Ultrafast memory loss and energy redistribution in the hydrogen bond network of liquid H2O (original) (raw)
- Letter
- Published: 10 March 2005
- B. D. Bruner1 na1,
- N. Huse2 na1,
- J. R. Dwyer1,
- B. Chugh1,
- E. T. J. Nibbering2,
- T. Elsaesser2 &
- …
- R. J. D. Miller1
Nature volume 434, pages 199–202 (2005) Cite this article
- 8656 Accesses
- 722 Citations
- 131 Altmetric
- Metrics details
Abstract
Many of the unusual properties of liquid water are attributed to its unique structure, comprised of a random and fluctuating three-dimensional network of hydrogen bonds that link the highly polar water molecules1,2. One of the most direct probes of the dynamics of this network is the infrared spectrum of the OH stretching vibration3,4,5,6,7,8,9,10,11, which reflects the distribution of hydrogen-bonded structures and the intermolecular forces controlling the structural dynamics of the liquid. Indeed, water dynamics has been studied in detail5,6,7,8,9,10,11,12,13,14, most recently using multi-dimensional nonlinear infrared spectroscopy15,16 for acquiring structural and dynamical information on femtosecond timescales. But owing to technical difficulties, only OH stretching vibrations in D2O or OD vibrations in H2O could be monitored. Here we show that using a specially designed, ultrathin sample cell allows us to observe OH stretching vibrations in H2O. Under these fully resonant conditions, we observe hydrogen bond network dynamics more than one order of magnitude faster than seen in earlier studies that include an extremely fast sweep in the OH frequencies on a 50-fs timescale and an equally fast disappearance of the initial inhomogeneous distribution of sites. Our results highlight the efficiency of energy redistribution within the hydrogen-bonded network, and that liquid water essentially loses the memory of persistent correlations in its structure within 50 fs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Experimental set-up.
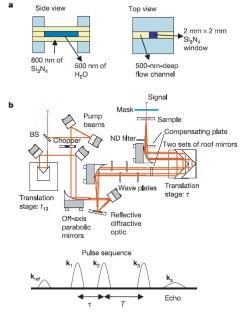
The alternative text for this image may have been generated using AI.
Figure 2: Spectrally integrated transient grating data in pure H2O.
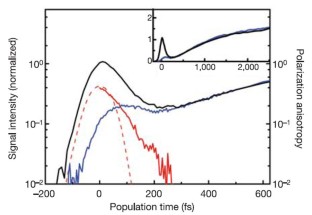
The alternative text for this image may have been generated using AI.
Figure 3: Absorptive component of the spectrally resolved transient grating signal, plotted as a function of population time T.
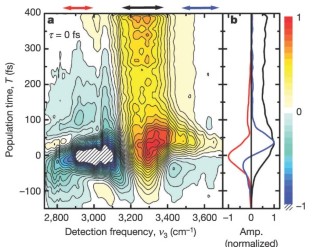
The alternative text for this image may have been generated using AI.
Figure 4: Absorptive components of the two-dimensional-infrared echo spectra of pure liquid H2O for different population times.
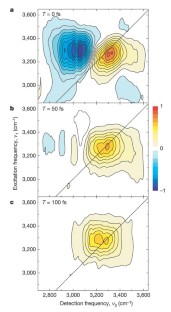
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Eisenberg, D. & Kauzmann, W. The Structure and Properties of Water (Oxford Univ. Press, New York, 1969)
Google Scholar - Franks, F. (ed.) Water, a Comprehensive Treatise (Plenum, New York, 1972)
- Luzar, A. & Chandler, D. Hydrogen-bond kinetics in liquid water. Nature 379, 55–57 (1996)
Article ADS CAS Google Scholar - Marx, D., Tuckerman, M. E., Hutter, J. & Parrinello, M. The nature of the hydrated excess proton in water. Nature 397, 601–604 (1999)
Article ADS CAS Google Scholar - Graener, H., Seifert, G. & Laubereau, A. New spectroscopy of water using tunable picosecond pulses in the infrared. Phys. Rev. Lett. 66, 2092–2095 (1991)
Article ADS CAS Google Scholar - Woutersen, S. & Bakker, H. J. Resonant intermolecular transfer of vibrational energy in liquid water. Nature 402, 507–509 (1999)
Article ADS CAS Google Scholar - Gale, G. M. et al. Femtosecond dynamics of hydrogen bonds in liquid water: A real-time study. Phys. Rev. Lett. 82, 1068–1071 (1999)
Article ADS CAS Google Scholar - Stenger, J., Madsen, D., Hamm, P., Nibbering, E. T. J. & Elsaesser, T. Ultrafast vibrational dephasing of liquid water. Phys. Rev. Lett. 87, 027401 (2001)
Article ADS Google Scholar - Møller, K. B., Rey, R. & Hynes, J. T. Hydrogen bond dynamics in water and ultrafast infrared spectroscopy: a theoretical study. J. Phys. Chem. A 108, 1275–1289 (2004)
Article Google Scholar - Lawrence, C. P. & Skinner, J. L. Vibrational spectroscopy of HOD in liquid D2O. Infrared line shapes and vibrational Stokes shift. J. Chem. Phys. 117, 8847–8854 (2002)
Article ADS CAS Google Scholar - Torre, R., Bartolini, P. & Righini, R. Structural relaxation in supercooled water by time-resolved spectroscopy. Nature 428, 296–299 (2004)
Article ADS CAS Google Scholar - Asbury, J. B. et al. Water dynamics: vibrational echo correlation spectroscopy and comparison to molecular dynamics simulations. J. Phys. Chem. A 108, 1107–1119 (2004)
Article CAS Google Scholar - Fecko, C. J., Eaves, J. D., Loparo, J. J., Tokmakoff, A. & Geissler, P. L. Ultrafast hydrogen-bond dynamics in the infrared spectroscopy of water. Science 301, 1698–1702 (2003)
