Inhibition of quantal release from motor nerve by wortmannin - PubMed (original) (raw)
Inhibition of quantal release from motor nerve by wortmannin
S J Hong et al. Br J Pharmacol. 1999 Sep.
Abstract
1. The effects of wortmannin, an inhibitor of phosphatidylinositol (PI) kinases and myosin light chain kinase, on the quantal release of neurotransmitter from mouse phrenic nerve were investigated. 2. Wortmannin (10 - 100 microM) initially enhanced, thereafter progressively depressed spontaneous quantal discharge (miniature endplate potential, mepp). The mean amplitude and the amplitude distribution of mepp were not altered. 3. The compound inhibited and prevented the intensive quantal release evoked by high KC1 solution as well as the mepp burst induced by alpha-latrotoxin, a polypeptide toxin that possesses Ca2+-independent synaptic action to trigger quantal release. The inhibitory actions of wortmannin were partially reversible. 4. Wortmannin depressed the amplitude of endplate potentials (epps) and increased the coefficient of variance of epps. The profile of epps in response to high frequency nerve stimulation exhibited fluctuations between run-down and run-up. The phenomenon is thus different from the consistency of run-up characteristic as the motor nerve Ca2+ channel is blocked by omega-agatoxin IVA. 5. LY294002, another inhibitor of PI 3-kinase, raised mepp frequency without causing late phase suppressions. The compound did not inhibit KC1-, alpha-latrotoxin- or nerve stimulation-evoked quantal release. 6. The results suggest that wortmannin could depress quantal release beyond the step of Ca2+ channel blockade, probably by interfering with the exocytotic cascade.
Figures
Figure 1
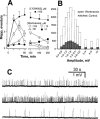
Effects of wortmannin and LY294002 on the spontaneous quantal discharge. Isolated phrenic nerve-diaphragms were incubated in Tyrode solution. (A) Time-dependent actions of wortmannin and of LY294002 on the firing of miniature endplate potentials (mepps). (B) mepp amplitude distribution histograms before or 20–30 min after treatment with wortmannin. (C) Traces of mepps from different endplates of the same preparation in the absence (top panel), or treated with wortmannin 30 μ
M
for 20 min (middle), or for 220 min (bottom). Resting membrane potentials were −76∼–82 mV. Data in (A and B) were pooled from 21–32 endplates, _n_=4–6.
Figure 2
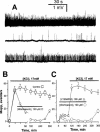
Wortmannin, but not LY294002, inhibits quantal discharge evoked by high KC1 solution. (A) The preparation was incubated in high KC1 (17 m
M
) Tyrode solution for 20 min (top panel) and then further treated with wortmannin 100 μ
M
for 220 min (middle). The bottom trace was 120 min after washout of wortmannin. Traces from different endplates. (B) Inhibition time course. Preparations were incubated in high KC1 solution in the absence or presence of wortmannin as indicated by the respective bars. The total incubation time for wortmannin was 220 min. (C) Prevention of high KC1 solution-evoked quantal release by wortmannin. Preparations were firstly treated with wortmannin or LY294002 for 90 min and then KC1 concentration was raised to 17 m
M
(total incubation time 270 min) as annotated. The total incubation duration for wortmannin and LY294002 was 240 min. Resting membrane potentials were −60∼−64 mV. Data in (B and C) were pooled from 22–27 endplates, _n_=5.
Figure 3
Inhibition of α-latrotoxin-induced mepp burst by wortmannin. (A) The preparation was treated with α-latrotoxin 3 n
M
for 20 min (top panel), and 40 min later further with wortmannin 100 μ
M
for 220 min (bottom, a different endplate). A part of mepp burst is displayed with a 200 fold expansion of the time scale (middle panel). (B) The preparation was first treated with wortmannin 100 μ
M
for 220 min and then further with α-latrotoxin 10 n
M
for 40 min (top panel). Bottom panel shows a segment of recording continued from top panel 120 min after washout of α-latrotoxin and wortmannin. Note the significant increase of mepps and partial restoration of mepp burst. Resting membrane potentials were −70∼−82 mV.
Figure 4
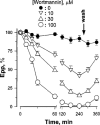
Dose- and time-dependent actions of wortmannin on endplate potentials. Phrenic nerve was stimulated at 0.3 Hz, and 20 endplate potentials (epps) were averaged for each endplate. Wortmannin was washed out at 250 min. Data were pooled from 21–30 endplates, _n_=4–6.
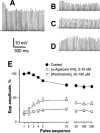
Figure 5
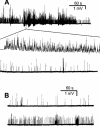
Effects of wortmannin on epps evoked by high frequency stimulation. Phrenic nerve was stimulated at 50 Hz for 4 s in the absence (A), or presence of wortmannin 100 μ
M
treated for 30–40 min (B–D). Shown are typical patterns of epps with run-down (B), flat (C) or run-up (D). (E) is the overall statistical epp profile of control, after treatment with wortmannin for 30–180 min, or after treatment with ω-agatoxin IVA for 20–40 min. Data were pooled from 19–37 endplates, (_n_=4–6). Resting membrane potentials were −76∼−82 mV.
