disordered metal, and the conductivity could be increased by processing.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Strategy to measure in situ biofilm conductivity.
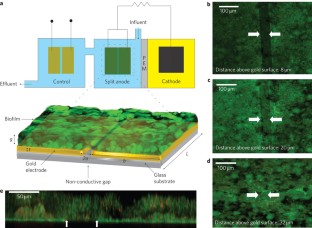
The alternative text for this image may have been generated using AI.
Figure 2: Measurement setup and conductance data.
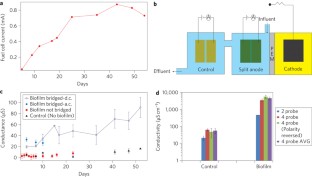
The alternative text for this image may have been generated using AI.
Figure 3: Evidence for pili being associated with biofilm conductivity.
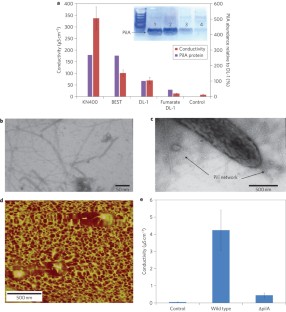
The alternative text for this image may have been generated using AI.
Figure 4: Observation of metallic-like nature of conductivity.
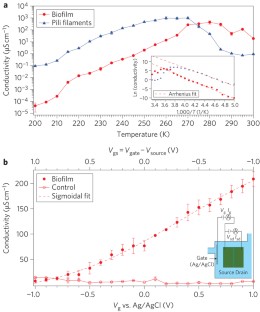
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Hauser, C. & Zhang, S. Nanotechnology: peptides as biological semiconductors. Nature 468, 516–517 (2010).
Article CAS Google Scholar - Skotheim, T. A. & Reynolds, J. R. Handbook of Conducting Polymers (CRC, 2007).
Google Scholar - Ashkenasy, N., Horne, W. & Ghadiri, M. Design of self-assembling peptide nanotubes with delocalized electronic states. Small 2, 99–102 (2006).
Article CAS Google Scholar - Dheilly, A. et al. Monitoring of microbial adhesion and biofilm growth using electrochemical impedancemetry. Appl. Microbiol. Biotechnol. 79, 157–164 (2008).
Article CAS Google Scholar - Muñoz-Berbel, X., Muñoz, F. J., Vigués, N. & Mas, J. On-chip impedance measurements to monitor biofilm formation in the drinking water distribution network. Sens. Actuat. B 118, 129–134 (2006).
Article Google Scholar - Herbert-Guillou, D., Tribollet, B., Festy, D. & Kiéné, L. In situ detection and characterization of biofilm in water by electrochemical methods. Electrochim. Acta 45, 1067–1075 (1999).
Article CAS Google Scholar - Yi, H. et al. Selection of a variant of Geobacter sulfurreducens with enhanced capacity for current production in microbial fuel cells. Biosens. Bioelectron. 24, 3498–3503 (2009).
Article CAS Google Scholar - Reguera, G. et al. Extracellular electron transfer via microbial nanowires. Nature 435, 1098–1101 (2005).
Article CAS Google Scholar - Gorby, Y. A. et al. Electrically conductive bacterial nanowires produced by Shewanella oneidensis strain MR-1 and other microorganisms. Proc. Natl Acad. Sci. USA 103, 11358–11363 (2006).
Article CAS Google Scholar - Smart, A. G. Two experiments, two takes on electric bacteria. Phys. Today 63, 18–20 (December 2010).
Article Google Scholar - Heeger, A. J., Sariciftci, N. S. & Namdas, E. B. Semiconducting and Metallic Polymers (Oxford Univ. Press, 2010).
Google Scholar - Kaiser, A. Systematic conductivity behaviour in conducting polymers: effects of heterogeneous disorder. Adv. Mater. 13, 927–941 (2001).
Article CAS Google Scholar - Noy, A., Artyukhin, A. & Misra, N. Bionanoelectronics with 1D materials. Mater. Today 12, 22–31 (2009).
Article Google Scholar - Huebsch, N. & Mooney, D. J. Inspiration and application in the evolution of biomaterials. Nature 462, 426–432 (2009).
Article CAS Google Scholar - Yuen, J. et al. Electrochemical doping in electrolyte-gated polymer transistors. J. Am. Chem. Soc. 129, 14367–14371 (2007).
Article CAS Google Scholar - Nevin, K. P. et al. Power output and coulombic efficiencies from biofilms of Geobacter sulfurreducens comparable to mixed community microbial fuel cells. Environ. Microbiol. 10, 2505–2514 (2008).
Article CAS Google Scholar - Reguera, G. et al. Biofilm and nanowire production leads to increased current in Geobacter sulfurreducens fuel cells. Appl. Environ. Microbiol. 72, 7345–7348 (2006).
Article CAS Google Scholar - Lange, U. & Mirsky, V. M. Separated analysis of bulk and contact resistance of conducting polymers: comparison of simultaneous two- and four-point measurements with impedance measurements. J. Electroanal. Chem. 622, 246–251 (2008).
Article CAS Google Scholar - Schwan, H. P. Electrode polarization impedance and measurements in biological materials. Ann. NY Acad. Sci. 148, 191–209 (1968).
Article CAS Google Scholar - Hadziioannou, G. & Van Hutter, P. F. Semiconducting Polymers: Chemistry, Physics, and Engineering 2nd edn, Vol. 2 (Wiley-VCH, 2007).
Google Scholar - Kankare, J. & Kupila, E. L. In-situ conductance measurement during electropolymerization. J. Electroanal. Chem. 322, 167–181 (1992).
Article CAS Google Scholar - Lanthier, M., Gregory, K. B. & Lovley, D. R. Growth with high planktonic biomass in Shewanella oneidensis fuel cells. FEMS Microbiol. Lett. 278, 29–35 (2008).
Article CAS Google Scholar - Voordeckers, J. W., Kim, B.-C., Izallalen, M. & Lovley, D. R. Role of Geobacter sulfurreducens outer surface _c_-type cytochromes in reduction of soil humic acid and anthraquinone-2,6-disulfonate. Appl. Environ. Microbiol. 76, 2371–2375 (2010).
Article CAS Google Scholar - Dallas, P. et al. Characterization, magnetic and transport properties of polyaniline synthesized through interfacial polymerization. Polymer 48, 3162–3169 (2007).
Article CAS Google Scholar - Abthagir, P. S. & Saraswathi, R. Charge transport and thermal properties of polyindole, polycarbazole and their derivatives. Thermochim. Acta 424, 25–35 (2004).
Article CAS Google Scholar - Lee, K. et al. Metallic transport in polyaniline. Nature 441, 65–68 (2006).
Article CAS Google Scholar - Zotti, G. et al. Potential-driven conductivity of polypyrroles, poly-_n_-alkylpyrroles, and polythiophenes: role of the pyrrole nh moiety in the doping-charge dependence of conductivity. Chem. Mater. 15, 4642–4650 (2003).
Article CAS Google Scholar - Zotti, G. et al. Conductivity in redox modified conducting polymers. 2. Enhanced redox conductivity in ferrocene-substituted polypyrroles and polythiophenes. Chem. Mater. 7, 2309–2315 (1995).
Article CAS Google Scholar - Lindsay, S. Molecular wires and devices: advances and issues. Faraday Discuss. 131, 403–409 (2006).
Article CAS Google Scholar - Chiang, J. C. & MacDiarmid, A. G. ‘Polyaniline’: protonic acid doping of the emeraldine form to the metallic regime. Synth. Metals 13, 193–205 (1986).
Article CAS Google Scholar - Agrell, H., Boschloo, G. & Hagfeldt, A. Conductivity studies of nanostructured TiO2 films permeated with electrolyte. J. Phys. Chem. B 108, 12388–12396 (2004).
Article CAS Google Scholar - Hélaine, S. et al. PilX, a pilus-associated protein essential for bacterial aggregation, is a key to pilus-facilitated attachment of Neisseria meningitidis to human cells. Mol. Microbiol. 55, 65–77 (2005).
Article Google Scholar - Roine, E. et al. Hrp pilus: a hrp-dependent bacterial surface appendage produced by Pseudomonas syringae pv. tomato DC3000. Proc. Natl Acad. Sci. USA 94, 3459–3464 (1997).
Article CAS Google Scholar
Acknowledgements
The authors thank D. Venkataraman, C. Salthouse, M. Aklujkar, J. Nicholson, R. Krotvov, A. Ursache, O. Yavuzcetin and S. Ebru Yalcin for helpful discussions and technical assistance. This research was supported by the Office of Naval Research (grant no. N00014-10-1-0084), the Office of Science (BER), US Department of Energy (award no. DE-SC0004114 and Cooperative Agreement no. DE-FC02-02ER63446 as well as the NSF Center for Hierarchical Manufacturing (grant no. CMMI-0531171)).
Author information
Author notes
- Byoung-Chan Kim, Kengo Inoue, Tünde Mester, Sean F. Covalla & Jessica P. Johnson
Present address: Present address: Korea Research Institute of Bioscience and Biotechnology, Yusong-gu, Daejeon 305-806, South Korea (B.C.K.); Interdisciplinary Research Organization, University of Miyazaki, Kiyotake, Miyazaki 889-1692, Japan (K.I.); University of Michigan Medical School and Veterans Affairs Medical Research Centre, Ann Arbor, Michigan 48105, USA. (T.M.); Mascoma Corporation, Lebanon, New Hampshire 03766, USA (S.F.C. and J.P.J.),
Authors and Affiliations
- Department of Physics, University of Massachusetts, Amherst, 01003, Massachusetts, USA
Nikhil S. Malvankar & Mark T. Tuominen - Department of Microbiology, University of Massachusetts, Amherst, 01003, Massachusetts, USA
Nikhil S. Malvankar, Madeline Vargas, Kelly P. Nevin, Ashley E. Franks, Ching Leang, Byoung-Chan Kim, Kengo Inoue, Tünde Mester, Sean F. Covalla, Jessica P. Johnson & Derek R. Lovley - Department of Biology, College of the Holy Cross, Worcester, 01610, Massachusetts, USA
Madeline Vargas - Department of Chemistry, University of Massachusetts, Amherst, 01003, Massachusetts, USA
Vincent M. Rotello
Authors
- Nikhil S. Malvankar
- Madeline Vargas
- Kelly P. Nevin
- Ashley E. Franks
- Ching Leang
- Byoung-Chan Kim
- Kengo Inoue
- Tünde Mester
- Sean F. Covalla
- Jessica P. Johnson
- Vincent M. Rotello
- Mark T. Tuominen
- Derek R. Lovley
Contributions
The experiments were designed by N.S.M., K.P.N. and M.T.T., with suggestions from A.E.F., S.F.C, V.M.T. and D.R.L. N.S.M. performed electrical measurements, X-ray studies and AFM imaging of pili preparations. M.V. prepared and TEM-imaged pilin filaments and performed haem staining. N.S.M., M.V., B.C.K., K.I. and T.M. performed protein measurements. B.C.K. generated the BEST strain. C.L. generated the CL-1 strain and performed the peeling and TEM-imaging of biofilms. A.E.F. and J.P.J. carried out the confocal imaging of biofilms. N.S.M., M.T.T. and D.R.L. analysed the data and wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Correspondence toMark T. Tuominen or Derek R. Lovley.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Malvankar, N., Vargas, M., Nevin, K. et al. Tunable metallic-like conductivity in microbial nanowire networks.Nature Nanotech 6, 573–579 (2011). https://doi.org/10.1038/nnano.2011.119
- Received: 18 April 2011
- Accepted: 23 June 2011
- Published: 07 August 2011
- Issue date: September 2011
- DOI: https://doi.org/10.1038/nnano.2011.119