Article ADS CAS Google Scholar - Stenger, J., Madsen, D., Hamm, P., Nibbering, E. T. J. & Elsaesser, T. A photon echo peak shift study of liquid water. J. Phys. Chem. A 106, 2341–2350 (2002)
Article CAS Google Scholar - Asplund, M. C., Zanni, M. T. & Hochstrasser, R. M. Two dimensional infrared spectroscopy of peptides by phase-controlled femtosecond vibrational photon echoes. Proc. Natl Acad. Sci. USA 97, 8219–8224 (2000)
Article ADS CAS Google Scholar - Mukamel, S. Multidimensional femtosecond correlation spectroscopies of electronic and vibrational excitations. Annu. Rev. Phys. Chem. 51, 691–729 (2000)
Article ADS CAS Google Scholar - Cowan, M. L., Ogilvie, J. P. & Miller, R. J. D. Two-dimensional spectroscopy using diffractive optics based phased-locked photon echoes. Chem. Phys. Lett. 386, 184–189 (2004)
Article ADS CAS Google Scholar - Lepetit, L., Cheriaux, G. & Joffre, M. Linear techniques of phase measurement by femtosecond spectral interferometry for applications in spectroscopy. J. Opt. Soc. Am. B 104, 2467–2474 (1995)
Article ADS Google Scholar - Hybl, J. D., Albrecht, A. W., Faeder, S. M. G. & Jonas, D. M. Two-dimensional electronic spectroscopy. Chem. Phys. Lett. 297, 307–313 (1998)
Article ADS CAS Google Scholar - Rice, S. A., Bergren, M. S., Beich, A. C. & Nielson, G. A theoretical analysis of the OH stretching spectra of ice Ih, liquid water, and amorphous solid water. J. Phys. Chem. 87, 4295–4308 (1983)
Article CAS Google Scholar - Wojcik, M. J., Buch, V. & Devlin, J. P. Spectra of isotopic ice mixtures. J. Chem. Phys. 99, 2332–2344 (1993)
Article ADS CAS Google Scholar - Lock, A. J. & Bakker, H. J. Temperature dependence of vibrational relaxation in liquid H2O. J. Chem. Phys. 117, 1708–1713 (2002)
Article ADS CAS Google Scholar - Pakoulev, A., Wang, Z. & Dlott, D. Vibrational relaxation and spectral evolution following ultrafast OH stretch excitation of water. Chem. Phys. Lett. 371, 594–600 (2003)
Article ADS CAS Google Scholar - Jimenez, R., Fleming, G. R., Kumar, P. V. & Maroncelli, M. Femtosecond solvation dynamics in water. Nature 369, 471–473 (1994)
Article ADS CAS Google Scholar - Castner, E. W. Jr, Chang, Y. J., Chu, Y. C. & Walrafen, G. E. The intermolecular dynamics of liquid water. J. Chem. Phys. 102, 653–659 (1995)
Article ADS CAS Google Scholar - Saito, S. & Ohmine, I. Third order nonlinear response of liquid water. J. Chem. Phys. 106, 4889–4893 (1997)
Article ADS CAS Google Scholar - Pohorille, A., Pratt, L. R., LaViolette, R. A., Wilson, M. A. & MacElroy, R. D. Comparison of the structure of harmonic aequous glasses and liquid water. J. Chem. Phys. 87, 6070–6077 (1987)
Article ADS CAS Google Scholar - Poulsen, J. A., Nyman, G. & Nordholm, S. Wave packet study of ultrafast relaxation in ice Ih and liquid water. Resonant intermolecular vibrational energy transfer. J. Phys. Chem. A 107, 8420–8428 (2003)
Article CAS Google Scholar
Acknowledgements
We thank F. Weik for help with the use of a thermal imaging camera. Financial support by the Deutsche Forschungsgemeinschaft, the Humboldt foundation (R.J.D.M.), the Canadian Foundation for Innovation, the Natural Sciences and Engineering Research Council of Canada, and Photonics Research Ontario is acknowledged.
Author information
Author notes
- M. L. Cowan, B. D. Bruner and N. Huse: These authors contributed equally to this work
Authors and Affiliations
- Departments of Chemistry and Physics, University of Toronto, 80 St George Street, Toronto, Ontario, M5S3H6, Canada
M. L. Cowan, B. D. Bruner, J. R. Dwyer, B. Chugh & R. J. D. Miller - Max-Born-Institut für Nichtlineare Optik und Kurzzeitspektroskopie, Max-Born-Strasse 2A, D-12489, Berlin, Germany
N. Huse, E. T. J. Nibbering & T. Elsaesser
Authors
- M. L. Cowan
- B. D. Bruner
- N. Huse
- J. R. Dwyer
- B. Chugh
- E. T. J. Nibbering
- T. Elsaesser
- R. J. D. Miller
Corresponding author
Correspondence toR. J. D. Miller.
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Notes (download DOC )
This material describes control experiments illustrating the performance of our system. Supplementary Figure 1 illustrates the effects of isotopic substitution on the relaxation dynamics of liquid water. Supplementary Figure 2 shows that ultrathin Si3N4 windows eliminate nonlinear window signals. This file also contains additional references. (DOC 141 kb)
Rights and permissions
About this article
Cite this article
Cowan, M., Bruner, B., Huse, N. et al. Ultrafast memory loss and energy redistribution in the hydrogen bond network of liquid H2O.Nature 434, 199–202 (2005). https://doi.org/10.1038/nature03383
- Received: 18 October 2004
- Accepted: 21 January 2005
- Issue date: 10 March 2005
- DOI: https://doi.org/10.1038/nature03383