Similar articles
- Physiological and regenerative acetylcholine release from motor nerve: differential inhibitions by vesamicol and omega-agatoxin IVA.
Hong SJ, Lee SH, Chang CC. Hong SJ, et al. Neuroscience. 1995 Jul;67(1):169-75. doi: 10.1016/0306-4522(95)00046-l. Neuroscience. 1995. PMID: 7477897 - Inhibition of acetylcholine release from mouse motor nerve by a P-type calcium channel blocker, omega-agatoxin IVA.
Hong SJ, Chang CC. Hong SJ, et al. J Physiol. 1995 Jan 15;482 ( Pt 2)(Pt 2):283-90. doi: 10.1113/jphysiol.1995.sp020517. J Physiol. 1995. PMID: 7714822 Free PMC article. - Coupling of L-type calcium channels to neurotransmitter release at mouse motor nerve terminals.
Urbano FJ, Depetris RS, Uchitel OD. Urbano FJ, et al. Pflugers Arch. 2001 Mar;441(6):824-31. doi: 10.1007/s004240000489. Pflugers Arch. 2001. PMID: 11316267 - Relative potencies of metal ions on transmitter release at mouse motor nerve terminals.
Porter VA, Wray D. Porter VA, et al. Br J Pharmacol. 1996 May;118(1):27-32. doi: 10.1111/j.1476-5381.1996.tb15362.x. Br J Pharmacol. 1996. PMID: 8733572 Free PMC article. - P-type Ca2+ channels trigger stimulus-evoked [3H]acetylcholine release from mammalian motor endplates.
Wessler I, Dooley DJ, Lohr B. Wessler I, et al. Eur J Pharmacol. 1995 May 4;278(1):83-6. doi: 10.1016/0014-2999(95)00133-6. Eur J Pharmacol. 1995. PMID: 7664817
Cited by
- Inhibition of mouse neuromuscular transmission and contractile function by okadaic acid and cantharidin.
Hong SJ. Hong SJ. Br J Pharmacol. 2000 Jul;130(6):1211-8. doi: 10.1038/sj.bjp.0703418. Br J Pharmacol. 2000. PMID: 10903957 Free PMC article. - Capsaicin modulates acetylcholine release at the myoneural junction.
Thyagarajan B, Potian JG, Baskaran P, McArdle JJ. Thyagarajan B, et al. Eur J Pharmacol. 2014 Dec 5;744:211-9. doi: 10.1016/j.ejphar.2014.09.044. Epub 2014 Oct 23. Eur J Pharmacol. 2014. PMID: 25446918 Free PMC article. - Phosphatidylinositol 3-kinase C2alpha is essential for ATP-dependent priming of neurosecretory granule exocytosis.
Meunier FA, Osborne SL, Hammond GR, Cooke FT, Parker PJ, Domin J, Schiavo G. Meunier FA, et al. Mol Biol Cell. 2005 Oct;16(10):4841-51. doi: 10.1091/mbc.e05-02-0171. Epub 2005 Jul 29. Mol Biol Cell. 2005. PMID: 16055506 Free PMC article. - Importance of Full-Collapse Vesicle Exocytosis for Synaptic Fatigue-Resistance at Rat Fast and Slow Muscle Neuromuscular Junctions.
Rudling JE, Drever BD, Reid B, Bewick GS. Rudling JE, et al. Int J Mol Sci. 2018 Jul 2;19(7):1936. doi: 10.3390/ijms19071936. Int J Mol Sci. 2018. PMID: 30004407 Free PMC article. - Plasmalemmal phosphatidylinositol-4,5-bisphosphate level regulates the releasable vesicle pool size in chromaffin cells.
Milosevic I, Sørensen JB, Lang T, Krauss M, Nagy G, Haucke V, Jahn R, Neher E. Milosevic I, et al. J Neurosci. 2005 Mar 9;25(10):2557-65. doi: 10.1523/JNEUROSCI.3761-04.2005. J Neurosci. 2005. PMID: 15758165 Free PMC article.
References
- BROADIE K.S. Genetic dissection of the molecular mechanisms of transmitter vesicle release during synaptic transmission. J. Physiol., (Paris) 1995;89:59–70. - PubMed
- BURKE E.P., GERTHOFFER W.T., SANDERS K.M., PUBLICOVER N.G. Wortmannin inhibits contraction without altering electrical activity in canine gastric smooth muscle. Am. J. Physiol. 1996;270:C1405–C1412. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous