Distinct proinflammatory host responses to Neisseria gonorrhoeae infection in immortalized human cervical and vaginal epithelial cells - PubMed (original) (raw)
Distinct proinflammatory host responses to Neisseria gonorrhoeae infection in immortalized human cervical and vaginal epithelial cells
R N Fichorova et al. Infect Immun. 2001 Sep.
Abstract
In this study we utilized immortalized morphologically and functionally distinct epithelial cell lines from normal human endocervix, ectocervix, and vagina to characterize gonococcal epithelial interactions pertinent to the lower female genital tract. Piliated, but not nonpiliated, N. gonorrhoeae strain F62 variants actively invaded these epithelial cell lines, as demonstrated by an antibiotic protection assay and confocal microscopy. Invasion of these cells by green fluorescent protein-expressing gonococci was characterized by colocalization of gonococci with F actin, which were initially detected 30 min postinfection. In all three cell lines, upregulation of interleukin 8 (IL-8) and IL-6, intercellular adhesion molecule 1 (CD54), and the nonspecific cross-reacting antigen (CD66c) were detected 4 h after infection with piliated and nonpiliated gonococci. Furthermore, stimulation of all three cell lines with gonococcal whole-cell lysates resulted in a similar upregulation of IL-6 and IL-8, confirming that bacterial uptake is not essential for this response. Increased levels of IL-1 were first detected 8 h after infection with gonococci, suggesting that the earlier IL-8 and IL-6 responses were not mediated through the IL-1 signaling pathway. The IL-1 response was limited to cultures infected with piliated gonococci and was more vigorous in the endocervical epithelial cells. The ability of gonococci to stimulate distinct proinflammatory host responses in these morphologically and functionally different compartments of the lower female genital tract may contribute directly to the inflammatory signs and symptoms characteristic of disease caused by N. gonorrhoeae.
Figures
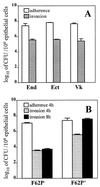
FIG. 1
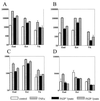
Interactions of N. gonorrhoeae with human cervical and vaginal epithelial cells. (A) Adherence of the N. gonorrhoeae F62 piliated variant to and invasion of the endocervical (End), ectocervical (Ect), and vaginal (Vk) immortalized epithelial cell lines 4 h postinfection. (B) Adherence and invasion of nonpiliated (F62P−) and piliated (F62P+) N. gonorrhoeae F62 following 4 and 8 h infection of endocervical epithelial cells. Values are means plus standard deviations of logarithmically transformed determinations obtained from three independent experiments.
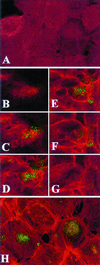
FIG. 2
Confocal microscopy analysis of endocervical epithelial cell monolayers infected with GFP-expressing N. gonorrhoeae. F actin was visualized using red fluorescent Alexa-Fluor. Images were obtained by combining a Z series of 15 optical sections taken at 2-μm intervals. The slides were consecutively read for red and green fluorescence using 485- and 568-nm filters, respectively. (A) Control uninfected culture. Diffuse red staining indicates lack of F-actin polymerization. (B to G) Single apical-basal confocal slices taken in consecutive order (, , , , , and optical sections) from a Z series 30 min postinfection. The Alexa-Fluor labeling (C to G) demonstrates increased cortical polymerization of F actin in most epithelial cells. The GFP Alexa-Fluor overlap (yellow color [C to F]) indicates colocalization of F actin with gonococci. (H) Composite image of a 15-section Z series 4 h postinfection. GFP-expressing gonococci appear in large round clusters within the infected epithelial cells (typically seen between the sixth and tenth optical sections). Polymerized actin bundles extend toward the middle of infected epithelial cells.
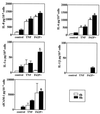
FIG. 3
Levels of IL-8, IL-6, IL-1β, IL-1α, and sICAM-1 following infection of endocervical cells with N. gonorrhoeae. Culture supernatants were collected 4 and 8 h after incubation under the following conditions: uninfected nonstimulated (control), uninfected stimulated with TNF-α, and infected with the N. gonorrhoeae F62 piliated variant (F62P+) expressing GFP. Values are means plus standard deviations of quadruplicate determinations and are representative of two independent experiments. ∗, P < 0.05 versus nonstimulated control.
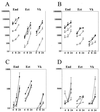
FIG. 4
Levels of IL-8 (A), IL-6 (B), IL-1α (C), and sICAM-1 (D) in supernatants collected from endocervical (End), ectocervical (Ect), and vaginal (Vk) epithelial cell cultures during a 24-h time course of infection with N. gonorrhoeae. Data (picograms per 106 epithelial cells) are means plus standard deviations of duplicate determinations obtained in two independent experiments for each cell line. Closed symbol represent levels of mediators in cultures infected with piliated (circles) and nonpiliated (triangles) N. gonorrhoeae strain F62. Controls included uninfected nonstimulated (open circles) and TNF-α-stimulated cultures (open triangles) examined at the same time points.
FIG. 5
Levels of IL-8 (A), IL-6 (B), IL-1α (C), and sICAM-1 (D) in 24-h supernatants from endocervical (End), ectocervical (Ect), and vaginal (Vk) epithelial cell cultures exposed to whole-cell lysates obtained from N. gonorrhoeae F62 piliated (F62P+) and nonpiliated (F62P−) variants. Controls include uninfected unstimulated cultures (control) and TNF-α-stimulated cultures (TNFα) examined at the same time point (24 h). Values (picograms per 106 epithelial cells) are means plus standard deviations of triplicate determinations for each cell line and represent two independent experiments.
FIG. 6
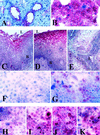
Immunocytochemical analysis of adhesion molecule expression by cervicovaginal epithelial cells in vitro and in vivo. Positive cells appear red. (A and B) Constitutive expression of CD46 in endocervical tissue (A) and endocervical cell culture (B). (C to G) Expression of CD66 in vaginal tissue (C), ectocervical tissue (D), endocervical tissue (E), uninfected endocervical cell culture (F), and endocervical cell culture after 8 h of infection with the N. gonorrhoeae F62 piliated variant (G). (H to K) Expression of ICAM-1 in endocervical cell cultures with no infection (H), following 8 h of TNF-α stimulation (I), following 8 h of infection with N. gonorrhoeae piliated F62 (J), or following 8 h of infection with N. gonorrhoeae nonpiliated F62 (K). L. lumenal epithelial surface: B. basal epithelial layers. Magnification, ×125 (A, C to E, and H to K) and ×250 (B, F, and G).
Similar articles
- Response to Neisseria gonorrhoeae by cervicovaginal epithelial cells occurs in the absence of toll-like receptor 4-mediated signaling.
Fichorova RN, Cronin AO, Lien E, Anderson DJ, Ingalls RR. Fichorova RN, et al. J Immunol. 2002 Mar 1;168(5):2424-32. doi: 10.4049/jimmunol.168.5.2424. J Immunol. 2002. PMID: 11859134 - Fibroblast growth factor-inducible 14 (Fn14) is expressed in the lower genital tract and may play a role in amplifying inflammation during infection.
Han ES, Mekasha S, Ingalls RR. Han ES, et al. J Reprod Immunol. 2010 Jan;84(1):16-23. doi: 10.1016/j.jri.2009.09.009. Epub 2009 Dec 5. J Reprod Immunol. 2010. PMID: 19963275 Free PMC article. - CEACAM is not necessary for Neisseria gonorrhoeae to adhere to and invade female genital epithelial cells.
Swanson KV, Jarvis GA, Brooks GF, Barham BJ, Cooper MD, Griffiss JM. Swanson KV, et al. Cell Microbiol. 2001 Oct;3(10):681-91. doi: 10.1046/j.1462-5822.2001.00147.x. Cell Microbiol. 2001. PMID: 11580753 - The molecular mechanisms used by Neisseria gonorrhoeae to initiate infection differ between men and women.
Edwards JL, Apicella MA. Edwards JL, et al. Clin Microbiol Rev. 2004 Oct;17(4):965-81, table of contents. doi: 10.1128/CMR.17.4.965-981.2004. Clin Microbiol Rev. 2004. PMID: 15489357 Free PMC article. Review. - Pathogenesis of Neisseria gonorrhoeae in the female reproductive tract: neutrophilic host response, sustained infection, and clinical sequelae.
Stevens JS, Criss AK. Stevens JS, et al. Curr Opin Hematol. 2018 Jan;25(1):13-21. doi: 10.1097/MOH.0000000000000394. Curr Opin Hematol. 2018. PMID: 29016383 Free PMC article. Review.
Cited by
- Exposure to HIV-1 directly impairs mucosal epithelial barrier integrity allowing microbial translocation.
Nazli A, Chan O, Dobson-Belaire WN, Ouellet M, Tremblay MJ, Gray-Owen SD, Arsenault AL, Kaushic C. Nazli A, et al. PLoS Pathog. 2010 Apr 8;6(4):e1000852. doi: 10.1371/journal.ppat.1000852. PLoS Pathog. 2010. PMID: 20386714 Free PMC article. - Adhesion and invasion of gingival epithelial cells by Porphyromonas gulae.
Inaba H, Nomura R, Kato Y, Takeuchi H, Amano A, Asai F, Nakano K, Lamont RJ, Matsumoto-Nakano M. Inaba H, et al. PLoS One. 2019 Mar 14;14(3):e0213309. doi: 10.1371/journal.pone.0213309. eCollection 2019. PLoS One. 2019. PMID: 30870452 Free PMC article. - Effects of feminine hygiene products on the vaginal mucosal biome.
Fashemi B, Delaney ML, Onderdonk AB, Fichorova RN. Fashemi B, et al. Microb Ecol Health Dis. 2013 Feb 25;24. doi: 10.3402/mehd.v24i0.19703. eCollection 2013. Microb Ecol Health Dis. 2013. PMID: 24009546 Free PMC article. - The ironclad truth: how in vivo transcriptomics and in vitro mechanistic studies shape our understanding of Neisseria gonorrhoeae gene regulation during mucosal infection.
Moreau MR, Massari P, Genco CA. Moreau MR, et al. Pathog Dis. 2017 Jul 31;75(5):ftx057. doi: 10.1093/femspd/ftx057. Pathog Dis. 2017. PMID: 28520925 Free PMC article. Review. - Glycerol monolaurate does not alter rhesus macaque (Macaca mulatta) vaginal lactobacilli and is safe for chronic use.
Schlievert PM, Strandberg KL, Brosnahan AJ, Peterson ML, Pambuccian SE, Nephew KR, Brunner KG, Schultz-Darken NJ, Haase AT. Schlievert PM, et al. Antimicrob Agents Chemother. 2008 Dec;52(12):4448-54. doi: 10.1128/AAC.00989-08. Epub 2008 Oct 6. Antimicrob Agents Chemother. 2008. PMID: 18838587 Free PMC article.
References
- Anderson R M. Transmission dynamics of sexually transmitted infections. In: Holmes K K, Sparling P F, Mardh P A, Lemon S M, Satm W E, Piot P, Wasserheit J N, editors. Sexually transmitted diseases. New York, N.Y: McGraw-Hill; 1999. pp. 25–37.
- Christodoulides M, Everson J S, Liu B L, Lambden P R, Watt P J, Thomas E J, Heckels J E. Interaction of primary human endometrial cells with Neisseria gonorrhoeae expressing green fluorescent protein. Mol Microbiol. 2000;35:32–43. - PubMed
- Crowe S E, Alvarez L, Dytoc M, Hunt R H, Miller M, Sherman P, Patel J, Jin Y, Ernst P B. Expression of interleukin 8 and CD54 by human gastric epithelium after Helicobacter pylori infection in vitro. Gastroenterology. 1995;108:65–74. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical